Abstract
Background:
In neonates requiring chest compression (CC) during resuscitation, neonatal resuscitation program (NRP) recommends against relying on a single feedback device such as end-tidal carbon dioxide (ETCO2) or saturations (SpO2) to determine return of spontaneous circulation (ROSC) until more evidence becomes available.
Methods:
We evaluated the role of monitoring ETCO2 during resuscitation in a lamb model of cardiac arrest induced by umbilical cord occlusion (n = 21). Lambs were resuscitated as per NRP guidelines. Systolic blood pressure (SBP), carotid and pulmonary blood flows along with ETCO2 and blood gases were continuously monitored. Resuscitation was continued for 20 min or until ROSC (whichever was earlier). Adequate CC was arbitrarily defined as generation of 30 mmHg SBP during resuscitation. ETCO2 thresholds to predict adequacy of CC and detect ROSC were determined.
Results:
Significant relationship between ETCO2 and adequate CC was noted during resuscitation (AUC-0.735, P < 0.01). At ROSC (n = 12), ETCO2 rapidly increased to 57 ± 20 mmHg with a threshold of ≥32 mmHg being 100% sensitive and 97% specific to predict ROSC.
Conclusion:
In a large mammalian model of perinatal asphyxia, continuous ETCO2 monitoring predicted adequacy of CC and detected ROSC. These findings suggest ETCO2 in conjunction with other devices may be beneficial during CC and predict ROSC.
Similar content being viewed by others
Main
Ventilation of the lungs is the most effective action in neonatal resuscitation. In severely asphyxiated infants, chest compressions are indicated if profound bradycardia (heart rate < 60/min) or asystole persist despite adequate ventilation (1,2,3,4). The initiation of effective ventilation and chest compressions establishes blood flow through the lungs leading to gas exchange and expulsion of CO2 (5). Successful resuscitation will result in return of spontaneous circulation (ROSC). At present, there are no feedback devices approved by the neonatal resuscitation program (NRP) to assess adequacy of chest compressions and detect ROSC. As a result, chest compressions are frequently interrupted to assess heart rate and detect ROSC. Interruption of chest compressions delays the time to attain ROSC and can compromise outcomes (6,7). Studies have shown that clinical assessment of ventilation and chest compressions during resuscitation are highly subjective and inaccurate with significant variability between observers (8,9). Use of a noninvasive feedback device during resuscitation may address these limitations and assist in optimizing care and improving outcomes. An ideal feedback device when used during neonatal resuscitation may accurately assess effective ventilation and chest compressions, minimize hands off time during resuscitation and predict ROSC. The new guidelines recommend electrocardiography (EKG) monitoring during chest compressions. EKG monitoring will enhance our ability to detect ROSC but has two disadvantages—the pattern is likely to be distorted during chest compressions and it does not assess adequacy of chest compressions.
The purpose of performing chest compressions is to generate antegrade blood flow by increasing intravascular pressure (10). In this study, we used systolic blood pressure (SBP) achieved during the compression phase as a marker of adequacy of chest compressions.
We have previously demonstrated that use of capnography in addition to arterial carbon dioxide (PaCO2) monitoring in a term lamb model of meconium aspiration resulted in maintenance of carbon dioxide in a narrower range and prevented fluctuation of carotid blood flow (11). From our pilot studies, using an asphyxiated cardiac arrest model in which active resuscitation was uninterrupted, we noted that changes in end-tidal carbon dioxide (ETCO2) reflected gas exchange and also helped predict ROSC. In addition, following resuscitation, avoiding extremes of PaCO2 are important. Asphyxia is commonly associated with persistent pulmonary hypertension of the newborn (PPHN) and high PaCO2 is associated with acidosis and increase pulmonary vascular resistance (12). Hypocapnia can reduce cerebral blood flow and is associated with poor neurodevelopmental outcomes in infants with HIE (13). Monitoring ETCO2 can potentially avoid fluctuations in PaCO2 during the postresuscitation phase.
Based on these observations, our study aimed to see if capnography can be used as a feedback device during resuscitation. Our objectives were to study if (i) end-tidal (ETCO2) or expired CO2 can be used to predict adequacy of chest compressions during resuscitation, (ii) ETCO2 can predict ROSC, and (iii) ETCO2 and arterial carbon dioxide correlated postresuscitation.
Methods
This study was approved by the University at Buffalo Institutional Animal Care and Use Committee (IACUC). Time-dated pregnant ewes from May Farms (Buffalo Mills, PA) were fasted overnight and underwent cesarean section under ketamine, diazepam sedation and 2% isoflurane anesthesia.
Instrumentation
Under maternal anesthesia, term lambs (139–141 d) were partially exteriorized and instrumented while on placental circulation. Jugular venous and carotid lines were placed for intravenous access and blood draws. Carotid and pulmonary flow probes (Transonic systems, Ithaca, NY) were used to measure flows. Low lying umbilical arterial catheter was placed for transducing blood pressures. After instrumentation, the chest was closed in layers. The lambs were intubated and lung liquid drained. Cord occlusion resulted in asphyxia and cardiac arrest. Resuscitation was initiated after 5 min of cardiac arrest (Heart rate = 0 for 5 min by arterial tracing and EKG) and as per NRP protocol.
Resuscitation Protocol
Five NRP providers with designated roles were present at each delivery. The providers performed the following roles: (i) lead and direct resuscitation, (ii) provide PPV using a T-piece resuscitator, (iii) provide chest compressions, (iv) administer epinephrine, and (v) record events. Two research assistants drew blood gases and analyzed them at the bedside. An ETCO2 adapter was attached to the endotracheal tube (ETT) and lambs were ventilated with a T-piece resuscitator. We used a Philips NM3 monitor (Respironics, MA) with flow parameters.
Resuscitation was initiated with 30 s of effective PPV with peak inspiratory pressures of 35 cmH2O and PEEP of 5 cmH2O with 21% O2. If there was no ROSC, chest compressions were initiated with a 3:1 compression to ventilation ratio and inspired oxygen concentration was increased to 100%. Epinephrine (0.03 mg/kg, 1: 10,000 concentrations, 0.3 ml/kg) was administered at 3-min intervals until ROSC through an intravenous line. ROSC was defined as a sustained heart rate > 60/min with SBP of ≥ 30 mmHg. Resuscitation was continued until ROSC or until 20 min from onset of PPV (whichever was earlier).
PPV was initiated uniformly with pressures of 35/5 cmH2O with tidal volumes of 7–8 ml/kg. During resuscitation, in our study, effective SBP was defined as 50% of baseline systemic pressures prior to asphyxia which was approximately ≥ 30 mmHg. Arterial blood gases were collected and analyzed every minute until 20 min. The data were continuously collected by BIOPAC Systems (Goleta, CA) software version - 4.3.1.
Statistics
Ordinal data were analyzed using χ2-test while continuous data were analyzed using unpaired t-test. Mann–Whitney U-test was used as an alternative nonparametric test where applicable. Sensitivity and specificity analysis with receiver operator curve was generated for ETCO2 to predict effective chest compressions (SBP ≥ 30 mmHg) during resuscitation and ETCO2 to predict ROSC. A plot of the true positives against the false positives for different test cut-off points were plotted as receiver operator curve. The cut-off points were assessed based on area under the curve (AUC), accuracy and the best sensitivity/specificity. Pearson correlation analysis post ROSC for ETCO2 and PaCO2 was performed. Bland–Altman plot was used to analyze agreement between PaCO2 and ETCO2. Box plots for all ETCO2 values for SBP ≥ 30 mmHg and for ROSC vs. no ROSC were generated. Data were analyzed using SPSS 22 software (IBM, NY) and prediction analysis was done using XLSTAT (Addinsoft, NY). Probability of <5% was used for statistical significance.
Results
We studied 21 term asphyxiated lambs. Baseline characteristics such as birth weight, gender, lung liquid volume, and blood gas prior to resuscitation are shown in Table 1 . Following resuscitation with positive pressure ventilation (PPV), chest compressions and epinephrine, 12 lambs achieved ROSC while 9 did not achieve ROSC. Comparison of the characteristics between the lambs that achieved ROSC and the ones that did not, demonstrated no significant difference in baseline pH, PaCO2, base excess or difference in time to arrest, volume of lung liquid, gender or weight ( Table 1 ). The average time taken to achieve ROSC in those who survived was 4.9 ± 3 min.
Ventilation Parameters and Initial ETCO2
Tidal volumes generated by PPV for the first 5 min are shown in Figure 1 . The mean peak inspiratory pressures were constant at 35 ± 2.1 cmH2O which resulted in a tidal volume of 1.8 ± 0.5 ml/kg in the first minute and 6.9 ± 0.4 ml/kg by 5 min. At the time of initiation of ventilation, initial ETCO2 values (residual CO2— Figure 2a ) were 20 (IQR 10–30) mmHg compared with the remainder of the time where ETCO2 values were 15 (IQR 12–22) mmHg prior to ROSC or at the end of resuscitation which was significantly different (P < 0.01).
Respiratory parameters: Changes in tidal volume and peak inspiratory pressure in the first 5 min of resuscitation. The x-axis represents the time points in minutes. The primary y-axis represents the tidal volume (black triangle) in ml/kg. Secondary y-axis represents the peak inspiratory pressure (circles) (PIP) in cm H2O.
BIOPAC snapshot. (a) A representative snapshot of BIOPAC image showing changes in ETCO2, pulmonary blood flow and systolic blood pressure during resuscitation. Changes occurring with ventilation, CC, and ROSC are shown with downward pointing arrows. Residual CO2 is represented by black circle. Parenthesis denotes active resuscitation. Hyphenated arrow represents positive & negative pulmonary flow during CC. (b) A representative snapshot around the time of ROSC showing systolic blood pressure, carotid blood flow, pulmonary blood flow, EKG, and ETCO2. Downward arrow points at ROSC, detections of EKG and rise in ETCO2. CC, chest compression; ETCO2, end-tidal carbon dioxide; ROSC, return of spontaneous circulation.
Assessment of Adequacy of Chest Compressions (ETCO2 and SBP ≥ 30 mmHg)
We subsequently compared ETCO2 during chest compressions with systolic pressure generated by an umbilical arterial catheter with its tip in the ascending aorta. The ability of ETCO2 values to detect SBP ≥ 30 mmHg are tabulated with different sensitivities, specificities, positive predictive value (PPV) and negative predictive value (NPV) in Table 2 . Receiver operator curve for ETCO2 and SBP are shown ( Figure 3a ). The AUC was 0.735 (confidence interval (CI): 0.66–0.82, P < 0.01) for ETCO2 of 15 mmHg to predict SBP ≥ 30 mmHg. The ETCO2 values (12 (IQR 8–15) mmHg) associated with SBP < 30 mmHg were significantly lower than ETCO2 (18 (IQR 14–22) mmHg)) associated with SBP ≥ 30mm Hg ( Figure 3b ) (P < 0.01).
Prediction analysis ETCO2 and SBP. (a) Receiver operator curve for end-tidal CO2 (ETCO2) to predict systolic blood pressure ≥ 30 mmHg. (b) Box plots of all ETCO2 values (mmHg) associated with systolic blood pressure of <30 (black box plot) and ≥30 mmHg (white box plot). SBP = systolic blood pressure. *denotes a statistical significance P < 0.01.
During chest compressions, SBP ≥ 30 mm Hg predicted left carotid arterial blood flow of 3.2 ml/kg/min with AUC of 0.85 (CI: 0.75–0.96, P < 0.01).
ETCO2 and ROSC
The ETCO2 values for predicting ROSC are shown in Table 3 . The AUC was 0.990 for ETCO2 of 32 mmHg to predict ROSC ( Figure 4a ). The ETCO2 values before (17 (IQR 11–25) mmHg) and after (66 (IQR 59–72) mmHg) ROSC are shown in Figure 4b which were significantly different (P < 0.01).
Prediction analysis ETCO2 and ROSC. (a) Receiver operator curve for end-tidal CO2 (ETCO2) to predict return of spontaneous circulation (ROSC). (b) Box plots of all ETCO2 values before and after ROSC. *denotes a statistical significance P < 0.01.
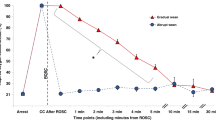
Prior to achieving ROSC (−2 min and −1 min), there was no significant difference in ETCO2 (18.7 ± 7.5 mmHg and 21.9 ± 6.9 mmHg respectively, Figure 5a ). At ROSC the ETCO2 rapidly increased to 56.8 ± 19.7 mmHg and was significantly higher than ETCO2 at −1 min prior to ROSC (P < 0.001).
Changes in ETCO2, PaCO2, and SBP. (a) Changes in ETCO2 (black squares), PaCO2 (white circles) and systolic blood pressure (SBP) (black circles) are shown in lambs that achieved return of spontaneous circulation (ROSC) and (b) in those that did not achieve ROSC. *denotes a statistical significance P < 0.01.
ETCO2 and PaCO2 Correlation Post ROSC
Analysis of data from 5–10 min after ROSC demonstrated that there was a positive correlation between ETCO2 and PaCO2 (157 pairs, R2=0.64, P < 0.001) ( Figure 6a ). Agreement between PaCO2 and ETCO2 are shown as Bland–Altman plot ( Figure 6b ). ETCO2 was lower than PaCO2 with a mean and SD of −6 ± 13 mmHg.
Post resuscitation ETCO2 & PaCO2. (a) Correlation analysis for PaCO2 and end-tidal CO2 (ETCO2) after 5 min of return of spontaneous circulation (ROSC). 157 pairs (black diamond), *R2 = 0.64, P < 0.001. (b) Bland and Altman plot showing the agreement between PaCO2 and ETCO2 (black circles). ETCO2 was lower than PaCO2 with a mean (center dark line) and SD (hyphenated lines at top and bottom) of −6 ± 13 mmHg.
ETCO2 and PaCO2 in no ROSC
The ETCO2 and PaCO2 values during the period of resuscitation in lambs that did not achieve ROSC are shown in Figure 5b . The ETCO2 during the first minute of resuscitation was 30.7 ± 6.3 mmHg and dropped to 17.1 ± 3.2 mmHg (P < 0.01) by second minute and remained below 20 mmHg for the remainder of the resuscitation.
Figure 2a is a graphic depiction of events during resuscitation of a lamb with perinatal cardiac arrest. With initiation of ventilation, there is expulsion of residual CO2 which is seen as a brisk increase in ETCO2 (14). Subsequently, during chest compressions, ETCO2, pulmonary blood flow, and SBP are low. Once ROSC is achieved, there is an abrupt increase in ETCO2 ( Figure 2a , b ), pulmonary blood flow, and SBP. Figure 2b shows the events around ROSC. Detectable EKG and rise in ETCO2 at ROSC are shown.
Discussion
Providing effective chest compressions and PPV without interruption and early detection of ROSC are essential components of neonatal resuscitation. To our knowledge, this is the first study in a lamb model of perinatal asphxial arrest to explore the utility of capnography to assess adequacy of resuscitation and detect ROSC.
Once effective ventilation is established, if the heart rate remains < 60/min, NRP recommends chest compressions to establish circulation, increase intravascular pressures and establish blood flow (5). In the absence of an intra-arterial line, it is difficult to assess the adequacy of chest compressions in the delivery room. In our study, in a fully instrumented lamb, the initiation of chest compressions caused changes in SBP, pulmonary blood flow and ETCO2 which was reflective of effective resuscitation ( Figure 2a , b ). An increase in SBP appears to be a good marker for effective chest compressions (10). In a previous prospective resuscitation study by Sutton et al. in children, effective chest compressions resulted in SBP of ≥ 80 mmHg (15). In the same study, for every 10 mm increase in depth of CC, the average SBP increased by 15 mmHg (15). They also found SBP had the most clinically relevant association with improved rate, depth, and force of chest compressions while the association with diastolic pressures was less clinically robust. In term lambs with baseline fetal systolic pressures of ~60 mmHg, and in the presence of diastolic run-off through a patent ductus, we arbitrarily assigned a SBP of ≥ 30 mmHg to define effective chest compressions. This value was approximately half of the fetal baseline SBP. In our study, ETCO2 was able to predict effective SBP (≥30 mmHg) with AUC – 0.735 ( Figure 3a ) with sensitivities ranging from 96 to 69% for a range of 10–15 mmHg during resuscitation. We understand that diastolic pressure, a more important measure of coronary perfusion, should have been targeted instead of SBP. However, in the presence of a patent ductus arteriosus, diastolic pressures were not an accurate measure as it was associated with reversal of flow.
Many studies involving cardiac arrest models after completion of perinatal transition have shown that ETCO2 is useful in predicting ROSC with high sensitivity and specificity (16,17,18). Chalak et al. have reported an ETCO2 value of 14 mmHg with high sensitivity and specificity to predict ROSC. In this model by Chalak et al., resuscitation was initiated after 1 min of asystole, while in our study resuscitation was initiated after 5 min of asystole. In our study, in the presence of fetal lung liquid, ETCO2 of 32 mmHg had 100% sensitivity and near 100% specificity. Our results were similar to previous animal models (16,19) where resuscitation was initiated 5–10 min after asphyxia. When ROSC is achieved there is a dramatic rise in ETCO2 ( Figure 6 ) and this finding may serve as a useful tool to assess ROSC without interrupting resuscitation in delivery room. In the clinical setting, the importance of ETCO2 during resuscitation has been validated in adults by Falk et al. (20). A retrospective study involving neonates using Pedi-cap a colorimetric CO2 detector has reported color change just prior to ROSC neonatal resuscitation (21) which points to the fact that a rise in ETCO2 regardless of the value can aid in confirming ROSC along with clinical cues without interrupting resuscitation.
We have previously shown that ETCO2 correlated well with PaCO2 in an ovine meconium aspiration model poststabilization (11). In this study using an asphyxia model, ETCO2 correlated well with PaCO2 after 5 min of established ROSC. Infants with asphyxia are at high risk of suffering from associated neurologic injury. Optimal management of these infants after resuscitation includes maintenance of appropriate perfusion, oxygenation and maintaining carbon dioxide levels (13). In these critically ill patients, having a noninvasive feedback device may be helpful to monitor efficacy of resuscitation. While blood gas parameters remain the standard of evaluating gas exchange, ETCO2 monitoring may also serve as a valuable tool in reducing fluctuations in cerebral and pulmonary blood flow, when trended along with intermittent blood gas (PaCO2) monitoring.
NRP recently recommended using EKG monitoring during resuscitation to accurately assess heart rate (1). EKG assesses heart rate with higher sensitivity compared with pulse oximetry (22). EKG is likely to be more sensitive in detecting ROSC. However, there are three potential limitations with exclusive use of EKG for detecting ROSC. (i) A case series by Koizumi et al. (23) reported a neonate with pulseless electrical activity with no palpable pulse or heart rate on auscultation. EKG only measures cardiac electrical activity and not perfusion of essential organs. (ii) EKG pattern is difficult to detect during active chest compressions; a brief interruption during chest compressions may be necessary to detect heart rate on EKG (probably during the ventilation period during 3:1 chest compressions to ventilation as recommended by NRP) ( Figure 2b ). (iii) Finally, EKG does not assess the adequacy and hemodynamic effectiveness of chest compressions. Hence, we recommend a combination of EKG and ETCO2 monitoring during neonatal resuscitation. The use of multiple devices to detect ROSC is more effective compared with a single feedback device.
A recent study by Li et al. (24) using capnography in a transitioned porcine model has showed significantly better respiratory and hemodynamic parameters during resuscitation in the surviving as compared to nonsurviving piglets. Monitoring ETCO2 and respiratory parameters along with optimal oxygen saturations (25) may be useful during and postresuscitation stabilization.
In interpreting the findings of our study we need to acknowledge several limitations. While we have identified ETCO2 values for predicting SBP of 30 mmHg (roughly half of fetal baseline value) during resuscitation, these are likely to vary by models and may not be true in neonates. Although use of a transitional model is the strength of this study, the presence of a patent ductus may have resulted in diastolic run off during chest compressions. While these findings simulate delivery room resuscitation, it may not reflect resuscitation of an older neonate in the intensive care unit.
Conclusion
In a large mammalian transitional model of asphyxial cardiac arrest, our study suggests a role for capnography in resuscitation as ETCO2 predicted adequacy of chest compressions as measured by generation of SBP. In addition, a rise in ETCO2 identified ROSC with 100% sensitivity and 97% specificity. Post ROSC, ETCO2 correlates well with PaCO2 and may assist in the ongoing management of those who are resuscitated. Future clinical studies monitoring ETCO2 along with EKG may assist in providing uninterrupted resuscitation and efficient detection of ROSC.
Statement of Financial Support
This work was supported by the Canadian Pediatric Society—Neonatal Resuscitation Program (SL) and 1R01HD072929 (SL), American Academy of Pediatrics—Neonatal Resuscitation Program (MR), and Henry C. and Bertha H. Buswell Fellowship—University at Buffalo—Salary Support (PC).
Disclosure
Authors have no other financial disclosure.
References
Perlman JM, Wyllie J, Kattwinkel J, et al.; Neonatal Resuscitation Chapter Collaborators. Part 7: Neonatal Resuscitation: 2015 International Consensus on Cardiopulmonary Resuscitation and Emergency Cardiovascular Care Science With Treatment Recommendations. Circulation 2015;132(16 Suppl 1):S204–41.
Perlman JM, Wyllie J, Kattwinkel J, et al.; Neonatal Resuscitation Chapter Collaborators. Part 7: Neonatal Resuscitation: 2015 International Consensus on Cardiopulmonary Resuscitation and Emergency Cardiovascular Care Science With Treatment Recommendations (Reprint). Pediatrics 2015;136 Suppl 2:S120–66.
Wyckoff MH, Aziz K, Escobedo MB, et al. Part 13: Neonatal Resuscitation: 2015 American Heart Association Guidelines Update for Cardiopulmonary Resuscitation and Emergency Cardiovascular Care. Circulation 2015;132(18 Suppl 2):S543–60.
Wyllie J, Perlman JM, Kattwinkel J, et al.; Neonatal Resuscitation Chapter Collaborators. Part 7: Neonatal Resuscitation: 2015 International Consensus on Cardiopulmonary Resuscitation and Emergency Cardiovascular Care Science with Treatment Recommendations. Resuscitation 2015;95:e169–201.
Wyckoff MH. Neonatal cardiopulmonary resuscitation: critical hemodynamics. NeoReviews 2010;11:123–129.
Berg RA, Hemphill R, Abella BS, et al. Part 5: adult basic life support: 2010 American Heart Association Guidelines for cardiopulmonary resuscitation and emergency cardiovascular care. Circulation 2010;122(18 Suppl 3):S685–705.
Garza AG, Gratton MC, Salomone JA, Lindholm D, McElroy J, Archer R. Improved patient survival using a modified resuscitation protocol for out-of-hospital cardiac arrest. Circulation 2009;119:2597–605.
Abella BS, Sandbo N, Vassilatos P, et al. Chest compression rates during cardiopulmonary resuscitation are suboptimal: a prospective study during in-hospital cardiac arrest. Circulation 2005;111:428–34.
Cheng A, Hunt EA, Grant D, et al.; International Network for Simulation-based Pediatric Innovation, Research, and Education CPR Investigators. Variability in quality of chest compressions provided during simulated cardiac arrest across nine pediatric institutions. Resuscitation 2015;97:13–9.
Maher KO, Berg RA, Lindsey CW, Simsic J, Mahle WT. Depth of sternal compression and intra-arterial blood pressure during CPR in infants following cardiac surgery. Resuscitation 2009;80:662–4.
Chandrasekharan PK, Rawat M, Nair J, et al. Continuous end-tidal carbon dioxide monitoring during resuscitation of asphyxiated term lambs. Neonatology 2016;109:265–73.
Rudolph AM, Yuan S. Response of the pulmonary vasculature to hypoxia and H+ ion concentration changes. J Clin Invest 1966;45:399–411.
Pappas A, Shankaran S, Laptook AR, et al.; Eunice Kennedy Shriver National Institute of Child Health and Human Development Neonatal Research Network. Hypocarbia and adverse outcome in neonatal hypoxic-ischemic encephalopathy. J Pediatr 2011;158:752–758.e1.
Berg RA, Henry C, Otto CW, et al. Initial end-tidal CO2 is markedly elevated during cardiopulmonary resuscitation after asphyxial cardiac arrest. Pediatr Emerg Care 1996;12:245–8.
Sutton RM, French B, Nishisaki A, et al. American Heart Association cardiopulmonary resuscitation quality targets are associated with improved arterial blood pressure during pediatric cardiac arrest. Resuscitation 2013;84:168–72.
Bhende MS, Karasic DG, Menegazzi JJ. Evaluation of an end-tidal CO2 detector during cardiopulmonary resuscitation in a canine model for pediatric cardiac arrest. Pediatr Emerg Care 1995;11:365–8.
Chalak LF, Barber CA, Hynan L, Garcia D, Christie L, Wyckoff MH. End-tidal CO2 detection of an audible heart rate during neonatal cardiopulmonary resuscitation after asystole in asphyxiated piglets. Pediatr Res 2011;69(5 Pt 1):401–5.
Hamrick JL, Hamrick JT, Lee JK, Lee BH, Koehler RC, Shaffner DH. Efficacy of chest compressions directed by end-tidal CO2 feedback in a pediatric resuscitation model of basic life support. J Am Heart Assoc 2014;3:e000450.
Bhende MS, Karasic DG, Karasic RB. End-tidal carbon dioxide changes during cardiopulmonary resuscitation after experimental asphyxial cardiac arrest. Am J Emerg Med 1996;14:349–50.
Falk JL, Rackow EC, Weil MH. End-tidal carbon dioxide concentration during cardiopulmonary resuscitation. N Engl J Med 1988;318:607–11.
Blank D, Rich W, Leone T, Garey D, Finer N. Pedi-cap color change precedes a significant increase in heart rate during neonatal resuscitation. Resuscitation 2014;85:1568–72.
Katheria A, Rich W, Finer N. Electrocardiogram provides a continuous heart rate faster than oximetry during neonatal resuscitation. Pediatrics 2012;130:e1177–81.
Koizumi M, Mizumoto H, Araki R, Kan H, Akashi R, Hata D. The utility of electrocardiogram for evaluation of clinical cardiac arrest in neonatal resuscitation. Resuscitation 2016;104:e3–4.
Li ES, Cheung PY, O’Reilly M, et al. Exhaled CO2 parameters as a tool to assess ventilation-perfusion mismatching during neonatal resuscitation in a Swine model of neonatal asphyxia. PLoS One 2016;11:e0146524.
Lakshminrusimha S, Swartz DD, Gugino SF, et al. Oxygen concentration and pulmonary hemodynamics in newborn lambs with pulmonary hypertension. Pediatr Res 2009;66:539–44.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chandrasekharan, P., Vali, P., Rawat, M. et al. Continuous capnography monitoring during resuscitation in a transitional large mammalian model of asphyxial cardiac arrest. Pediatr Res 81, 898–904 (2017). https://doi.org/10.1038/pr.2017.26
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/pr.2017.26
This article is cited by
-
Non-invasive carbon dioxide monitoring in neonates: methods, benefits, and pitfalls
Journal of Perinatology (2021)
-
Two-site regional oxygen saturation and capnography monitoring during resuscitation after cardiac arrest in a swine pediatric ventricular fibrillatory arrest model
Journal of Clinical Monitoring and Computing (2020)
-
Effect of various inspired oxygen concentrations on pulmonary and systemic hemodynamics and oxygenation during resuscitation in a transitioning preterm model
Pediatric Research (2018)
-
A family of NICU graduates!
Pediatric Research (2018)