Abstract
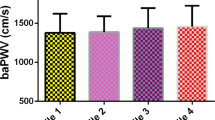
This study was performed to investigate and compare the association of albuminuria with the brachial–ankle pulse wave velocity (baPWV) and carotid–femoral pulse wave velocity (cfPWV) in a community-based population in Beijing. Subjects were enrolled from a follow-up survey conducted in 2018 from an atherosclerosis cohort in Shijingshan district, Beijing, China. The baPWV and cfPWV were measured using a BP-203 RPE III arteriosclerosis detection device and PulsePen, respectively. Albuminuria was defined as a urinary albumin–creatinine ratio of ≥30 mg/g. A multivariate logistic regression model was used to evaluate the impacts of different PWV measurements on the prevalence of albuminuria. In total, 5605 subjects were included in the analyses. Their mean age was 62.22 ± 7.55 years, and the prevalence of albuminuria was 8.22%. In the multivariate logistic regression model adjusted for potential covariates, both baPWV (odds ratio [OR], 1.06; 95% confidence interval [CI], 1.03–1.10; P < 0.001) and cfPWV (OR, 1.07; 95% CI, 1.01–1.14; P = 0.018) were significantly associated with albuminuria. Furthermore, when baPWV and cfPWV were entered into the logistic regression model simultaneously, only baPWV was significantly associated with albuminuria using either continuous value (OR, 1.05; 95% CI, 1.01–1.10; P = 0.007) or classified into quartiles (highest vs. lowest value group: OR, 1.55; 95% CI, 1.01–2.37; P for trend = 0.019). Both cfPWV and baPWV were positively associated with albuminuria, while baPWV had a stronger relationship than cfPWV. Thus, baPWV measurement could be considered for the purpose of community health screening.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
Data availability
The data used to support the findings of this study are available from the corresponding author upon request.
References
Ballew SH, Matsushita K. Cardiovascular risk prediction in CKD. Semin Nephrol. 2018;38:208–16.
Nowak KL, Chonchol M, Jovanovich A, You Z, Ambrosius WT, Cho ME, et al. Arterial stiffness and kidney disease progression in the systolic blood pressure intervention trial. Clin Nephrol. 2020;94:26–35.
Cavalcante JL, Lima JA, Redheuil A, Al-Mallah MH. Aortic stiffness: current understanding and future directions. J Am Coll Cardiol. 2011;57:1511–22.
Mattace-Raso FU, van der Cammen TJ, Hofman A, van Popele NM, Bos ML, Schalekamp MA, et al. Arterial stiffness and risk of coronary heart disease and stroke: the Rotterdam Study. Circulation 2006;113:657–63.
Aiumtrakul N, Supasyndh O, Krittayaphong R, Phrommintikul A, Satirapoj B. Cardio-ankle vascular index with renal progression and mortality in high atherosclerosis risk: a prospective cohort study in CORE-Thailand. Clin Exp Nephrol 2021; https://doi.org/10.1007/s10157-021-02149-x.
Ohkuma T, Ninomiya T, Tomiyama H, Kario K, Hoshide S, Kita Y, et al. Brachial–ankle pulse wave velocity and the risk prediction of cardiovascular disease: an individual participant data meta-analysis. Hypertension 2017;69:1045–52.
Williams B, Mancia G, Spiering W, Agabiti Rosei E, Azizi M, Burnier M, et al. 2018 ESC/ESH guidelines for the management of arterial hypertension: the Task Force for the management of arterial hypertension of the European Society of Cardiology and the European Society of Hypertension J Hypertens. 2018;36:1953–2041.
Joint Committee for Guideline Revision. 2018 Chinese guidelines for prevention and treatment of hypertension—a report of the Revision Committee of Chinese Guidelines for Prevention and Treatment of Hypertension. J Geriatr Cardiol. 2019;16:182–241.
Meyer M, Tanaka H, Palta P, Patel M, Camplain R, Couper D, et al. Repeatability of central and peripheral pulse wave velocity measures: the Atherosclerosis Risk in Communities (ARIC) Study. Am J Hypertens. 2016;29:470–5.
Yu WC, Chuang SY, Lin YP, Chen CH. Brachial–ankle vs. carotid–femoral pulse wave velocity as a determinant of cardiovascular structure and function. J Hum Hypertens. 2008;22:24–31.
Sugawara J, Hayashi K, Yokoi T, Cortez-Cooper MY, DeVan AE, Anton MA, et al. Brachial–ankle pulse wave velocity: an index of central arterial stiffness? J Hum Hypertens. 2005;19:401–6.
Vasan RS, Short MI, Niiranen TJ, Xanthakis V, DeCarli C, Cheng S, et al. Interrelations between arterial stiffness, target organ damage, and cardiovascular disease outcomes. J Am Heart Assoc. 2019;8:e012141.
Coutinho T, Turner ST, Kullo IJ. Aortic pulse wave velocity is associated with measures of subclinical target organ damage. JACC Cardiovasc Imaging. 2011;4:754–61.
Kim ED, Tanaka H, Ballew SH, Sang Y, Heiss G, Coresh J, et al. Associations between kidney disease measures and regional pulse wave velocity in a large community-based cohort: the Atherosclerosis Risk in Communities (ARIC) Study. Am J Kidney Dis. 2018;72:682–90.
Lu Y, Zhu M, Bai B, Chi C, Yu S, Teliewubai J, et al. Comparison of carotid–femoral and brachial–ankle pulse-wave velocity in association with target organ damage in the community-dwelling elderly chinese: the Northern Shanghai Study. J Am Heart Assoc. 2017;6:e004168.
Fan F, Qi L, Jia J, Xu X, Liu Y, Yang Y, et al. Noninvasive central systolic blood pressure is more strongly related to kidney function decline than peripheral systolic blood pressure in a Chinese community-based population. Hypertension 2016;67:1166–72.
Levey AS, Stevens LA, Schmid CH, Zhang YL, Castro AF, Feldman HI, et al. A new equation to estimate glomerular filtration rate. Ann Intern Med. 2009;150:604–12.
Salvi P, Lio G, Labat C, Ricci E, Pannier B, Benetos A. Validation of a new non-invasive portable tonometer for determining arterial pressure wave and pulse wave velocity: the PulsePen device. J Hypertens. 2004;22:2285–93.
Cândido JSA, Camelo LV, Mill JG, Lotufo PA, Ribeiro ALP, Duncan BB, et al. Greater aortic stiffness is associated with renal dysfunction in participants of the ELSA-Brasil cohort with and without hypertension and diabetes. PLoS ONE. 2019;14:e0210522.
O’Rourke MF, Safar ME. Relationship between aortic stiffening and microvascular disease in brain and kidney: cause and logic of therapy. Hypertension 2005;46:200–4.
Zhang Y, Lacolley P, Protogerou AD, Safar ME. Arterial stiffness in hypertension and function of large arteries. Am J Hypertens. 2020;33:291–6.
Wu S, Chen D, Zeng X, Wen J, Zhou C, Xiao K, et al. Arterial stiffness is associated with target organ damage in subjects with pre-hypertension. Arch Med Sci. 2018;14:1374–80.
Zhang X, Low S, Sum CF, Tavintharan S, Yeoh LY, Liu J, et al. Arterial stiffness is an independent predictor for albuminuria progression among Asians with type 2 diabetes—a prospective cohort study. J Diabetes Complicat. 2017;31:933–8.
Huang N, Foster MC, Mitchell GF, Andresdottir MB, Eiriksdottir G, Gudmundsdottir H, et al. Aortic stiffness and change in glomerular filtration rate and albuminuria in older people. Nephrol Dial Transpl. 2017;32:677–84.
Munakata M, Nunokawa T, Yoshinaga K, Toyota T. Pulse wave velocity is an independent risk factor for microalbuminuria in patients with essential hypertension—a Japanese Trial on the Prognostic Implication of Pulse Wave V elocity (J-TOPP). Hypertens Res. 2006;29:515–21.
Kim JH, Kim SS, Kim IJ, Kim BH, Park JY, Lee CW, et al. Arterial stiffness is more associated with albuminuria than decreased glomerular filtration rate in patients with type 2 diabetes mellitus: the REBOUND Study. J Diabetes Res. 2017;2017:7047909.
Jiang Y, Fan F, Jia J, He D, Sun P, Wu Z, et al. Brachial–ankle pulse wave velocity is independently associated with urine albumin-to-creatinine ratio in a Chinese community-based cohort. Int Urol Nephrol. 2020;52:713–20.
Ishikawa T, Hashimoto J, Morito RH, Hanazawa T, Aikawa T, Hara A, et al. Association of microalbuminuria with brachial–ankle pulse wave velocity: the Ohasama study. Am J Hypertens. 2008;21:413–8.
Bulas J, Potocarova M, Kupcova V, Gaspar L, Wimmer G, Murin J. Central systolic blood pressure increases with aortic stiffness. Bratisl Lek Listy. 2019;120:894–8.
Kitagawa N, Okada H, Tanaka M, Hashimoto Y, Kimura T, Nakano K, et al. Which measurement of blood pressure is more associated with albuminuria in patients with type 2 diabetes: central blood pressure or peripheral blood pressure? J Clin Hypertens. 2016;18:790–5.
Peralta CA, Katz R, Madero M, Sarnak M, Kramer H, Criqui MH, et al. The differential association of kidney dysfunction with small and large arterial elasticity: the multiethnic study of atherosclerosis. Am J Epidemiol. 2009;169:740–8.
Munakata M, Miura Y, Yoshinaga K. Higher brachial–ankle pulse wave velocity as an independent risk factor for future microalbuminuria in patients with essential hypertension: the J-TOPP study. J Hypertens. 2009;27:1466–71.
Munakata M. Brachial–ankle pulse wave velocity: a most predictable arterial measure for cerebral small vessel disease. J Atheroscler Thromb. 2020;27:919–21.
Zhang K, Jiang Y, Wang Y, Suo C, Xu K, Zhu Z, et al. Associations of arterial stiffness and carotid atherosclerosis with cerebral small vessel disease in a rural community-based population. J Atheroscler Thromb. 2020;27:922–33.
Chen YT, Hsu HJ, Hsu CK, Lee CC, Hsu KH, Sun CY, et al. Correlation between spot and 24h proteinuria: derivation and validation of equation to estimate daily proteinuria. PLoS ONE. 2019;14:e0214614.
Acknowledgements
We are very grateful to the staff of the Gucheng and Pingguoyuan Community Health Centers and the research coordinators who participated in this cohort study. This work was supported by funding from the following: UMHS-PUHSC Joint Institute for Translational and Clinical Research and the Fundamental Research Funds for the Central Universities (BMU20110177, BMU20160530); National Key Research and Development Program of China (2017YFC1307704); Scientific Research Seed Fund of Peking University First Hospital (2018SF003, 2018SF071, 2018SF073); Projects of National Natural Science Foundation of China (81703288); Key Laboratory of Molecular Cardiovascular Sciences(Peking University), Ministry of Education and NHC Key Laboratory of Cardiovascular Molecular Biology and Regulatory Peptides.
Author information
Authors and Affiliations
Contributions
TW was responsible for conducting the search, screening potentially eligible studies, interpreting results, updating reference lists and writing the paper; FF was responsible for conducting the search, extracting and analyzing data, interpreting results; YG contributed to screening potentially eligible studies; JJ contributed to writing the protocol and report, conducting the search, writing the funding resources; LG, ZL, ML, and YJ contributed to data extraction and provided feedback on the report; YZ and JL was responsible for designing the review protocol, screening potentially eligible studies, interpreting results and revising the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval
This research protocol was approved by the Biomedical Research Ethics Committee of Peking University First Hospital.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Wang, T., Fan, F., Gong, Y. et al. Comparison of brachial–ankle pulse wave velocity and carotid–femoral pulse wave velocity in association with albuminuria in a community of Beijing: a cross-sectional study. J Hum Hypertens 37, 412–418 (2023). https://doi.org/10.1038/s41371-022-00697-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41371-022-00697-7