Abstract
Background
The trafficking protein particle (TRAPP) complex subunit 9 (C9) protein is a member of TRAPP-II complexes and regulates vesicle trafficking. Biallelic mutations in the TRAPPC9 gene are responsible for intellectual disability with expanded developmental delay, epilepsy, microcephaly, and brain atrophy. TRAPPC9-related disease list is still expanding, however, the functional effects of only a limited fraction of these have been studied.
Methods
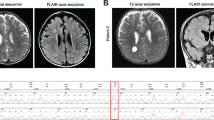
In a patient with a pathological variant in TRAPPC9, clinical examination and cranial imaging findings were evaluated. Whole-exome sequencing, followed by Sanger sequencing was performed to detect and verify the variant. To confirm the functional effect of the mutation; variant mRNA and protein expression levels were evaluated by qRT-PCR and Western blotting. Immunostaining for TRAPPC9 and lipid droplet accumulation were examined.
Results
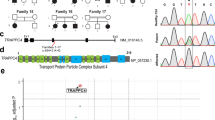
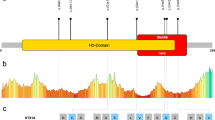
We have identified a novel homozygous c.696C>G (p.Phe232Leu) pathogenic variant in TRAPPC9 (NM_031466.6) gene as a cause of severe developmental delay. Functional characterization of the TRAPPC9 variant resulted in decreased mRNA and protein expression. The intracellular findings showed that TRAPPC9 protein build-up around the nucleus in mutant type while there was no specific accumulation in the control cell line. This disrupted protein pattern affected the amount of neutral lipid-carrying vesicles and their homogenous distribution at a decreasing level.
Conclusion
Biallelic variants in the TRAPPC9 gene have been reported as the underlying cause of intellectual disability. This study provides functional evidence of the novel variant in TRAPPC9 We demonstrated that the loss of function variant exclusively targeting TRAPPC9 may explicate the neurological findings through vesicle trafficking.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Musante L, Ropers HH. Genetics of recessive cognitive disorders. Trends Genet. 2014;30:32–9.
Vissers LE, Gilissen C, Veltman JA. Genetic studies in intellectual disability and related disorders. Nat Rev Genet. 2016;17:9–18.
Reuter MS, Tawamie H, Buchert R, Hosny Gebril O, Froukh T, Thiel C, et al. Diagnostic yield and novel candidate genes by exome sequencing in 152 consanguineous families with neurodevelopmental disorders. JAMA Psychiatry. 2017;74:293–9.
Mir A, Kaufman L, Noor A, Motazacker MM, Jamil T, Azam M, et al. Identification of mutations in TRAPPC9, which encodes the NIK- and IKK-beta-binding protein, in nonsyndromic autosomal-recessive mental retardation. Am J Hum Genet. 2009;85:909–15.
Mochida GH, Mahajnah M, Hill AD, Basel-Vanagaite L, Gleason D, Hill RS, et al. A truncating mutation of TRAPPC9 is associated with autosomal-recessive intellectual disability and postnatal microcephaly. Am J Hum Genet. 2009;85:897–902.
Philippe O, Rio M, Carioux A, Plaza JM, Guigue P, Molinari F, et al. Combination of linkage mapping and microarray-expression analysis identifies NF-kappaB signaling defect as a cause of autosomal-recessive mental retardation. Am J Hum Genet. 2009;85:903–8.
Mortreux J, Busa T, Germain DP, Nadeau G, Puechberty J, Coubes C, et al. The role of CNVs in the etiology of rare autosomal recessive disorders: the example of TRAPPC9-associated intellectual disability. Eur J Hum Genet. 2018;26:143–8.
Hnoonual A, Graidist P, Kritsaneepaiboon S, Limprasert P. Novel Compound heterozygous mutations in the TRAPPC9 gene in two siblings with autism and intellectual disability. Front Genet. 2019;10:61.
Wilton KM, Gunderson LB, Hasadsri L, Wood CP, Schimmenti LA. Profound intellectual disability caused by homozygous TRAPPC9 pathogenic variant in a man from Malta. Mol Genet Genom Med. 2020;8:e1211.
Yousefipour F, Mozhdehipanah H, Mahjoubi F. Identification of two novel homozygous nonsense mutations in TRAPPC9 in two unrelated consanguineous families with intellectual disability from Iran. Mol Genet Genomic Med. 2021:e1610.
Kramer J, Beer M, Bode H, Winter B. Two novel compound heterozygous mutations in the TRAPPC9 gene reveal a connection of non-syndromic intellectual disability and autism spectrum disorder. Front Genet. 2020;11:972.
Koifman A, Feigenbaum A, Bi W, Shaffer LG, Rosenfeld J, Blaser S, et al. A homozygous deletion of 8q24.3 including the NIBP gene associated with severe developmental delay, dysgenesis of the corpus callosum, and dysmorphic facial features. Am J Med Genet A. 2010;152A:1268–72.
Abou Jamra R, Wohlfart S, Zweier M, Uebe S, Priebe L, Ekici A, et al. Homozygosity mapping in 64 Syrian consanguineous families with non-specific intellectual disability reveals 11 novel loci and high heterogeneity. Eur J Hum Genet. 2011;19:1161–6.
Kakar N, Goebel I, Daud S, Nurnberg G, Agha N, Ahmad A, et al. A homozygous splice site mutation in TRAPPC9 causes intellectual disability and microcephaly. Eur J Med Genet. 2012;55:727–31.
Marangi G, Leuzzi V, Manti F, Lattante S, Orteschi D, Pecile V, et al. TRAPPC9-related autosomal recessive intellectual disability: report of a new mutation and clinical phenotype. Eur J Hum Genet. 2013;21:229–32.
Giorgio E, Ciolfi A, Biamino E, Caputo V, Di Gregorio E, Belligni EF, et al. Whole exome sequencing is necessary to clarify ID/DD cases with de novo copy number variants of uncertain significance: two proof-of-concept examples. Am J Med Genet A. 2016;170:1772–9.
Abbasi AA, Blaesius K, Hu H, Latif Z, Picker-Minh S, Khan MN, et al. Identification of a novel homozygous TRAPPC9 gene mutation causing non-syndromic intellectual disability, speech disorder, and secondary microcephaly. Am J Med Genet B Neuropsychiatr Genet. 2017;174:839–45.
Boonsawat P, Joset P, Steindl K, Oneda B, Gogoll L, Azzarello-Burri S, et al. Elucidation of the phenotypic spectrum and genetic landscape in primary and secondary microcephaly. Genet Med. 2019;21:2043–58.
Bai Z, Kong X. Diagnosis of a case with mental retardation due to novel compound heterozygous variants of TRAPPC9 gene. Zhonghua Yi Xue Yi Chuan Xue Za Zhi. 2019;36:1115–9.
Alvarez-Mora MI, Corominas J, Gilissen C, Sanchez A, Madrigal I, Rodriguez-Revenga L. Novel compound heterozygous mutation in TRAPPC9 gene: The relevance of whole genome sequencing. Genes. 2021;12:557.
Sacher M, Shahrzad N, Kamel H, Milev MP. TRAPPopathies: an emerging set of disorders linked to variations in the genes encoding transport protein particle (TRAPP)-associated proteins. Traffic. 2019;20:5–26.
Vangipuram M, Ting D, Kim S, Diaz R, Schule B. Skin punch biopsy explant culture for derivation of primary human fibroblasts. J Vis Exp. 2013;77:e3779.
Livak KJ, Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) method. Methods. 2001;25:402–8.
Schneider CA, Rasband WS, Eliceiri KW. NIH Image to ImageJ: 25 years of image analysis. Nat Methods. 2012;9:671–5.
Greenspan P, Mayer EP, Fowler SD. Nile red: a selective fluorescent stain for intracellular lipid droplets. J Cell Biol. 1985;100:965–73.
Kim JJ, Lipatova Z, Segev N. TRAPP complexes in secretion and autophagy. Front Cell Dev Biol. 2016;4:20.
Hu WH, Pendergast JS, Mo XM, Brambilla R, Bracchi-Ricard V, Li F, et al. NIBP, a novel NIK and IKK(beta)-binding protein that enhances NF-(kappa)B activation. J Biol Chem. 2005;280:29233–41.
Cox R, Chen SH, Yoo E, Segev N. Conservation of the TRAPPII-specific subunits of a Ypt/Rab exchanger complex. BMC Evol Biol. 2007;7:12.
Harris NJ, Jenkins ML, Dalwadi U, Fleming KD, Nam SE, Parson MAH, et al. Biochemical insight into novel Rab-GEF activity of the mammalian TRAPPIII complex. J Mol Biol. 2021;433:167145.
Wang J, Fourriere L, Gleeson PA. Local secretory trafficking pathways in neurons and the role of dendritic golgi outposts in different cell models. Front Mol Neurosci. 2020;13:597391.
Bodnar B, DeGruttola A, Zhu Y, Lin Y, Zhang Y, Mo X, et al. Emerging role of NIK/IKK2-binding protein (NIBP)/trafficking protein particle complex 9 (TRAPPC9) in nervous system diseases. Transl Res. 2020;224:55–70.
Zong M, Satoh A, Yu MK, Siu KY, Ng WY, Chan HC, et al. TRAPPC9 mediates the interaction between p150 and COPII vesicles at the target membrane. PLoS One. 2012;7:e29995.
Li C, Luo X, Zhao S, Siu GK, Liang Y, Chan HC, et al. COPI-TRAPPII activates Rab18 and regulates its lipid droplet association. EMBO J. 2017;36:441–57.
Thomas LL, Fromme JC. GTPase cross talk regulates TRAPPII activation of Rab11 homologues during vesicle biogenesis. J Cell Biol. 2016;215:499–513.
Acknowledgements
We are grateful to the patient and his family for their participation in the study.
Funding
This study was supported by the Research Fund of the Bezmialem Vakif University, project number of 42019/20.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Aslanger, A.D., Goncu, B., Duzenli, O.F. et al. Biallelic loss of TRAPPC9 function links vesicle trafficking pathway to autosomal recessive intellectual disability. J Hum Genet 67, 279–284 (2022). https://doi.org/10.1038/s10038-021-01007-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s10038-021-01007-8
This article is cited by
-
A novel homozygous mutation in TRAPPC9 gene causing autosomal recessive non-syndromic intellectual disability
BMC Medical Genomics (2022)
-
Postmortem Human Dura Mater Cells Exhibit Phenotypic, Transcriptomic and Genetic Abnormalities that Impact their Use for Disease Modeling
Stem Cell Reviews and Reports (2022)