Abstract
Background and aims
Randomized clinical trials have proven the efficacy and safety of Food and Drug Administration (FDA) approved anti-obesity medications (AOMs) for long-term use. It is unclear whether these outcomes can be replicated in real-world clinical practice where clinical complexities arise. The aim of this study was to evaluate the effectiveness and side effects of these medications in real-world multidisciplinary clinical practice settings.
Methods
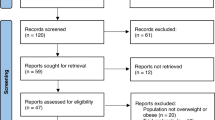
We reviewed the electronic medical records (EMR) of patients with obesity who were prescribed an FDA-approved AOM for long-term use in academic and community multidisciplinary weight loss programs between January 2016 and January 2020.
Intervention
We assessed percentage total body weight loss (%TBWL), metabolic outcomes, and side effect profile up to 24 months after AOM initiation.
Results
The full cohort consisted of 304 patients (76% women, 95.2% White, median age of 50 years old [IQR, 39–58]). The median follow-up time was 9.1 months [IQR, 4.2–14.1] with a median number of 3 visits [IQR, 2–4]. The most prescribed medication was phentermine/topiramate extended-release (ER) (51%), followed by liraglutide (26.3%), bupropion/naltrexone sustained-release (SR) (16.5%), and lorcaserin (6.2%). %TBWL was 5.0%, 6.8%, 9.3%, 10.3%, and 10.5% at 3, 6, 12, 18, and 24 months. 60.2% of the entire cohort achieved at least 5% TBWL. Overall, phentermine/topiramate-ER had the most robust weight loss response during follow-up, with the highest %TBWL at 12 months of 12.0%. Adverse events were reported in 22.4% of patients. Only 9% of patients discontinued the medication due to side effects.
Conclusions
AOMs resulted in significant long-term weight loss, that was comparable to outcomes previously reported in clinical trials.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Ogden CL, Carroll MD, Fryar CD, Flegal KM. Prevalence of obesity among adults and youth: United States, 2011–2014. NCHS Data Brief. 2015;219:1–8.
Waters H, Graf M. America’s obesity crisis. The Health and Economic Costs of Excess Weight. Santa Monica, California: Milken Institute; 2018.
Su W, Huang J, Chen F, Iacobucci W, Mocarski M, Dall TM, et al. Modeling the clinical and economic implications of obesity using microsimulation. J Med Econ. 2015;18:886–97.
Kim DD, Basu A. Estimating the medical care costs of obesity in the United States: systematic review, meta-analysis, and empirical analysis. Value Health. 2016;19:602–13.
Heymsfield SB, Wadden TA. Mechanisms, pathophysiology, and management of obesity. N Engl J Med. 2017;376:1492.
Acosta A, Streett S, Kroh MD, Cheskin LJ, Saunders KH, Kurian M, et al. White paper AGA: POWER - practice guide on obesity and weight management, education, and resources. Clin Gastroenterol Hepatol. 2017;15:631–49. e10.
Jensen MD, Ryan DH, Apovian CM, Ard JD, Comuzzie AG, Donato KA, et al. 2013 AHA/ACC/TOS guideline for the management of overweight and obesity in adults: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines and The Obesity Society. J Am Coll Cardiol. 2014;63:2985–3023.
Apovian CM, Aronne LJ, Bessesen DH, McDonnell ME, Murad MH, Pagotto U, et al. Pharmacological management of obesity: an endocrine Society clinical practice guideline. J Clin Endocrinol Metab. 2015;100:342–62.
Sharretts J, Galescu O, Gomatam S, Andraca-Carrera E, Hampp C, Yanoff L. Cancer risk associated with lorcaserin - the FDA’s review of the CAMELLIA-TIMI 61 trial. N Engl J Med. 2020;383:1000–2.
CllinicalTrials.gov. A research study of NNC0165-1562 and semaglutide in people who are overweight or obese. 2020. https://clinicaltrials.gov/ct2/show/NCT03574584.
Lewis KH, Fischer H, Ard J, Barton L, Bessesen DH, Daley MF, et al. Safety and effectiveness of longer-term phentermine use: clinical outcomes from an electronic health record cohort. Obesity. 2019;27:591–602.
Gadde KM, Allison DB, Ryan DH, Peterson CA, Troupin B, Schwiers ML, et al. Effects of low-dose, controlled-release, phentermine plus topiramate combination on weight and associated comorbidities in overweight and obese adults (CONQUER): a randomised, placebo-controlled, phase 3 trial. Lancet. 2011;377:1341–52.
Smith SR, Weissman NJ, Anderson CM, Sanchez M, Chuang E, Stubbe S, et al. Multicenter, placebo-controlled trial of lorcaserin for weight management. N Engl J Med. 2010;363:245–56.
Greenway FL, Fujioka K, Plodkowski RA, Mudaliar S, Guttadauria M, Erickson J, et al. Effect of naltrexone plus bupropion on weight loss in overweight and obese adults (COR-I): a multicentre, randomised, double-blind, placebo-controlled, phase 3 trial. Lancet. 2010;376:595–605.
Pi-Sunyer X, Astrup A, Fujioka K, Greenway F, Halpern A, Krempf M, et al. A randomized, controlled trial of 3.0 mg oF Liraglutide in Weight Management. N Engl J Med. 2015;373:11–22.
Wadden TA, Hollander P, Klein S, Niswender K, Woo V, Hale PM, et al. Weight maintenance and additional weight loss with liraglutide after low-calorie-diet-induced weight loss: the SCALE Maintenance randomized study. Int J Obes. 2013;37:1443–51.
Astrup A, Carraro R, Finer N, Harper A, Kunesova M, Lean ME, et al. Safety, tolerability and sustained weight loss over 2 years with the once-daily human GLP-1 analog, liraglutide. Int J Obes. 2012;36:843–54.
Garvey WT, Ryan DH, Look M, Gadde KM, Allison DB, Peterson CA, et al. Two-year sustained weight loss and metabolic benefits with controlled-release phentermine/topiramate in obese and overweight adults (SEQUEL): a randomized, placebo-controlled, phase 3 extension study. Am J Clin Nutr. 2012;95:297–308.
Allison DB, Gadde KM, Garvey WT, Peterson CA, Schwiers ML, Najarian T. et al. Controlled-release phentermine/topiramate in severely obese adults: a randomized controlled trial (EQUIP). Obesity. 2012;20:330–42.
Khera R, Murad MH, Chandar AK, Dulai PS, Wang Z, Prokop LJ, et al. Association of pharmacological treatments for obesity with weight loss and adverse events: a systematic review and meta-analysis. JAMA. 2016;315:2424–34.
Carey TA, Stiles WB. Some problems with randomized controlled trials and some viable alternatives. Clin Psychol Psychother. 2016;23:87–95.
Grudell AB, Sweetser S, Camilleri M, Eckert DJ, Vazquez-Roque MI, Carlson PJ, et al. A controlled pharmacogenetic trial of sibutramine on weight loss and body composition in obese or overweight adults. Gastroenterology. 2008;135:1142–54.
Holgerson AA, Clark MM, Ames GE, Collazo-Clavell ML, Kellogg TA, Graszer KM, et al. Association of adverse childhood experiences and food addiction to bariatric surgery completion and weight loss outcome. Obes Surg. 2018;28:3386–92.
Gorgojo-Martínez JJ, Basagoiti-Carreño B, Sanz-Velasco A, Serrano-Moreno C, Almodóvar-Ruiz F. Effectiveness and tolerability of orlistat and liraglutide in patients with obesity in a real-world setting: The XENSOR Study. Int J Clin Pract. 2019;73:e13399.
Park JH, Kim JY, Choi JH, Park HS, Shin H-Y, Lee JM, et al. Effectiveness of liraglutide 3 mg for the treatment of obesity in a real-world setting without intensive lifestyle intervention. Int J Obes. 2021;45:776–86.
Piantanida E, Gallo D, Tanda ML. Liraglutide is an effective drug for the treatment of obesity also in real life. J Endocrinol Investig. 2020;43:1827–8.
Wharton S, Liu A, Pakseresht A, Nørtoft E, Haase CL, Mancini J, et al. Real-world clinical effectiveness of liraglutide 3.0 mg for weight management in Canada. Obesity. 2019;27:917–24.
Ferrari F, Fierabracci P, Salvetti G, Jaccheri R, Vitti J, Scartabelli G, et al. Weight loss effect of liraglutide in real-life: the experience of a single Italian obesity center. J Endocrinol Investig. 2020;43:1779–85.
Fakhreddine AY, Bagsic S, Fujioka K, Frenette CT. Safety and efficacy of pharmacologic weight loss in patients with cirrhosis. Obes Sci Pract. 2021;7:159–67.
Ganguly R, Tian Y, Kong SX, Hersloev M, Hobbs T, Smolarz BG, et al. Persistence of newer anti-obesity medications in a real-world setting. Diabetes Res Clin Pract. 2018;143:348–56.
Shibuya K, Ali KF, Ji X, Milinoivh A, Bauman J, Kattan MW, et al. The benefit of short-term weight loss with anti-obesity medications in real-world clinical practice. Endocr Pract. 2019;25:1022–8.
Elangovan A, Shah R, Smith ZL. Pharmacotherapy for obesity—trends using a population level national database. Obes Surg. 2021;31:1105–12.
Wilding JPH, Batterham RL, Calanna S, Davies M, Van Gaal LF, Lingvay I, et al. Once-weekly semaglutide in adults with overweight or obesity. N Engl J Med. 2021;384:989–1002.
Fidler MC, Sanchez M, Raether B, Weissman NJ, Smith SR, Shanahan WR, et al. A one-year randomized trial of lorcaserin for weight loss in obese and overweight adults: the BLOSSOM trial. J Clin Endocrinol Metab. 2011;96:3067–77.
Wadden TA, Foreyt JP, Foster GD, Hill JO, Klein S, O’Neil PM, et al. Weight loss with naltrexone SR/bupropion SR combination therapy as an adjunct to behavior modification: the COR-BMOD trial. Obesity. 2011;19:110–20.
Apovian CM, Aronne L, Rubino D, Still C, Wyatt H, Burns C, et al. A randomized, phase 3 trial of naltrexone SR/bupropion SR on weight and obesity-related risk factors (COR-II). Obesity. 2013;21:935–43.
Lei XG, Ruan JQ, Lai C, Sun Z, Yang X. Efficacy and safety of phentermine/topiramate in adults with overweight or obesity: a systematic review and meta-analysis. Obesity 2021;29:985–94.
Lewis KH, Fischer H, Ard J, Barton L, Bessesen DH, Daley MF, et al. Safety and effectiveness of longer-term phentermine use: clinical outcomes from an electronic health record cohort. Obesity. 2019;27:591–602.
Acknowledgements
We would like to thank Dr. Michael Jensen for his critical feedback on the reporting of the findings and the discussion.
Author information
Authors and Affiliations
Contributions
GC, DGI, KLS, AA, and MAM conceived and design the study. MCC, MS, DH, HAL, KS, MMC, KG, MSM, MC, BAD, MDH, AA, and M.A.M coordinated data contributions. GC, DGI, KLS, OGV, AC, LC, AA, MeS, MaS, MDH analyzed the data. LC, AC, AA, and MDH produced the figures and tables. All authors contributed to the revision of the first draft, reviewed and approved the final version of the paper.
Corresponding authors
Ethics declarations
Competing interests
MSM has research grants from Fresenius Kabi, Nestle, Realfood Blends, and VectivBio and is a consultant for Baxter. AA reports other from Gila Therapeutics, other from Phenomix Sciences, personal fees from Rhythm Pharmaceuticals, personal fees from General Mills, outside the submitted work. BAD has served as a consultant for Boston Scientific, Metamodix, BFKW, DyaMx, and USGI Medical, has received research support for Boston Scientific, Apollo Endosurgery, USGI, Spatz Medical, GI Dynamics, Caim Diagnostics, Aspire Bariatrics, and Medtronic, and has been a speaker for Johnson & Johnson, Endogastric Solutions, and Olympus. MC is a stockholder in Phenomix Sciences and Enterin and serves as a consultant to Takeda, Allergan, Rhythm, Kallyope, and Arena with compensation to his employer, Mayo Clinic. Other authors declare no conflict of interest.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Calderon, G., Gonzalez-Izundegui, D., Shan, K.L. et al. Effectiveness of anti-obesity medications approved for long-term use in a multidisciplinary weight management program: a multi-center clinical experience. Int J Obes 46, 555–563 (2022). https://doi.org/10.1038/s41366-021-01019-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41366-021-01019-6
This article is cited by
-
Weight loss and cardiovascular disease risk outcomes of semaglutide: a one-year multicentered study
International Journal of Obesity (2024)
-
Narrative Review of Topiramate: Clinical Uses and Pharmacological Considerations
Advances in Therapy (2023)