Abstract
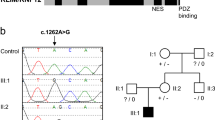
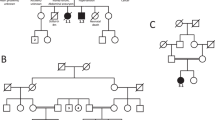
Peptidyl arginine deiminase, type VI (PADI6) is a member of the subcortical maternal complex (SCMC), which plays vital roles in mammalian embryogenesis. Most mutations in SCMC members have been reported to cause human embryonic arrest, and a total of 15 mutations in PADI6 have been shown to be responsible for early embryonic arrest according to previous studies. However, the genetic factors behind this phenotype remain to be understood in further detail. Here, we identified 13 novel mutations and 4 previously reported mutations of PADI6 in 14 patients who were diagnosed with abnormal embryonic development caused by early arrest, embryonic fragmentation, and recurrent implantation failure. Most of the mutations were predicted by in silico analysis to be deleterious or damaging to the function of PADI6. In addition, the total and East Asian population frequencies of the mutations were low or absent in the gnomAD database. Our study expands the mutational spectrum in PADI6 and will provide precise targets for genetic counseling in the future.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Data availability
The data underlying this article will be shared on reasonable request to the corresponding author.
Material availability
The material underlying this article will be shared on reasonable request to the corresponding author.
References
Lee MT, Bonneau AR, Giraldez AJ. Zygotic genome activation during the maternal-to-zygotic transition. Annu Rev Cell Dev Biol. 2014;30:581–613.
Zegers-Hochschild F, Adamson GD, de Mouzon J, Ishihara O, Mansour R, Nygren K, et al. International Committee for Monitoring Assisted Reproductive Technology (ICMART) and the World Health Organization (WHO) revised glossary of ART terminology, 2009. Fertil Steril. 2009;92:1520–4.
Gardner DK, Lane M. Culture and selection of viable blastocysts: a feasible proposition for human IVF? Hum Reprod Update. 1997;3:367–82.
Li L, Baibakov B, Dean J. A subcortical maternal complex essential for preimplantation mouse embryogenesis. Dev Cell. 2008;15:416–25.
Lu X, Gao Z, Qin D, Li L. A maternal functional module in the mammalian oocyte-to-embryo transition. Trends Mol Med. 2017;23:1014–23.
Bebbere D, Albertini DF, Coticchio G, Borini A, Ledda S. The subcortical maternal complex: emerging roles and novel perspectives. Mol Hum Reprod. 2021;27:gaab043.
Alazami AM, Awad SM, Coskun S, Al-Hassan S, Hijazi H, Abdulwahab FM, et al. TLE6 mutation causes the earliest known human embryonic lethality. Genome Biol. 2015;16:240.
Wang X, Song D, Mykytenko D, Kuang Y, Lv Q, Li B, et al. Novel mutations in genes encoding subcortical maternal complex proteins may cause human embryonic developmental arrest. Reprod Biomed Online. 2018;36:698–704.
Lin J, Xu H, Chen B, Wang W, Wang L, Sun X, et al. Expanding the genetic and phenotypic spectrum of female infertility caused by TLE6 mutations. J Assist Reprod Genet. 2020;37:437–42.
Mu J, Wang W, Chen B, Wu L, Li B, Mao X, et al. Mutations in NLRP2 and NLRP5 cause female infertility characterised by early embryonic arrest. J Med Genet. 2019;56:471–80.
Xu Y, Shi Y, Fu J, Yu M, Feng R, Sang Q, et al. Mutations in PADI6 cause female infertility characterized by early embryonic arrest. Am J Hum Genet. 2016;99:744–52.
Kan R, Yurttas P, Kim B, Jin M, Wo L, Lee B, et al. Regulation of mouse oocyte microtubule and organelle dynamics by PADI6 and the cytoplasmic lattices. Dev Biol. 2011;350:311–22.
Yurttas P, Vitale AM, Fitzhenry RJ, Cohen-Gould L, Wu W, Gossen JA, et al. Role for PADI6 and the cytoplasmic lattices in ribosomal storage in oocytes and translational control in the early mouse embryo. Development. 2008;135:2627–36.
Qian J, Nguyen NMP, Rezaei M, Huang B, Tao Y, Zhang X, et al. Biallelic PADI6 variants linking infertility, miscarriages, and hydatidiform moles. Eur J Hum Genet. 2018;26:1007–13.
Maddirevula S, Coskun S, Awartani K, Alsaif H, Abdulwahab FM, Alkuraya FS. The human knockout phenotype of PADI6 is female sterility caused by cleavage failure of their fertilized eggs. Clin Genet. 2017;91:344–45.
Zheng W, Chen L, Dai J, Dai C, Guo J, Lu C, et al. New biallelic mutations in PADI6 cause recurrent preimplantation embryonic arrest characterized by direct cleavage. J Assist Reprod Genet. 2020;37:205–12.
Begemann M, Rezwan FI, Beygo J, Docherty LE, Kolarova J, Schroeder C, et al. Maternal variants in NLRP and other maternal effect proteins are associated with multilocus imprinting disturbance in offspring. J Med Genet. 2018;55:497–504.
Cubellis MV, Pignata L, Verma A, Sparago A, Del Prete R, Monticelli M, et al. Loss-of-function maternal-effect mutations of PADI6 are associated with familial and sporadic Beckwith-Wiedemann syndrome with multi-locus imprinting disturbance. Clin Epigenetics. 2020;12:139.
Eggermann T, Kadgien G, Begemann M, Elbracht M. Biallelic PADI6 variants cause multilocus imprinting disturbances and miscarriages in the same family. Eur J Hum Genet. 2021;29:575–80.
Liu J, Tan Z, He J, Jin T, Han Y, Hu L, et al. Two novel mutations in PADI6 and TLE6 genes cause female infertility due to arrest in embryonic development. J Assist Reprod Genet. 2021;38:1551–59.
Mogessie B, Schuh M. Actin protects mammalian eggs against chromosome segregation errors. Science. 2017;357:eaal1647.
Lu YQ, He XC, Zheng P. Decrease in expression of maternal effect gene Mater is associated with maternal ageing in mice. Mol Hum Reprod. 2016;22:252–60.
Acknowledgements
The authors thank the patients and their families for participating in this study, as well as the doctors for collecting samples and providing clinical support.
Funding
This work was supported by the National Natural Science Foundation of China (81725006, 81822019, 81771581, 81771649, 81971450, 81971382, 82001538, 82071642 and 81901561), the project supported by the Shanghai Municipal Science and Technology Major Project (2017SHZDZX01), the Project of the Shanghai Municipal Science and Technology Commission (19JC1411001), the Natural Science Foundation of Shanghai (19ZR1444500, 21ZR1404800), the Shuguang Program of the Shanghai Education Development Foundation and the Shanghai Municipal Education Commission (18SG03), the Foundation of the Shanghai Health and Family Planning Commission (20154Y0162), the Capacity Building Planning Program for Shanghai Women and Children’s Health Service, and the collaborative innovation center project construction for Shanghai Women and Children’s Health.
Author information
Authors and Affiliations
Contributions
QingS, LW, and JD conceived and designed the research study. JF, ZY, LL, QiuwenS, RS, FD, and YQ collected the clinical samples. BC organized the medical records and analyzed the whole-exome data. JD, YZ, and RL performed the genome sequencing. QingS and JD drafted and revised the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval and consent to participate
Our study was approved by the Ethics Committee of the Medical College of Fudan University and the Reproductive Study Ethics Committees of the hospital. All blood samples were donated for the investigation after informed consent.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Dong, J., Fu, J., Yan, Z. et al. Novel biallelic mutations in PADI6 in patients with early embryonic arrest. J Hum Genet 67, 285–293 (2022). https://doi.org/10.1038/s10038-021-00998-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s10038-021-00998-8