Abstract
Background
Obesity often originates in early life, and is linked to excess sugar intake. Nonnutritive sweeteners (NNS) are widely consumed as “healthier” alternatives to sugar, yet recent evidence suggests NNS may adversely influence weight gain and metabolic health. The impact of NNS during critical periods of early development has rarely been studied. We investigated the effect of prenatal NNS exposure on postnatal adiposity and adipocyte development.
Methods
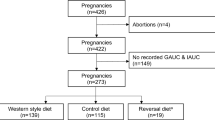
In the CHILD birth cohort (N = 2298), we assessed maternal NNS beverage intake during pregnancy and child body composition at 3 years, controlling for maternal BMI and other potential confounders. To investigate causal mechanisms, we fed NNS to pregnant C57BL6J mice at doses relevant to human consumption (42 mg/kg/day aspartame or 6.3 mg/kg/day sucralose), and assessed offspring until 12 weeks of age for: body weight, adiposity, adipose tissue morphology and gene expression, glucose and insulin tolerance. We also studied the effect of sucralose on lipid accumulation and gene expression in cultured 3T3-L1 pre-adipocyte cells.
Results
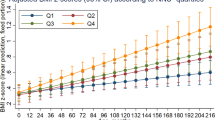
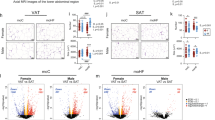
In the CHILD cohort, children born to mothers who regularly consumed NNS beverages had elevated body mass index (mean z-score difference +0.23, 95% CI 0.05–0.42 for daily vs. no consumption, adjusted for maternal BMI). In mice, maternal NNS caused elevated body weight, adiposity, and insulin resistance in offspring, especially in males (e.g., 47% and 15% increase in body fat for aspartame and sucralose vs. controls, p < 0.001). In cultured adipocytes, sucralose exposure at early stages of differentiation caused increased lipid accumulation and expression of adipocyte differentiation genes (e.g., C/EBP-α, FABP4, and FASN). These genes were also upregulated in adipose tissue of male mouse offspring born to sucralose-fed dams.
Conclusion
By triangulating evidence from humans, mice, and cultured adipocytes, this study provides new evidence that maternal NNS consumption during pregnancy may program obesity risk in offspring through effects on adiposity and adipocyte differentiation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Abarca-Gomez L, Abdeen ZA, Hamid ZA, Abu-Rmeileh NM, Acosta-Cazares B, Acuin C, et al. Worldwide trends in body-mass index, underweight, overweight, and obesity from 1975 to 2016: a pooled analysis of 2416 population-based measurement studies in 128.9 million children, adolescents, and adults. Lancet. 2017;390:2627–42.
Agarwal P, Morriseau TS, Kereliuk SM, Doucette CA, Wicklow BA, Dolinsky VW. Maternal obesity, diabetes during pregnancy and epigenetic mechanisms that influence the developmental origins of cardiometabolic disease in the offspring. Crit Rev Clin Lab Sci. 2018;55:71–101.
Borengasser SJ, Zhong Y, Kang P, Lindsey F, Ronis MJ, Badger TM, et al. Maternal obesity enhances white adipose tissue differentiation and alters genome-scale DNA methylation in male rat offspring. Endocrinology. 2013;154:4113–25.
Guan H, Arany E, van Beek JP, Chamson-Reig A, Thyssen S, Hill DJ, et al. Adipose tissue gene expression profiling reveals distinct molecular pathways that define visceral adiposity in offspring of maternal protein-restricted rats. Am J Physiol Endocrinol Metab. 2005;288:E663–73.
Liang X, Yang Q, Fu X, Rogers CJ, Wang B, Pan H, et al. Maternal obesity epigenetically alters visceral fat progenitor cell properties in male offspring mice. J Physiol. 2016;594:4453–66.
Wen J, Hong Q, Wang X, Zhu L, Wu T, Xu P, et al. The effect of maternal vitamin D deficiency during pregnancy on body fat and adipogenesis in rat offspring. Sci Rep. 2018;8:365.
Yang QY, Liang JF, Rogers CJ, Zhao JX, Zhu MJ, Du M. Maternal obesity induces epigenetic modifications to facilitate Zfp423 expression and enhance adipogenic differentiation in fetal mice. Diabetes. 2013;62:3727–35.
Hu FB. Resolved: there is sufficient scientific evidence that decreasing sugar-sweetened beverage consumption will reduce the prevalence of obesity and obesity-related diseases. Obes Rev. 2013;14:606–19.
Malik VS, Schulze MB, Hu FB. Intake of sugar-sweetened beverages and weight gain: a systematic review. Am J Clin Nutr. 2006;84:274–88.
Seferidi P, Millett C, Laverty AA. Sweetened beverage intake in association to energy and sugar consumption and cardiometabolic markers in children. Pediatr Obes. 2018;13:195–203.
Vos MB, Kaar JL, Welsh JA, Van Horn LV, Feig DI, Anderson CAM, et al. Added sugars and cardiovascular disease risk in children: a scientific statement from the American Heart Association. Circulation. 2017;135:e1017–34.
Fitch C, Keim KS. Position of the Academy of Nutrition and Dietetics: use of nutritive and nonnutritive sweeteners. J Acad Nutr Diet. 2012;112:739–58.
Gardner C, Wylie-Rosett J, Gidding SS, Steffen LM, Johnson RK, Reader D, et al. Nonnutritive sweeteners: current use and health perspectives: a scientific statement from the American Heart Association and the American Diabetes Association. Diabetes Care. 2012;35:1798–808.
Azad MB, Sharma AK, de Souza RJ, Dolinsky VW, Becker AB, Mandhane PJ, et al. Association between artificially sweetened beverage consumption during pregnancy and infant body mass index. JAMA. 2016;170:662–70.
Sylvetsky AC, Figueroa J, Rother KI, Goran MI, Welsh JA. Trends in low-calorie sweetener consumption among pregnant women in the United States. Curr Dev Nutr. 2019;3:nzz004.
Zhu Y, Olsen SF, Mendola P, Halldorsson TI, Rawal S, Hinkle SN, et al. Maternal consumption of artificially sweetened beverages during pregnancy, and offspring growth through 7 years of age: a prospective cohort study. Int J Epidemiol. 2017;46:1499–508.
Azad MB, Abou-Setta AM, Chauhan BF, Rabbani R, Lys J, Copstein L, et al. Nonnutritive sweeteners and cardiometabolic health: a systematic review and meta-analysis of randomized controlled trials and prospective cohort studies. Can Med Assoc J. 2017;189:E929–39.
Pearlman M, Obert J, Casey L. The association between artificial sweeteners and obesity. Curr Gastroenterol Rep. 2017;19:64.
Toews I, Lohner S, Kullenberg de Gaudry D, Sommer H, Meerpohl JJ. Association between intake of non-sugar sweeteners and health outcomes: systematic review and meta-analyses of randomised and non-randomised controlled trials and observational studies. BMJ. 2019;364:k4718.
Archibald AJ, Dolinsky VW, Azad MB. Early-life exposure to non-nutritive sweeteners and the developmental origins of childhood obesity: global evidence from human and rodent studies. Nutrients. 2018;10:194.
Gillman MW, Rifas-Shiman SL, Fernandez-Barres S, Kleinman K, Taveras EM, Oken E. Beverage intake during pregnancy and childhood adiposity. Pediatrics. 2017;140. https://pediatrics.aappublications.org/content/140/2/e20170031.long.
Araujo JR, Martel F, Keating E. Exposure to non-nutritive sweeteners during pregnancy and lactation: impact in programming of metabolic diseases in the progeny later in life. Reprod Toxicol. 2014;49:196–201.
Collison KS, Makhoul NJ, Zaidi MZ, Saleh SM, Andres B, Inglis A, et al. Gender dimorphism in aspartame-induced impairment of spatial cognition and insulin sensitivity. PLoS ONE. 2012;7:e31570.
Olivier-Van Stichelen S, Rother KI, Hanover JA. Maternal exposure to non-nutritive sweeteners impacts progeny’s metabolism and microbiome. Front Microbiol. 2019;10:1360.
von Poser Toigo E, Huffell AP, Mota CS, Bertolini D, Pettenuzzo LF, Dalmaz C. Metabolic and feeding behavior alterations provoked by prenatal exposure to aspartame. Appetite. 2015;87:168–74.
Soffritti M, Belpoggi F, Manservigi M, Tibaldi E, Lauriola M, Falcioni L, et al. Aspartame administered in feed, beginning prenatally through life span, induces cancers of the liver and lung in male Swiss mice. Am J Ind Med. 2010;53:1197–206.
Simon BR, Parlee SD, Learman BS, Mori H, Scheller EL, Cawthorn WP, et al. Artificial sweeteners stimulate adipogenesis and suppress lipolysis independently of sweet taste receptors. J Biol Chem. 2013;288:32475–89.
Masubuchi Y, Nakagawa Y, Ma J, Sasaki T, Kitamura T, Yamamoto Y, et al. A novel regulatory function of sweet taste-sensing receptor in adipogenic differentiation of 3T3-L1 cells. PLoS ONE. 2013;8:e54500.
Pandurangan M, Park J, Kim E. Aspartame downregulates 3T3-L1 differentiation. In Vitro Cell Dev Biol Anim. 2014;50:851–7.
Subbarao P, Anand SS, Becker AB, Befus AD, Brauer M, Brook JR, et al. The Canadian Healthy Infant Longitudinal Development (CHILD) Study: examining developmental origins of allergy and asthma. Thorax. 2015;70:998–1000.
Patterson RE, Kristal AR, Tinker LF, Carter RA, Bolton MP, Agurs-Collins T. Measurement characteristics of the Women’s Health Initiative food frequency questionnaire. Ann Epidemiol. 1999;9:178–87.
Qiu C, Coughlin KB, Frederick IO, Sorensen TK, Williams MA. Dietary fiber intake in early pregnancy and risk of subsequent preeclampsia. Am J Hypertens. 2008;21:903–9.
Maslova E, Strom M, Olsen SF, Halldorsson TI. Consumption of artificially-sweetened soft drinks in pregnancy and risk of child asthma and allergic rhinitis. PLoS ONE. 2013;8:e57261.
Guenther PM, Casavale KO, Reedy J, Kirkpatrick SI, Hiza HA, Kuczynski KJ, et al. Update of the Healthy Eating Index: HEI-2010. J Acad Nutr Diet. 2013;113:569–80.
Pereira TJ, Fonseca MA, Campbell KE, Moyce BL, Cole LK, Hatch GM, et al. Maternal obesity characterized by gestational diabetes increases the susceptibility of rat offspring to hepatic steatosis via a disrupted liver metabolome. J Physiol. 2015;593:3181–97.
Dolinsky VW, Gilham D, Hatch GM, Agellon LB, Lehner R, Vance DE. Regulation of triacylglycerol hydrolase expression by dietary fatty acids and peroxisomal proliferator-activated receptors. Biochim Biophys Acta. 2003;1635:20–8.
Vandesompele J, De Preter K, Pattyn F, Poppe B, Van Roy N, De Paepe A, et al. Accurate normalization of real-time quantitative RT-PCR data by geometric averaging of multiple internal control genes. Genome Biol. 2002;3:Research0034.
Guariguata L, Linnenkamp U, Beagley J, Whiting DR, Cho NH. Global estimates of the prevalence of hyperglycaemia in pregnancy. Diabetes Res Clin Pract. 2014;103:176–85.
Moseti D, Regassa A, Kim WK. Molecular regulation of adipogenesis and potential anti-adipogenic bioactive molecules. Int J Mol Sci. 2016;17:E124.
Roberts A, Renwick AG, Sims J, Snodin DJ. Sucralose metabolism and pharmacokinetics in man. Food Chem Toxicol. 2000;38 Suppl 2:S31–41.
Nettleton JE, Reimer RA, Shearer J. Reshaping the gut microbiota: Impact of low calorie sweeteners and the link to insulin resistance? Physiol Behav. 2016;164:488–93.
Ruiz-Ojeda FJ, Plaza-Diaz J, Saez-Lara MJ, Gil A. Effects of sweeteners on the gut microbiota: a review of experimental studies and clinical trials. Adv Nutr. 2019;10 Suppl 1:S31–48.
Asnicar F, Manara S, Zolfo M, Truong DT, Scholz M, Armanini F, et al. Studying vertical microbiome transmission from mothers to infants by strain-level metagenomic profiling. mSystems. 2017;2:e00164–16.
Le Doare K, Holder B, Bassett A, Pannaraj PS. Mother’s milk: a purposeful contribution to the development of the infant microbiota and immunity. Front Immunol. 2018;9:361.
Liou AP, Paziuk M, Luevano JM Jr, Machineni S, Turnbaugh PJ, Kaplan LM. Conserved shifts in the gut microbiota due to gastric bypass reduce host weight and adiposity. Sci Transl Med. 2013;5:178ra41.
Meijnikman AS, Gerdes VE, Nieuwdorp M, Herrema H. Evaluating causality of gut microbiota in obesity and diabetes in humans. Endocr Rev. 2018;39:133–53.
Turnbaugh PJ, Ley RE, Mahowald MA, Magrini V, Mardis ER, Gordon JI. An obesity-associated gut microbiome with increased capacity for energy harvest. Nature. 2006;444:1027–31.
Acknowledgements
We are grateful to all the families who took part in the CHILD study, and the whole CHILD team, which includes interviewers, nurses, computer and laboratory technicians, clerical workers, research scientists, volunteers, managers, and receptionists. We also acknowledge the excellent technical work of Mario Fonseca and Bo Xiang (University of Manitoba), critical review by Shirin Moossavi (University of Manitoba), and editorial assistance from John Schellenberg (University of Manitoba). A preprint of this work was posted on bioRxiv (https://doi.org/10.1101/713974).
Funding
The Canadian Institutes of Health Research (CIHR) and the Allergy, Genes and Environment Network of Centers of Excellence (AllerGen NCE) provided core support for the CHILD Study. This research was supported, in part, by the Canada Research Chairs program. MBA holds the Tier 2 Canada Research Chair in the Developmental Origins of Chronic Disease, and is a Canadian Institute for Advanced Research Fellow in the Humans and the Microbiome Program. VWD holds the Allen Rouse-Manitoba Medical Services Foundation Basic Scientist Award. MMT is the recipient of a Research Manitoba/CHRIM studentship. This research was supported by a Children’s Hospital Research Institute of Manitoba Grant, a CIHR Operating Grant #151540, and a CIHR Environments, Genes and Chronic Disease Team Grant #144626. These entities had no role in the design and conduct of the study; collection, management, analysis, and interpretation of the data; and preparation, review, or approval of the paper.
Author information
Authors and Affiliations
Contributions
MBA and VWD conceived of the study design, obtained funding for this research, and drafted the paper. MRS, PS, TJM, SET, PJM, and ABB obtained funding for and oversaw recruitment of the CHILD cohort and data collection. AA performed the statistical analysis of clinical data from the CHILD cohort under the supervision of MBA. RJS contributed nutritional expertise. MMT, AH, and KGC performed mouse and cell culture experiments under the supervision of VWD. All authors critically reviewed and approved the paper. MBA had full access to the human data and VWD had full access to the mouse and adipocyte data, and take final responsibility for the decision to submit for publication.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Azad, M.B., Archibald, A., Tomczyk, M.M. et al. Nonnutritive sweetener consumption during pregnancy, adiposity, and adipocyte differentiation in offspring: evidence from humans, mice, and cells. Int J Obes 44, 2137–2148 (2020). https://doi.org/10.1038/s41366-020-0575-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41366-020-0575-x