Abstract
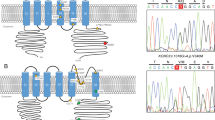
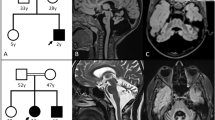
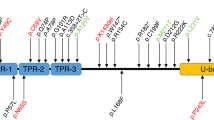
Spinocerebellar ataxia type 14 (SCA14) is an autosomal dominant SCA caused by variants of the PRKCG encoding protein kinase C gamma (PKCγ). Although the toxic gain-of-function mechanism is the main cause of SCA14, its molecular pathophysiology remains unclear. To elucidate the molecular pathogenesis of SCA14, we analyzed two families with the variants in PRKCG. Clinical symptoms and neurological findings of two Japanese families were evaluated by neurologists. Exome sequencing was performed using the BGI platform. GFP-tagged PRKCGs harboring the identified variants were transfected into the HeLa cells, and aggregation of PKCγ was analyzed using confocal laser microscopy. Solubility of PKCγ was evaluated by assessing the proportion of insoluble fraction present in1% Triton-X. Patients in family 1 presented with only cerebellar atrophy without ataxia; however, patients in family 2 exhibited cerebellar ataxia, dystonia, and more severe cerebellar atrophy than those in family 1. Exome sequencing identified two novel missense variants of PRKCG:c.171 G > C,p.W57C (family 1), and c.400 T > C,p.C134R (family 2). Both the mutant PKCγ aggregated in the cytoplasm. Although the solubility of PKCγ of the C134R variant was lower than that of the wild-type, PKCγ of W57C retained its solubility. In conclusion, we identified two novel variants of PRKCG. The difference in severity between the two families may be due to the difference in solubility changes observed between the two variants. Decreased solubility of the PKCγ may play an important role in the pathogenesis of SCA14.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Change history
07 July 2022
A Correction to this paper has been published: https://doi.org/10.1038/s10038-022-01064-7
References
Krygier M, Mazurkiewicz-Bełdzińska M. Milestones in genetics of cerebellar ataxias. Neurogenetics. 2021;22:225–34.
Müller U. Spinocerebellar ataxias (SCAs) caused by common mutations. Neurogenetics. 2021;22:235–50.
Tada Y, Kume K, Matsuda Y, Kurashige T, Kanaya Y, Ohsawa R, et al. Genetic screening for potassium channel mutations in Japanese autosomal dominant spinocerebellar ataxia. J Hum Genet. 2020;65:363–9.
Chen DH, Brkanac Z, Verlinde CL, Tan XJ, Bylenok L, Nochlin D, et al. Missense mutations in the regulatory domain of PKC gamma: a new mechanism for dominant nonepisodic cerebellar ataxia. Am J Hum Genet. 2003;72:839–49.
Chelban V, Wiethoff S, Fabian-Jessing BK, Haridy NA, Khan A, Efthymiou S, et al. Genotype-phenotype correlations, dystonia and disease progression in spinocerebellar ataxia type 14. Mov Dis. 2018;33:1119–29.
Seki T, Adachi N, Ono Y, Mochizuki H, Hiramoto K, Amano T, et al. Mutant protein kinase Cgamma found in spinocerebellar ataxia type 14 is susceptible to aggregation and causes cell death. J Biol Chem. 2005;280:29096–106.
Seki T, Takahashi H, Adachi N, Abe N, Shimahara T, Saito N, et al. Aggregate formation of mutant protein kinase C gamma found in spinocerebellar ataxia type 14 impairs ubiquitin-proteasome system and induces endoplasmic reticulum stress. Eur J Neurosci. 2007;26:3126–40.
Verbeek DS, Knight MA, Harmison GG, Fischbeck KH, Howell BW. Protein kinase C gamma mutations in spinocerebellar ataxia 14 increase kinase activity and alter membrane targeting. Brain. 2005;128:436–42.
Adachi N, Kobayashi T, Takahashi H, Kawasaki T, Shirai Y, Ueyama T, et al. Enzymological analysis of mutant protein kinase Cgamma causing spinocerebellar ataxia type 14 and dysfunction in Ca2+ homeostasis. J Biol Chem. 2008;283:19854–63.
Wong MMK, Hoekstra SD, Vowles J, Watson LM, Fuller G, Németh AH, et al. Neurodegeneration in SCA14 is associated with increased PKCγ kinase activity, mislocalization and aggregation. Acta Neuropathol Commun. 2018;6:99.
Matsuda Y, Morino H, Miyamoto R, Kurashige T, Kume K, Mizuno N, et al. Biallelic mutation of HSD17B4 induces middle age-onset spinocerebellar ataxia. Neurol Genet. 2020;6:e396.
Kume K, Morino H, Miyamoto R, Matsuda Y, Ohsawa R, Kanaya Y, et al. Middle-age-onset cerebellar ataxia caused by a homozygous TWNK variant: a case report. BMC Med Genet. 2020;21:68.
Asai H, Hirano M, Shimada K, Kiriyama T, Furiya Y, Ikeda M, et al. Protein kinase C gamma, a protein causative for dominant ataxia, negatively regulates nuclear import of recessive-ataxia-related aprataxin. Hum Mol Genet. 2009;18:3533–43.
Acknowledgements
We thank Ms. Eiko Nakajima and Dr. Mayumi Miyamoto for their excellent technical assistance. This work was supported by JSPS KAKENHI under the grant numbers JP21K15698, the Uehara Memorial Foundation, and the Research Committee for Ataxic Disease. We would like to thank Editage (www.editage.com) for English language editing.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Tada, Y., Kume, K., Noguchi, S. et al. Comparison of two families with and without ataxia harboring novel variants in PRKCG. J Hum Genet 67, 595–599 (2022). https://doi.org/10.1038/s10038-022-01057-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s10038-022-01057-6