Abstract
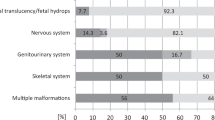
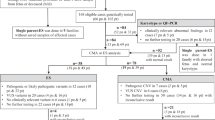
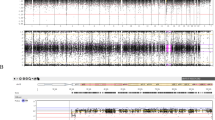
Müllerian anomaly (M.A.) is a group of congenital anatomic abnormalities caused by aberrations of the development process of the Müllerian duct. M.A. can either be isolated or be involved in Mendelian syndromes, such as Dandy-Walker syndrome, Holt–Oram syndrome and Bardet–Biedl syndrome, which are often associated with both uterus and kidney malformations. In this study, we applied a genotype-first approach to analyze the whole-exome sequencing data of 492 patients with M.A. Six potential pathogenic variants were found in five genes previously related to female urogenital deformities (PKD1, SON, SALL1, BMPR1B, ITGA8), which are partially overlapping with our patients’ phenotypes. We further identified eight incidental findings in seven genes related to Mendelian syndromes without known association with reproductive anomalies (TEK, COL11A1, ANKRD11, LEMD3, DLG5, SPTB, BMP2), which represent potential phenotype expansions of these genes.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Alvelos MI, Rodrigues M, Lobo L, Medeira A, Sousa AB, Simão C, et al. A novel mutation of the HNF1B gene associated with hypoplastic glomerulocystic kidney disease and neonatal renal failure: a case report and mutation update. Medicine (Baltimore). 2015;94:e469.
Botzenhart EM, Green A, Ilyina H, König R, Lowry RB, Lo IF, et al. SALL1 mutation analysis in Townes-Brocks syndrome: twelve novel mutations and expansion of the phenotype. Hum Mutat. 2005;26:282.
Kobayashi A, Shawlot W, Kania A, Behringer RR. Requirement of Lim1 for female reproductive tract development. Development 2004;131:539–49.
Orvis GD, Behringer RR. Cellular mechanisms of Müllerian duct formation in the mouse. Dev Biol. 2007;306:493–504.
Stessman HA, Bernier R, Eichler EE. A genotype-first approach to defining the subtypes of a complex disease. Cell 2014;156:872–7.
Sanna-Cherchi S, Khan K, Westland R, Krithivasan P, Fievet L, Rasouly HM, et al. Exome-wide association study identifies GREB1L mutations in congenital kidney malformations. Am J Hum Genet. 2017;101:789–802.
Schrauwen I, Kari E, Mattox J, Llaci L, Smeeton J, Naymik M, et al. De novo variants in GREB1L are associated with non-syndromic inner ear malformations and deafness. Hum Genet. 2018;137:459–70.
Jacquinet A, Boujemla B, Fasquelle C, Thiry J, Josse C, Lumaka A, et al. GREB1L variants in familial and sporadic hereditary urogenital adysplasia and Mayer-Rokitansky-Kuster-Hauser syndrome. Clin Genet. 2020;98:126–37.
Herlin MK, Le VQ, H Jland AT, Ernst A, Okkels H, Petersen AC, et al. Whole-exome sequencing identifies a GREB1L variant in a three-generation family with Müllerian and renal agenesis: a novel candidate gene in Mayer-Rokitansky-Küster-Hauser (MRKH) syndrome. A case report. Hum Reprod. 2019;34:1838–46.
Macchia PE, Lapi P, Krude H, Pirro MT, Missero C, Chiovato L, et al. PAX8 mutations associated with congenital hypothyroidism caused by thyroid dysgenesis. Nat Genet. 1998;19:83–86.
Chen N, Zhao S, Jolly A, Wang L, Pan H, Yuan J, et al. Perturbations of genes essential for Müllerian duct and Wölffian duct development in Mayer-Rokitansky-Küster-Hauser syndrome. Am J Hum Genet. 2021;108:337–45.
Zhao S, Zhang Y, Chen W, Li W, Wang S, Wang L, et al. Diagnostic yield and clinical impact of exome sequencing in early-onset scoliosis (EOS). J Med Genet. 2020;58:41–47.
Richards S, Aziz N, Bale S, Bick D, Das S, Gastier-Foster J, et al. Standards and guidelines for the interpretation of sequence variants: a joint consensus recommendation of the American College of Medical Genetics and Genomics and the Association for Molecular Pathology. Genet Med. 2015;17:405–24.
Little MH, Brennan J, Georgas K, Davies JA, Davidson DR, Baldock RA, et al. A high-resolution anatomical ontology of the developing murine genitourinary tract. Gene Expr Patterns. 2007;7:680–99.
Ali H, Hussain N, Naim M, Zayed M, Al-Mulla F, Kehinde EO, et al. A novel PKD1 variant demonstrates a disease-modifying role in trans with a truncating PKD1 mutation in patients with autosomal dominant polycystic kidney disease. BMC Nephrol. 2015;16:26.
Zhang L, Li LX, Zhou JX, Harris PC, Calvet JP, Li X. RNA helicase p68 inhibits the transcription and post-transcription of Pkd1 in ADPKD. Theranostics 2020;10:8281–97.
Song X, Di Giovanni V, He N, Wang K, Ingram A, Rosenblum ND, et al. Systems biology of autosomal dominant polycystic kidney disease (ADPKD): computational identification of gene expression pathways and integrated regulatory networks. Hum Mol Genet. 2009;18:2328–43.
Denne C, Gerstl EM, Mayer K, Steinborn M, Hahn H, Burdach S. [Uncommon presentation of tuberous sclerosis in an infant]. Arch Pediatr. 2011;18:660–4.
Jiang S, Chiou Y, Wang E, Lin H, Lin Y, Chi Y, et al. Defining a link with autosomal-dominant polycystic kidney disease in mice with congenitally low expression of Pkd1. Am J Pathol 2006;168:205–20.
Webb BD, Metikala S, Wheeler PG, Sherpa MD, Houten SM, Horb ME, et al. Heterozygous pathogenic variant in DACT1 causes an autosomal-dominant syndrome with features overlapping Townes-Brocks syndrome. Hum Mutat. 2017;38:373–7.
Ahn E, DeKelver RC, Lo M, Nguyen TA, Matsuura S, Boyapati A, et al. SON controls cell-cycle progression by coordinated regulation of RNA splicing. Mol Cell. 2011;42:185–98.
Lu X, Göke J, Sachs F, Jacques P, Liang H, Feng B, et al. SON connects the splicing-regulatory network with pluripotency in human embryonic stem cells. Nat Cell Biol. 2013;15:1141–52.
Kim JH, Shinde DN, Reijnders M, Hauser NS, Belmonte RL, Wilson GR, et al. De novo mutations in SON disrupt RNA splicing of genes essential for brain development and metabolism, causing an intellectual-disability syndrome. Am J Hum Genet. 2016;99:711–9.
Kim JH, Park EY, Chitayat D, Stachura DL, Schaper J, Lindstrom K, et al. SON haploinsufficiency causes impaired pre-mRNA splicing of CAKUT genes and heterogeneous renal phenotypes. Kidney Int. 2019;95:1494–504.
Yıldırım Y, Ouriachi T, Woehlbier U, Ouahioune W, Balkan M, Malik S, et al. Linked homozygous BMPR1B and PDHA2 variants in a consanguineous family with complex digit malformation and male infertility. Eur J Hum Genet. 2018;26:876–85.
Backhouse B, Hanna C, Robevska G, van den Bergen J, Pelosi E, Simons C, et al. Identification of candidate genes for Mayer-Rokitansky-Küster-Hauser syndrome using genomic approaches. Sex Dev. 2019;13:26–34.
Demirhan O, Türkmen S, Schwabe GC, Soyupak S, Akgül E, Tastemir D, et al. A homozygous BMPR1B mutation causes a new subtype of acromesomelic chondrodysplasia with genital anomalies. J Med Genet. 2005;42:314–7.
Humbert C, Silbermann F, Morar B, Parisot M, Zarhrate M, Masson C, et al. Integrin α8 recessive mutations are responsible for bilateral renal agenesis in humans. Am J Hum Genet. 2014;94:288–94.
Hartner A, Cordasic N, Menendez-Castro C, Volkert G, Yabu JM, Kupraszewicz-Hutzler M, et al. Lack of α8-integrin aggravates podocyte injury in experimental diabetic nephropathy. Am J Physiol Ren Physiol. 2010;299:F1151–F1157.
Schnapp LM, Breuss JM, Ramos DM, Sheppard D, Pytela R. Sequence and tissue distribution of the human integrin α8 subunit: a β1-associated α subunit expressed in smooth muscle cells. J Cell Sci. 1995;108:537–44.
Müller U, Wang D, Denda S, Meneses JJ, Pedersen RA, Reichardt LF. Integrin α8β1 is critically important for epithelial–mesenchymal interactions during kidney morphogenesis. Cell 1997;88:603–13.
Nixon T, Richards A, Towns LK, Fuller G, Abbs S, Alexander P, et al. Bone morphogenetic protein 4 (BMP4) loss-of-function variant associated with autosomal dominant Stickler syndrome and renal dysplasia. Eur J Hum Genet. 2019;27:369–77.
Yüksel Z, Vogel F, Alhashem AM, Alanzi T, Tabarki B, Kampe K, et al. A homozygous frameshift variant in an alternatively spliced exon of DLG5 causes hydrocephalus and renal dysplasia. Clin Genet. 2019;95:631–3.
Sezaki T, Tomiyama L, Kimura Y, Ueda K, Kioka N. Dlg5 interacts with the TGF-β receptor and promotes its degradation. Febs Lett. 2013;587:1624–9.
Marquez J, Mann N, Arana K, Deniz E, Ji W, Konstantino M, et al. DLG5 variants are associated with multiple congenital anomalies including ciliopathy phenotypes. J Med Genet. 2021;58:453–64.
Murtaugh LC, Chyung JH, Lassar AB. Sonic hedgehog promotes somitic chondrogenesis by altering the cellular response to BMP signaling. Genes Dev. 1999;13:225–37.
Hartwig S, Hu M, Cella C, Piscione T, Filmus J, Rosenblum ND. Glypican-3 modulates inhibitory Bmp2-Smad signaling to control renal development in vivo. Mech Dev. 2005;122:928–38.
Tan TY, Gonzaga-Jauregui C, Bhoj EJ, Strauss KA, Brigatti K, Puffenberger E, et al. Monoallelic BMP2 variants predicted to result in haploinsufficiency cause craniofacial, skeletal, and cardiac features overlapping those of 20p12 deletions. Am J Hum Genet. 2017;101:985–94.
Le Gloan L, Pichon O, Isidor B, Boceno M, Rival JM, David A, et al. A 8.26 Mb deletion in 6q16 and a 4.95 Mb deletion in 20p12 including JAG1 and BMP2 in a patient with Alagille syndrome and Wolff-Parkinson-White syndrome. Eur J Med Genet. 2008;51:651–7.
Lybaek H, Øyen N, Fauske L, Houge G. A 2.1 Mb deletion adjacent but distal to a 14q21q23 paracentric inversion in a family with spherocytosis and severe learning difficulties. Clin Genet. 2008;74:553–9.
Griswold AJ, Ma D, Sacharow SJ, Robinson JL, Jaworski JM, Wright HH, et al. A de novo 1.5 Mb microdeletion on chromosome 14q23.2-23.3 in a patient with autism and spherocytosis. Autism Res. 2011;4:221–7.
Chesi A, Mitchell JA, Kalkwarf HJ, Bradfield JP, Lappe JM, Cousminer DL, et al. A genomewide association study identifies two sex-specific loci, at SPTB and IZUMO3, influencing pediatric bone mineral density at multiple skeletal sites. J Bone Miner Res. 2017;32:1274–81.
Zhou M, Jiang R, Zhao G, Wang L, Wang H, Li W, et al. Classification and Tie2 mutations in spinal and soft tissue vascular anomalies. Gene 2015;571:91–96.
Bergmann C, Guay-Woodford LM, Harris PC, Horie S, Peters DJM, Torres VE. Polycystic kidney disease. Nat Rev Dis Primers. 2018;4:50.
Carpenter TO, Shaw NJ, Portale AA, Ward LM, Abrams SA, Pettifor JM. Rickets. Nat Rev Dis Primers 2017;3:17101.
Ledig S, Schippert C, Strick R, Beckmann MW, Oppelt PG, Wieacker P. Recurrent aberrations identified by array-CGH in patients with Mayer-Rokitansky-Küster-Hauser syndrome. Fertil Steril. 2011;95:1589–94.
Acknowledgements
We appreciate all of the patients, their families, and clinical research coordinators who participated in this project. We thank GeneSeeq Inc. for exome sequencing technical support. We thank Beijing Ekitech Co. Ltd. for the support in bioinformatics analyses.
Funding
This research was funded in part by the National Natural Science Foundation of China (81822030 and 82072391 to N.W., 81930068 and 81772299 to Z.W., 81972132 to G.Q., 81972037 to J.Z., and 81830043 to Z.L.), National Science Foundation for Youth grant no. 81801401 (to C.N.), Beijing Natural Science Foundation (JQ20032 to N.W. and 7191007 to Z.W.), Capital’s Funds for Health Improvement and Research (2020-4-40114 to N.W.), Tsinghua University-Peking Union Medical College Hospital Initiative Scientific Research Program, CAMS Innovation Fund for Medical Sciences (CIFMS, 2020-I2M-C&T-B-030 to J.Z., 2020-I2M-C&T-A-015 to Y.M., and CAMS-2017-I2M-1-002 to L.Z.), and Non-profit Central Research Institute Fund of Chinese Academy of Medical Sciences (No. 2019PT320025).
Author information
Authors and Affiliations
Contributions
Conceptualization: N.W. and L.Z. Cohort enrollment: N.C., W.T., H.P., C.M., Z.L., S.S., Y.Y., Y.N., X.L., L.Z., Z.C., J.L., and J.D. Funding acquisition: N.W., Z.L., J.Z., G.Q., and Z.W. Genetic data analysis: S.Z. and N.W. Bioinformatics analysis: S.Z. and H.Z. Writing - review & editing: N.W. and L.Z. Writing original draft: W.T., S.Z., N.C., L.Z., and N.W. All authors provided crucial input on several iterations of the manuscript, and all authors have approved the final version.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval
Approval for the study was obtained from the ethics committee at Peking Union Medical College Hospital (ZS-1858, S-452).
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Tian, W., Chen, N., Ye, Y. et al. A genotype-first analysis in a cohort of Mullerian anomaly. J Hum Genet 67, 347–352 (2022). https://doi.org/10.1038/s10038-021-00996-w
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s10038-021-00996-w
This article is cited by
-
Update on Mayer—Rokitansky—Küster—Hauser syndrome
Frontiers of Medicine (2022)