Abstract
Preeclampsia, a pregnancy-specific syndrome of hypertension and proteinuria, is characterized by defective placental vasculogenesis and widespread maternal endothelial dysfunction. Although the manifestations of preeclampsia are primarily maternal, the burden of morbidity and mortality is often on the neonate, since the only effective treatment—delivery of the fetus and placenta—often results in iatrogenic prematurity. In this review, we summarize recent advances in our understanding of the pathophysiology of preeclampsia, including normal and aberrant placental vascular development and evidence for endothelial dysfunction. We describe recent evidence that supports a novel mechanism in which a maladaptive shift in placental production of angiogenic factors such as soluble fms-like tyrosine kinase 1 (a circulating antiangiogenic protein) may play an important role in the pathogenesis of preeclampsia.
Similar content being viewed by others
Main
Preeclampsia affects 3–5% of all pregnancies (1) and is classically defined as the new onset of hypertension and proteinuria after wk 20 of gestation. Since E.W. Page first posited in 1939 that a placental defect might cause preeclampsia (2), our understanding of normal placental vasculogenesis and of the pathophysiology of preeclampsia has advanced significantly. Here we will review recent advances and summarize our current understanding of the pathogenesis of preeclampsia. We will describe evidence that preeclampsia may involve an imbalance among the pro- and antiangiogenic factors that regulate placental and systemic endothelial health.
CLINICAL FEATURES AND EPIDEMIOLOGY OF PREECLAMPSIA
Preeclampsia is characterized by the new onset of hypertension and proteinuria during the last trimester of pregnancy. It is also usually associated with hyperuricemia and edema. Severe preeclampsia may also lead to SGA babies. The clinical onset of preeclampsia is often insidious and asymptomatic, but may include headache, visual disturbances, epigastric pain, weight gain, and edema of the hands and face. These early signs and symptoms are important to recognize clinically, since they may herald progression to a more severe and often life-threatening disease. Severe complications of preeclampsia can include acute renal failure; cerebral edema, cerebral hemorrhage, and seizures (eclampsia); pulmonary edema; thrombocytopenia, hemolytic anemia, coagulopathy; and liver injury, including HELLP, the syndrome of hemolysis, elevated liver enzymes, and low platelets. Although antihypertensive medications help to lower blood pressure and magnesium sulfate is effective in seizure prophylaxis (3), delivery remains the only definitive treatment. When preeclampsia threatens to lead to severe maternal complications, urgent delivery of the fetus and placenta are often undertaken to preserve maternal health.
In the developed world, where safe emergent cesarean delivery is available, the burden of morbidity and mortality due to preeclampsia is on the neonate. Preeclampsia is associated with placental hypoperfusion, which can lead to intrauterine growth restriction and oligohydramnios. Abruptio placentae complicates about 4% of cases of severe preeclampsia (4). Neonatal morbidity is most often due to the sequelae of prematurity and low birth weight, including prolonged neonatal intensive care unit stays, respiratory distress, necrotizing enterocolitis, intraventricular hemorrhage, sepsis, and death (5,6). The HELLP syndrome has been associated with a 10–20% incidence of perinatal mortality, attributable largely to premature delivery (7,8).
The epidemiology of preeclampsia provides clues about the pathophysiology that scientists are still deciphering. Although most preeclampsia occurs in healthy nulliparous women, several risk factors are reminiscent of cardiovascular risk factors, including chronic hypertension [odds ratio (OR), 2.7] (9,10), renal disease (OR, 7.8) (11,12), diabetes mellitus (13), high body mass index and obesity (OR, 2.1–3.2) (9,14,15), and family history of cardiovascular disease (OR, 1.9) (16). The importance of cardiovascular risk factors has strengthened the hypothesis that preexisting maternal vascular dysfunction or susceptibility may have a pathologic role in at least some cases of preeclampsia. The association of preeclampsia with primiparity, increased interpregnancy interval, and possibly primipaternity (17) has generated hypotheses regarding dysregulation of placental immune tolerance, which remains an area for further study.
ROLE OF THE PLACENTA
Several early observations support the hypothesis for a placental trigger for preeclampsia. Preeclampsia only occurs in the presence of a placenta (though not necessarily a fetus, as in the case of hydatidiform mole). It almost always remits after delivery of the placenta. In cases of preeclampsia with extrauterine pregnancy, removal of the fetus alone is insufficient; symptoms persist until the placenta is delivered (18).
Long-standing and severe preeclampsia is associated with pathologic evidence of placental hypoperfusion and ischemia. Findings include acute atherosis, a lesion of diffuse vascular obstruction first described in 1945 (19): fibrin deposition, intimal thickening, necrosis, and endothelial damage. Infarcts, likely resulting directly from occlusion of maternal spiral arteries (20), are also common. Although these findings are not universal, they appear to be correlated with severity of clinical disease (21).
Abnormal uterine artery Doppler ultrasound, suggesting increased uteroplacental resistance to blood flow, is observed before the onset of clinical signs of preeclampsia (22), although this finding is nonspecific, limiting its use as a clinical screening test (23). In a recent study, abnormalities in uterine artery Doppler correlated with poor clinical outcome among women with pregnancy-induced hypertension and preeclampsia (24). Hypertension and proteinuria can be induced by constriction of uterine blood flow in pregnant primates (25,26) and rodents (27,28). These observations suggest placental ischemia may be an early, and perhaps precipitating, event. However, evidence for a causative role for placental ischemia remains circumstantial, and several observations call the hypothesis into question. For example, the animal models based on uterine hypoperfusion universally fail to induce several of the multiorgan features of preeclampsia, including seizures and the HELLP syndrome. In most cases of human preeclampsia, there is no evidence of growth restriction or fetal intolerance of labor, which would be expected from significant placental hypoperfusion. It may be that the pathologic evidence of placental ischemic damage that accompanies late-stage preeclampsia may be a secondary event: no studies have examined placental changes before the onset of clinical signs of preeclampsia. Nevertheless, recent studies provide evidence for inadequate spiral artery remodeling, the understanding of which requires a closer review of normal placentation.
PLACENTAL VASCULAR REMODELING
Early in placentation, extravillous cytotrophoblasts invade the uterine spiral arteries of the decidua and myometrium. These invasive fetal cells replace the endothelial layer of the uterine vessels, transforming them from small resistance vessels to flaccid, high-caliber capacitance vessels (29,30). This vascular transformation allows the increase in uterine blood flow needed to sustain the fetus through pregnancy.
In preeclampsia, this transformation is incomplete (31,32). Cytotrophoblast invasion of the arteries is limited to the superficial decidua, and the myometrial segments remain narrow and undilated (33,34). Fisher and colleagues (35,36) have shown that in normal placental development, invasive cytotrophoblasts down-regulate the expression of adhesion molecules characteristic of their epithelial cell origin and adopt an endothelial cell-surface adhesion phenotype, a process referred to as pseudovasculogenesis. In preeclampsia, cytotrophoblasts fail to undergo this switching of cell-surface integrins and adhesion molecules (37) and fail to adequately invade the myometrial spiral arteries.
The factors that regulate this process are just beginning to be elucidated. Hypoxia-inducible factor-1 (HIF-1) is up-regulated in preeclampsia and HIF-1 target genes such as transforming growth factor beta 3 may block cytotrophoblast invasion (38,39). Invasive cytotrophoblasts express several angiogenic factors and receptors, including VEGF-A, PlGF, and VEGFR-1 (Flt1); expression of these proteins by immunolocalization is altered in preeclampsia (40). Interestingly, uteroplacental ischemia produced in monkeys by aortic constriction late in gestation appears to enhance trophoblast invasion, thus producing a pathologic change quite different from that observed in preeclampsia (41). This again suggests the inadequacy of animal models based on aortic or uterine vessel constriction.
MATERNAL ENDOTHELIAL DYSFUNCTION
Placental pathology notwithstanding, preeclampsia is a multisystem disorder, and its manifestations reflect widespread endothelial dysfunction, often resulting in vasoconstriction and end-organ ischemia (42,43). Several serum markers of endothelial activation are altered in women with preeclampsia, including von Willebrand antigen (44), cellular fibronectin (45), soluble tissue factor, soluble E-selectin, platelet-derived growth factor, and endothelin (46). There is evidence for oxidative stress (47), increased lipid peroxidation (48), and platelet activation (49). Leptin levels are increased as early as 20 wk (50). Decreased PGI2 (an endothelial cell–derived prostaglandin) production well before the onset of clinical symptoms is consistent with the hypothesis that dysfunctional endothelial cells are central to the pathogenesis of the syndrome (51). Maternal vascular reactivity to the vasopressors such as angiotensin II and norepinephrine is increased in preeclampsia (52). Endothelium-dependent vasorelaxation is impaired, both in the myometrial vessels in vitro (53) and in forearm blood flow in vivo (54).
In the kidney, endothelial damage results in proteinuria and produces the characteristic pathologic lesion, glomerular endotheliosis (55,56). Glomerular endotheliosis is characterized by generalized swelling and vacuolization of the endothelial cells with obliteration of the endothelial fenestrae and loss of the capillary space. Mesangial cells may occasionally show nonspecific changes that are likely related to the proteinuria. There are deposits of fibrinogen and fibrin within and under the endothelial cells (57). The injury is specific to endothelial cells: the foot processes of the podocytes are almost completely normal early in disease, an unusual finding in proteinuric renal disease. Although once considered pathognomonic for preeclampsia, recent studies have shown that mild glomerular endotheliosis may also occur in a significant percentage of normal pregnancies at term and that it is more severe in preeclamptics (58). This suggests the endothelial dysfunction of preeclampsia may in fact be an exaggeration of a process present near term in all pregnancy.
Long-term outcomes among women with a history of preeclampsia suggest the endothelial changes are not limited to pregnancy. Cardiovascular morbidity and mortality are increased among such women, including stroke (59), ischemic heart disease (60,61), and chronic hypertension (62). Impaired endothelium-dependent vasodilation, a marker for endothelial dysfunction, persists for years postpartum (54,63) It is also interesting to note that women with preeclampsia appear to have a decreased risk of malignancy in several studies (59,64,65), consistent with an antiangiogenic milieu that may extend beyond the pregnancy itself. Whether these long-term observations are due to endothelial damage done as a result of preeclampsia, or simply reflect the consequences of the vascular risk factors that are more common in these women remains speculative.
LINKING THE PLACENTA AND THE MATERNAL RESPONSE
These two lines of evidence—the central role of the placenta and the subsequent endothelial dysfunction responsible for the end-organ damage—have converged to produce a pathophysiologic paradigm that has been the framework for investigation over the past few decades. The placenta, possibly as a result of ischemia, secretes a factor or factors into the maternal circulation that directly or indirectly damages maternal endothelial cells. This paradigm begs two essential questions about the pathophysiology of preeclampsia: first, why does placental vasculogenesis fail, resulting in incomplete remodeling of decidual arteries? And second, what is the placental factor that produces the clinical manifestations of disease?
In vitro data provide compelling supportive evidence for a factor in preeclamptic serum that produces endothelial dysfunction. Plasma from women with preeclampsia increases production of nitric oxide (66), cellular fibronectin (67), prostacyclin (68), and other markers of activation/dysfunction in vitro. Incubation of uterine vessels with plasma from women with preeclampsia results in impaired endothelium-dependent vasorelaxation (69). Many other observations also support this hypothesis and are recently reviewed elsewhere (70).
The identity of the “preeclampsia factor” has remained elusive, however. Several candidate factors have been studied. Page and colleagues (71) found that levels of neurokinin-B (NK-B), a neurotransmitter apparently produced by the placenta, were elevated in women with preeclampsia, and that rats infused with high doses of NK-B developed transient hypertension. However, their findings have not borne out in subsequent studies (72,73). There is also recent data that increased syncytiotrophoblast shedding in preeclampsia as a consequence of placental apoptosis may contribute to endothelial dysfunction in cell culture studies. However, there is no in vivo evidence so far that circulating placental debris induce preeclampsia-like syndrome (74). AbdAlla et al. (75) have found increased heterodimerization between the angiotensin AT-1 receptor and the bradykinin B-2 receptors; this resulted in increased responsiveness to angiotensin-2. The T235 polymorphism of the angiotensinogen gene also appears to increase risk (76). However, no single gene mutation/polymorphism has been consistently associated with preeclampsia in all populations. Patients with preeclampsia have also been reported to have circulating AT1-AA (77). These autoantibodies are thought to antagonize the AT1 receptor and have been hypothesized to participate in the angiotensin II–induced vascular lesions in preeclamptic patients (78,79). However, direct causal evidence implicating AT1-AA in preeclampsia has not yet been established. Moreover, the temporal relationship of these auto-antibodies with the clinical syndrome of preeclampsia has not been studied.
sFLT1: AN ANTI-ANGIOGENIC FACTOR AND ITS ROLE IN PREECLAMPSIA
The search for the elusive preeclampsia factor led our group to use gene expression profiling of placental tissue using microarray technology. Using this approach, the antiangiogenic protein sFlt1 (soluble fms-like tyrosine kinase or sVEGFR1) was found to be up-regulated in the placenta of women with preeclampsia (80). In addition, circulating levels of sFlt-1 were found to be elevated in conjunction with decreased VEGF and PlGF in the bloodstream at the time of disease presentation, a finding that has been reported by multiple groups (80–84). The in vitro effects of sFlt1 included vasoconstriction and endothelial dysfunction, mimicking the effects of plasma from women with preeclampsia (80). Adenoviral gene transfer of sFlt1 to pregnant rats produced a syndrome resembling preeclampsia, including hypertension, proteinuria, and glomerular endotheliosis (80). Furthermore, a soluble form of VEGF receptor-2 antagonist (sFlk-1), which does not antagonize PlGF when given exogenously, did not induce a preeclamptic phenotype in pregnant rats. This suggests that antagonism of both VEGF and PlGF is necessary to induce the maternal syndrome. This work has generated considerable enthusiasm for sFlt1 as an important mediator in preeclampsia. Much of the remainder of this article will explore the nature of this protein and how it might improve our understanding of preeclampsia.
sFlt1 is an antiangiogenic molecule that antagonizes VEGF and PlGF. VEGF is important in both angiogenesis (the growth of new blood vessels) and in the maintenance of endothelial cell health in the basal state. Although the function of PlGF is still ill defined, it appears to act synergistically with VEGF and may be necessary for wound healing and angiogenesis in ischemic tissues (85,86).
VEGF has a family of receptors, the most important of which are Flt1 (VEGFR1) and Flk1 (VEGFR2) (87). sFlt1 is a truncated form of the Flt1 receptor (Fig. 1). It includes the extracellular ligand-binding domain, but not the transmembrane and intracellular domains; it is secreted (hence “soluble”) and antagonizes VEGF and PlGF in the circulation by binding and preventing their interaction with their endothelial receptors (88). The regulation of Flt1 splicing to produce the full Flt1 receptor versus the truncated sFlt1 remains unexplored. It was recently shown that hypoxic trophoblasts grown in vitro produces excess quantities of sFlt-1 (89), however, whether this phenomenon occurs in vivo remains unknown. Although sFlt1 is made in small amounts by other tissues (endothelial cells and monocytes), the placenta seems to be the major source of circulating sFlt1 during pregnancy, as evidenced by the dramatic fall in circulating concentrations of sFlt1 after the delivery of the placenta (80).
Protein structure of Flt-1 and sFlt-1 are illustrated. Flt-1 has 7 immunoglobulin domains (IgG), which are thought to mediate ligand binding to VEGF and PlGF. sFlt-1 protein has a unique 31 AA C-terminus region derived from alternative splicing and lacks the transmembrane and cytoplasmic domains. Figure reproduced with permission from Karumanchi SA et al. 2004 Pathogenesis of preeclampsia. In: Rose BD (ed) UpToDate. UpToDate, Wellesley, MA. ©2004 UpToDate Inc.
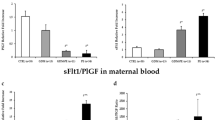
sFlt1 levels are increased, and free (unbound) PlGF and free VEGF levels are suppressed, in the serum of women during clinical preeclampsia (80). The changes in these markers precede the onset of clinical disease by at least 5 wk (90); in fact, decreased free PlGF levels are observed even before 20 wk gestation in women who go on to develop preeclampsia (84,90,91). The diminished PlGF levels early in pregnancy are not accompanied by reciprocal increases in systemic sFlt1. This may reflect either less placental production of PlGF or increased binding to local circulating and membrane-bound receptors, but the precise mechanisms for this remain unexplored.
There is much supportive evidence suggesting VEGF and PlGF antagonism by sFlt1 may produce the endothelial dysfunction in preeclampsia. VEGF is highly expressed by glomerular podocytes, and VEGF receptors are present on glomerular endothelial cells (92,93). In experimental glomerulonephritis, VEGF is necessary for glomerular capillary repair (94,95). In antiangiogenesis cancer trials, VEGF antagonists produce proteinuria and hypertension in human subjects (96,97). Recent data suggests that VEGF may be particularly important in maintaining the health of fenestrated endothelium (98), which is found in the renal glomerulus, brain, and liver—organs disproportionately affected in preeclampsia. It is tempting to speculate that cigarette smoking may exert its protective influence on preeclampsia risk (99) by its pro-angiogenic effects (100). A 50% reduction of renal VEGF production in genetically modified mice resulted in glomerular endotheliosis and proteinuria, providing genetic evidence that impaired VEGF signaling may lead to preeclampsia-like phenotype (101). Finally, trisomy 13 has long been associated with preeclampsia (102), suggesting that a protein on this chromosome may be important. The Flt1/sFlt1 gene is located on chromosome 13q12. Hence, one could hypothesize that 50% higher concentrations of circulating sFlt1 in trisomy 13 patients may account for the increased risk of preeclampsia; however, definitive proof for this hypothesis is lacking. On the other hand, other reported genetic associations with preeclampsia have no apparent link to sFlt1. For example, both an Australian/New Zealand cohort (103) and an Icelandic cohort (104) have suggested a maternal susceptibility locus on chromosome 2, bearing no known relationship to sFlt1. Although it is possible that such loci are associated with transcription factors or splicing factors affecting sFlt1 production, it seems more likely that there are other yet unidentified genetic factors that contribute to this multifactorial disease.
The excess sFlt1 theory certainly does not offer a complete picture of preeclampsia. There are several elements that remain unexplained. No coagulation, liver function, or brain abnormalities (eclampsia) were reported in sFlt-1-treated animals. Elements of maternal predisposition such as insulin resistance and hypertension seem to be independent of the placenta (105). Preeclampsia is associated not only with primiparity, but also with increased interpregnancy interval (106) and shorter cohabitation of partners before pregnancy (107). Some have suggested that this implies some role for reduced maternal-fetal immune tolerance or decreased exposure to paternal sperm antigens. It is interesting to note, however, that primiparous pregnant women appear to have higher baseline levels of sFlt1 compared with multiparous pregnant women, which may account for the increase in risk (91). Several maternal cardiovascular risk factors are also risk factors for preeclampsia, most notably diabetes mellitus, hypertension, and renal disease. It is possible that placental factors such as PlGF and sFlt1 interact with elements of maternal susceptibility in complex ways to produce a disease that varies dramatically in its timing and severity (108). Finally, the relationship of sFlt-1 with previously hypothesized mechanisms of systemic damage in preeclampsia such as increased angiotensin II sensitivity, increased circulating AT1-AA, hyperuricemia, and increased sympathetic activation is unknown at the present time.
ROLE OF ANGIOGENIC FACTORS IN PLACENTAL VASCULAR DEVELOPMENT
VEGF ligands and receptors are highly expressed by placental tissue in the first trimester (40). Thus, it is intuitive to hypothesize that placental vascular development might be regulated by a local balance between pro- and antiangiogenic factors, and that excess sFlt1 in early gestation could contribute to inadequate placental vasculogenesis. Circulating sFlt1 levels are relatively low early in pregnancy and begin to distinctly rise in the third trimester. Although the reason for this increase is unclear, we posit that it may reflect an antiangiogenic shift in the placental milieu toward the end of pregnancy, corresponding to completion of the vasculogenic phase of placental growth. In preeclampsia, this rise in sFlt1 production is early and exaggerated. Furthermore, circulating concentrations of free PlGF levels is altered well before 20 wk of gestation in preeclamptic patients. We therefore have hypothesized that this early alteration in the angiogenic balance may therefore contribute to inadequate vascular cytotrophoblast invasion in the early stages of pregnancy, and its overflow into the circulation may produce maternal endothelial dysfunction in the third trimester. In this case, placental ischemia may not be causative, but rather one of the several mechanisms affected by this deranged angiogenic balance. Although in vitro studies using primary cytotrophoblast cultures and villous explants have demonstrated a role for sFlt1 in interfering with trophoblast invasion (40,109,110), definitive in vivo evidence for this hypothesis is lacking.
CONCLUSIONS
In summary, we believe that preeclampsia is a state of endothelial dysfunction secondary to excessive amounts of circulating antiangiogenic factors of placental origin, such as sFlt1 (110). If other investigators confirm the importance of sFlt1 in preeclampsia, the clinical implications could be significant. Currently, there is no useful and practical screening test for preeclampsia. The syndrome can be silent in its early stages, and it sometimes presents as severe and even explosive disease. If serum sFlt1 or PlGF levels were found to be sensitive and specific predictors of preeclampsia in prospective clinical trials, the efficacy of various nonspecific interventions could be investigated, including bedrest and antihypertensive and antiplatelet therapy. Of course, a screening/diagnostic test will become truly useful only when an effective treatment becomes available. Investigations are currently underway of various pharmacologic agents to counteract the effects of sFlt1 as a treatment for preeclampsia. If such agents are effective in alleviating the end-organ manifestations to safely postpone delivery for even a few weeks, a significant impact on neonatal morbidity and mortality could be made.
Clearly, a multifactorial approach is required as we continue to deepen our understanding of the preeclampsia syndrome. As our understanding continues to advance based on molecular and genetic techniques, we are hopeful that new interventions may improve our management of this important syndrome in the near future.
Abbreviations
- AT1-AA:
-
angiotensin receptor 1 auto-antibody
- PlGF:
-
placental growth factor
- sFlt1:
-
soluble fms-like tyrosine kinase 1
- SGA:
-
small for gestational age
- VEGF:
-
vascular endothelial growth factor
References
Walker JJ 2000 Pre-eclampsia. Lancet 356: 1260–1265
Page EW 1939 The relation between hydatid moles, relative ischemia of the gravid uterus and the placental origin of eclampsia. Am J Obstet Gynecol 37: 291–293
Sibai BM 2003 Diagnosis and management of gestational hypertension and preeclampsia. Obstet Gynecol 102: 181–192
Sibai BM, Mercer BM, Schiff E, Friedman SA 1994 Aggressive versus expectant management of severe preeclampsia at 28 to 32 weeks' gestation: a randomized controlled trial. Am J Obstet Gynecol 171: 818–822
Sibai BM, Taslimi M, Abdella TN, Brooks TF, Spinnato JA, Anderson GD 1985 Maternal and perinatal outcome of conservative management of severe preeclampsia in midtrimester. Am J Obstet Gynecol 152: 32–37
Friedman SA, Schiff E, Kao L, Sibai BM 1995 Neonatal outcome after preterm delivery for preeclampsia. Am J Obstet Gynecol 172: 1785–1788
Roelofsen AC, van Pampus MG, Aarnoudse JG 2003 The HELLP-syndrome; maternal-fetal outcome and follow up of infants. J Perinat Med 31: 201–208
Abramovici D, Friedman SA, Mercer BM, Audibert F, Kao L, Sibai BM 1999 Neonatal outcome in severe preeclampsia at 24 to 36 weeks' gestation: does the HELLP (hemolysis, elevated liver enzymes, and low platelet count) syndrome matter?. Am J Obstet Gynecol 180: 221–225
Sibai BM, Ewell M, Levine RJ, Klebanoff MA, Esterlitz J, Catalano PM, Goldenberg RL, Joffe G 1997 Risk factors associated with preeclampsia in healthy nulliparous women. The Calcium for Preeclampsia Prevention (CPEP) Study Group. Am J Obstet Gynecol 177: 1003–1010
Caritis S, Sibai B, Hauth J, Lindheimer M, VanDorsten P, Klebanoff M, Thom E, Landon M, Paul R, Miodovnik M, Meis P, Thurnau G, Dombrowski M, McNellis D, Roberts J 1998 Predictors of pre-eclampsia in women at high risk. National Institute of Child Health and Human Development Network of Maternal-Fetal Medicine Units. Am J Obstet Gynecol 179: 946–951
Mostello D, Catlin TK, Roman L, Holcomb WL Jr, Leet T 2002 Preeclampsia in the parous woman: who is at risk?. Am J Obstet Gynecol 187: 425–429
Cunningham FG, Cox SM, Harstad TW, Mason RA, Pritchard JA 1990 Chronic renal disease and pregnancy outcome. Am J Obstet Gynecol 163: 453–459
Sibai BM 2000 Risk factors, pregnancy complications, and prevention of hypertensive disorders in women with pregravid diabetes mellitus. J Matern Fetal Med 9: 62–65
Weiss JL, Malone FD, Emig D, Ball RH, Nyberg DA, Comstock CH, Saade G, Eddleman K, Carter SM, Craigo SD, Carr SR, D'Alton ME, FASTER Research Consortium 2004 Obesity, obstetric complications and cesarean delivery rate—a population-based screening study. Am J Obstet Gynecol 190: 1091–1097
Thadhani R, Stampfer MJ, Hunter DJ, Manson JE, Solomon CG, Curhan GC 1999 High body mass index and hypercholesterolemia: risk of hypertensive disorders of pregnancy. Obstet Gynecol 94: 543–550
Ness RB, Markovic N, Bass D, Harger G, Roberts JM 2003 Family history of hypertension, heart disease, and stroke among women who develop hypertension in pregnancy. Obstet Gynecol 102: 1366–1371
Basso O, Christensen K, Olsen J 2001 Higher risk of pre-eclampsia after change of partner. An effect of longer interpregnancy intervals?. Epidemiology 12: 624–629
Shembrey MA, Noble AD 1995 An instructive case of abdominal pregnancy. Aust N Z J Obstet Gynaecol 35: 220–221
Hertig AT 1945 Vascular pathology in the hypertensive albuminuric toxemias of pregnancy. Clinics 4: 602–614
Zeek PM, Assali NS 1950 Vascular changes with in the decidua associated eclamptogenic toxemia of pregnancy. Am J Clin Pathol 20: 1099–1109
Salafia CM, Pezzullo JC, Ghidini A, Lopez-Zeno JA, Whittington SS 1998 Clinical correlations of patterns of placental pathology in preterm pre-eclampsia. Placenta 19: 67–72
Bower S, Schuchter K, Campbell S 1993 Doppler ultrasound screening as part of routine antenatal scanning: prediction of pre-eclampsia and intrauterine growth retardation. Br J Obstet Gynaecol 100: 989–994
North RA, Ferrier C, Long D, Townend K, Kincaid-Smith P 1994 Uterine artery Doppler flow velocity waveforms in the second trimester for the prediction of preeclampsia and fetal growth retardation. Obstet Gynecol 83: 378–386
Aardema MW, Saro MC, Lander M, De Wolf BT, Oosterhof H, Aarnoudse JG 2004 Second trimester Doppler ultrasound screening of the uterine arteries differentiates between subsequent normal and poor outcomes of hypertensive pregnancy: two different pathophysiological entities?. Clin Sci (Lond) 106: 377–382
Cavanagh D, Rao PS, Knuppel RA, Desai U, Balis JU 1985 Pregnancy-induced hypertension: development of a model in the pregnant primate (Papio anubis). Am J Obstet Gynecol 151: 987–999
Combs CA, Katz MA, Kitzmiller JL, Brescia RJ 1993 Experimental preeclampsia produced by chronic constriction of the lower aorta: validation with longitudinal blood pressure measurements in conscious rhesus monkeys. Am J Obstet Gynecol 169: 215–223
Llinas MT, Alexander BT, Seedek M, Abram SR, Crell A, Granger JP 2002 Enhanced thromboxane synthesis during chronic reductions in uterine perfusion pressure in pregnant rats. Am J Hypertens 15: 793–797
Isler CM, Bennett WA, Rinewalt AN, Cockrell KL, Martin JN Jr, Morrison JC, Granger JP 2003 Evaluation of a rat model of preeclampsia for HELLP syndrome characteristics. J Soc Gynecol Investig 10: 151–153
De Wolf F, De Wolf-Peeters C, Brosens I, Robertson WB 1980 The human placental bed: electron microscopic study of trophoblastic invasion of spiral arteries. Am J Obstet Gynecol 137: 58–70
Brosens IA, Robertson WB, Dixon HG 1972 The role of the spiral arteries in the pathogenesis of preeclampsia. Obstet Gynecol Annu 1: 177–191
Kumar D 1962 Chronic placental ischemia in relation to toxemias of pregnancy. A preliminary report. Am J Obstet Gynecol 84: 1323–1329
Robertson WB, Brosens I, Dixon HG 1967 The pathological response of the vessels of the placental bed to hypertensive pregnancy. J Pathol Bacteriol 93: 581–592
Meekins JW, Pijnenborg R, Hanssens M, McFadyen IR, van Asshe A 1994 A study of placental bed spiral arteries and trophoblast invasion in normal and severe pre-eclamptic pregnancies. Br J Obstet Gynaecol 101: 669–674
Pijnenborg R, Anthony J, Davey DA, Rees A, Tiltman A, Vercruysse L, van Assche A 1991 Placental bed spiral arteries in the hypertensive disorders of pregnancy. Br J Obstet Gynaecol 98: 648–655
Zhou Y, Damsky CH, Chiu K, Roberts JM, Fisher SJ 1993 Preeclampsia is associated with abnormal expression of adhesion molecules by invasive cytotrophoblasts. J Clin Invest 91: 950–960
Zhou Y, Fisher SJ, Janatpour M, Genbacev O, Dejana E, Wheelock M, Damsky CH 1997 Human cytotrophoblasts adopt a vascular phenotype as they differentiate. A strategy for successful endovascular invasion?. J Clin Invest 99: 2139–2151
Zhou Y, Damsky CH, Fisher SJ 1997 Preeclampsia is associated with failure of human cytotrophoblasts to mimic a vascular adhesion phenotype. One cause of defective endovascular invasion in this syndrome?. J Clin Invest 99: 2152–2164
Caniggia I, Grisaru-Gravnosky S, Kuliszewsky M, Post M, Lye SJ 1999 Inhibition of TGF-beta 3 restores the invasive capability of extravillous trophoblasts in preeclamptic pregnancies. J Clin Invest 103: 1641–1650
Caniggia I, Mostachfi H, Winter J, Gassmann M, Lye SJ, Kuliszewski M, Post M 2000 Hypoxia-inducible factor-1 mediates the biological effects of oxygen on human trophoblast differentiation through TGFbeta(3). J Clin Invest 105: 577–587
Zhou Y, McMaster M, Woo K, Janatpour M, Perry J, Karpanen T, Alitalo K, Damsky C, Fisher SJ 2002 Vascular endothelial growth factor ligands and receptors that regulate human cytotrophoblast survival are dysregulated in severe preeclampsia and hemolysis, elevated liver enzymes, and low platelets syndrome. Am J Pathol 160: 1405–1423
Zhou Y, Chiu K, Brescia RJ, Combs CA, Katz MA, Kitzmiller JL, Heilbron DC, Fisher SJ 1993 Increased depth of trophoblast invasion after chronic constriction of the lower aorta in rhesus monkeys. Am J Obstet Gynecol 169: 224–229
Ferris TF 1991 Pregnancy, preeclampsia, and the endothelial cell. N Engl J Med 325: 1439–1440
Roberts JM, Taylor RN, Musci TJ, Rodgers GM, Hubel CA, McLaughlin MK 1989 Preeclampsia: an endothelial cell disorder. Am J Obstet Gynecol 161: 1200–1204
Calvin S, Corrigan J, Weinstein L, Jeter M 1988 Factor VIII: von Willebrand factor patterns in the plasma of patients with pre-eclampsia. Am J Perinatol 5: 29–32
Lockwood CJ, Peters JH 1990 Increased plasma levels of ED1+ cellular fibronectin precede the clinical signs of preeclampsia. Am J Obstet Gynecol 162: 358–362
Nova A, Sibai BM, Barton JR, Mercer BM, Mitchell MD 1991 Maternal plasma level of endothelin is increased in preeclampsia. Am J Obstet Gynecol 165: 724–727
Davidge ST 1998 Oxidative stress and altered endothelial cell function in preeclampsia. Semin Reprod Endocrinol 16: 65–73
Hubel CA, McLaughlin MK, Evans RW, Hauth BA, Sims CJ, Roberts JM 1996 Fasting serum triglycerides, free fatty acids, and malondialdehyde are increased in preeclampsia, are positively correlated, and decrease within 48 hours post partum. Am J Obstet Gynecol 174: 975–982
Kolben M, Lopens A, Blaser J, Huber A, Frank M, Wilhelm O, Wilhelm S, Schneider KT, Ulm K, Tschesche H 1995 Measuring the concentration of various plasma and placenta extract proteolytic and vascular factors in pregnant patients with HELLP syndrome, pre-eclampsia and highly pathologic Doppler flow values. Gynakolo Geburtshilfliche Rundschau 35( suppl 1): 126–131
Chappell LC, Seed PT, Briley A, Kelly FJ, Hunt BJ, Charnock-Jones DS, Mallet AI, Poston L 2002 A longitudinal study of biochemical variables in women at risk of preeclampsia. Am J Obstet Gynecol 187: 127–136
Mills JL, DerSimonian R, Raymond E, Morrow JD, Roberts LJ II, Clemens JD, Hauth JC, Catalano P, Sibai B, Curet LB, Levine RJ 1999 Prostacyclin and thromboxane changes predating clinical onset of preeclampsia: a multicenter prospective study. JAMA 282: 356–362
Gant NF, Daley GL, Chand S, Whalley PJ, McDonald PC 1973 A study of angiotensin II pressor response throughout primigravid pregnancy. J Clin Invest 52: 2682–2689
Ashworth JR, Warren AY, Baker PN, Johnson IR 1997 Loss of endothelium-dependent relaxation in myometrial resistance arteries in pre-eclampsia. Br J Obstet Gynaecol 104: 1152–1158
Chambers JC, Fusi L, Malik IS, Haskard DO, De Swiet M, Kooner JS 2001 Association of maternal endothelial dysfunction with preeclampsia. JAMA 285: 1607–1612
Spargo BH, McCartney C, Winemiller R 1959 Glomerular capillary endotheliosis in toxemia of pregnancy. Arch Pathol 13: 593–599
Fisher KA, Luger A, Spargo BH, Lindheimer MD 1981 Hypertension in pregnancy: clinical-pathological correlations and remote prognosis. Medicine (Baltimore) 60: 267–276
Morris RH, Vassaldi PI, Beller FK, McCluskey RT 1964 Immunofluorescent studies of renal biopsies in the diagnosis of toxemia of pregnancy. Obstet Gynecol 24: 32–46
Strevens H, Wide-Swensson D, Hansen A, Horn T, Ingemarsson I, Larsen S, Willner J, Olsen S 2003 Glomerular endotheliosis in normal pregnancy and pre-eclampsia. BJOG 110: 831–836
Irgens HU, Reisaeter L, Irgens LM, Lie RT 2001 Long term mortality of mothers and fathers after pre-eclampsia: population based cohort study. BMJ 323: 1213–1217
Haukkamaa L, Salminen M, Laivuori H, Leinonen H, Hiilesmaa V, Kaaja R 2004 Risk for subsequent coronary artery disease after preeclampsia. Am J Cardiol 93: 805–808
Smith GC, Pell JP, Walsh D 2001 Pregnancy complications and maternal risk of ischaemic heart disease: a retrospective cohort study of 129,290 births. Lancet 357: 2002–2006
Epstein FH 1964 Late vascular effects of toxemia of pregnancy. N Eng J Med 271: 391–395
Agatisa PK, Ness RB, Roberts JM, Constantino JP, Kuller LH, McLaughlin MK 2004 Impairment of endothelial function in women with a history of preeclampsia: an indicator of cardiovascular risk. Am J Physiol Heart Circ Physiol 286: 1389–1393
Polednak AP, Janerich DT 1983 Characteristics of first pregnancy in relation to early breast cancer. A case-control study. J Reprod Med 28: 314–318
Vatten LJ, Romundstad PR, Trichopoulos D, Skjaerven R 2002 Pre-eclampsia in pregnancy and subsequent risk for breast cancer. Br J Cancer 87: 971–973
Baker PN, Davidge ST, Roberts JM 1995 Plasma from women with preeclampsia increases endothelial cell nitric oxide production. Hypertension 26: 244–248
Roberts JM, Edep ME, Goldfien A, Taylor RN 1992 Sera from preeclamptic women specifically activate human umbilical vein endothelial cells in vitro: morphological and biochemical evidence. Am J Reprod Immunol 27: 101–108
de Groot CJ, Murai JT, Vigne JL, Taylor RN 1998 Eicosanoid secretion by human endothelial cells exposed to normal pregnancy and preeclampsia plasma in vitro. Prostaglandins Leukot Essent Fatty Acids 58: 91–97
Hayman R, Warren A, Brockelsby J, Johnson I, Baker P 2000 Plasma from women with pre-eclampsia induces an in vitro alteration in the endothelium-dependent behaviour of myometrial resistance arteries. BJOG 107: 108–115
Roberts JM, Lain KY 2002 Recent Insights into the pathogenesis of pre-eclampsia. Placenta 23: 359–372
Page NM, Woods RJ, Gardiner SM, Lomthaisong K, Gladwell RT, Butlin DJ, Manyonda IT, Lowry PJ 2000 Excessive placental secretion of neurokinin B during the third trimester causes pre-eclampsia. Nature 405: 797–800
Schlembach D, Scalera F, Fischer T, Marx SG, Beinder E, Garfield RE 2003 Neurokinin B peptide serum levels are higher in normotensive pregnant women than in preeclamptic pregnant women. Am J Obstet Gynecol 189: 1418–1422
Wareing M, Bhatti H, O'Hara M, Kenny L, Warren AY, Taggart MJ, Baker PN 2003 Vasoactive effects of neurokinin B on human blood vessels. Am J Obstet Gynecol 188: 196–202
Smarason AK, Sargent IL, Starkey PM, Redman CW 1993 The effect of placental syncytiotrophoblast microvillous membranes from normal and pre-eclamptic women on the growth of endothelial cells in vitro. BJOG 100: 943–949
AbdAlla S, Lother H, el Massiery A, Quitterer U 2001 Increased AT(1) receptor heterodimers in preeclampsia mediate enhanced angiotensin II responsiveness. Nat Med 7: 1003–1009
Ward K, Hata A, Jeunemaitre X, Helin C, Nelson L, Namikawa C, Farrington PF, Ogasawara M, Suzumori K, Tomoda S 1993 A molecular variant of angiotensinogen associated with preeclampsia. Nat Genet 4: 59–61
Wallukat G, Homuth V, Fischer T, Lindschau C, Horstkamp B, Jupner A, Baur E, Nissen E, Vetter K, Neichel D, Dudenhausen JW, Haller H, Luft FC 1999 Patients with preeclampsia develop agonistic autoantibodies against the angiotensin AT1 receptor. J Clin Invest 103: 945–952
Xia Y, Wen H, Bobst S, Day MC, Kellems RE 2003 Maternal autoantibodies from preeclamptic patients activate angiotensin receptors on human trophoblast cells. J Soc Gynecol Investig 10: 82–93
Dechend R, Viedt C, Muller DN, Ugele B, Brandes RP, Wallukat G, Park JK, Janke J, Barta P, Theuer J, Fiebeler A, Homuth V, Dietz R, Haller H, Kreuzer J, Luft FC 2003 AT1 receptor agonistic antibodies from preeclamptic patients stimulate NADPH oxidase. Circulation 107: 1632–1639
Maynard SE, Min JY, Merchan J, Lim KH, Li J, Mondal S, Libermann TA, Morgan JP, Sellke FW, Stillman IE, Epstein FH, Sukhatme VP, Karumanchi SA 2003 Excess placental soluble fms-like tyrosine kinase 1 (sFlt1) may contribute to endothelial dysfunction, hypertension, and proteinuria in preeclampsia. J Clin Invest 111: 649–658
Koga K, Osuga Y, Yoshino O, Hirota Y, Ruimeng X, Hirata T, Takeda S, Yano T, Tsutsumi O, Taketani Y 2003 Elevated serum soluble vascular endothelial growth factor receptor 1 (sVEGFR-1) levels in women with preeclampsia. J Clin Endocrinol Metab 88: 2348–2351
Tsatsaris V, Goffin F, Munaut C, Brichant JF, Pignon MR, Noel A, Schaaps JP, Cabrol D, Frankenne F, Foidart JM 2003 Overexpression of the soluble vascular endothelial growth factor receptor in preeclamptic patients: pathophysiological consequences. J Clin Endocrinol Metab 88: 5555–5563
Chaiworapongsa T, Romero R, Espinoza J, Bujold E, Mee Kim Y, Goncalves LF, Gomez R, Edwin S 2004 Evidence supporting a role for blockade of the vascular endothelial growth factor system in the pathophysiology of preeclampsia. Young Investigator Award. Am J Obstet Gynecol 190: 1541–1547
Taylor RN, Grimwood J, Taylor RS, McMaster MT, Fisher SJ, North RA 2003 Longitudinal serum concentrations of placental growth factor: evidence for abnormal placental angiogenesis in pathologic pregnancies. Am J Obstet Gynecol 188: 177–182
Luttun A, Tjwa M, Moons L, Wu Y, Angelillo-Scherrer A, Liao F, Nagy JA, Hooper A, Priller J, De Klerck B, Compernolle V, Daci E, Bohlen P, Dewerchin M, Herbert JM, Fava R, Matthys P, Carmeliet G, Collen D, Dvorak HF, Hicklin DJ, Carmeliet P 2002 Revascularization of ischemic tissues by PlGF treatment, and inhibition of tumor angiogenesis, arthritis and atherosclerosis by anti-Flt1. Nat Med 8: 831–840
Carmeliet P, Moons L, Luttun A, Vincenti V, Compernolle V, De Mol M, Wu Y, Bono F, Devy L, Beck H, Scholz D, Acker T, DiPalma T, Dewerchin M, Noel A, Stalmans I, Barra A, Blacher S, Vandendriessche T, Ponten A, Eriksson U, Plate KH, Foidart JM, Schaper W, Charnock-Jones DS, Hicklin DJ, Herbert JM, Collen D, Persico MG 2001 Synergism between vascular endothelial growth factor and placental growth factor contributes to angiogenesis and plasma extravasation in pathological conditions. Nat Med 7: 575–583
Dvorak HF 2002 Vascular permeability factor/vascular endothelial growth factor: a critical cytokine in tumor angiogenesis and a potential target for diagnosis and therapy. J Clin Oncol 20: 4368–4380
Kendall RL, Thomas KA 1993 Inhibition of vascular endothelial cell growth factor activity by an endogenously encoded soluble receptor. Proc Natl Acad Sci U S A 90: 10705–10709
Nagamatsu T, Fujii T, Kusumi M, Zou L, Yamashita T, Osuga Y, Momoeda M, Kozuma S, Taketani Y 2004 Cytotrophoblasts up-regulate soluble fms-like tyrosine kinase-1 expression under reduced oxygen: an implication for the placental vascular development and the pathophysiology of preeclampsia. Endocrinology 145: 4838–4845
Levine RJ, Maynard SE, Qian C, Lim KH, England LJ, Yu KF, Schisterman EF, Thadhani R, Sachs BP, Epstein FH, Sibai BM, Sukhatme VP, Karumanchi SA 2004 Circulating angiogenic factors and the risk of preeclampsia. N Engl J Med 350: 672–683
Thadhani R, Mutter WP, Wolf M, Levine RJ, Taylor RN, Sukhatme VP, Ecker J, Karumanchi SA 2004 First trimester placental growth factor and soluble fms-like tyrosine kinase 1 and risk for preeclampsia. J Clin Endocrinol Metab 89: 770–775
Simon M, Grone HJ, Johren O, Kullmer J, Plate KH, Risau W, Fuchs E 1995 Expression of vascular endothelial growth factor and its receptors in human renal ontogenesis and in adult kidney. Am J Physiol 268: F240–F250
Simon M, Rockl W, Hornig C, Grone EF, Theis H, Weich HA, Fuchs E, Yayon A, Grone HJ 1998 Receptors of vascular endothelial growth factor/vascular permeability factor (VEGF/VPF) in fetal and adult human kidney: localization and [125I]VEGF binding sites. J Am Soc Nephrol 9: 1032–1044
Masuda Y, Shimizu A, Mori T, Ishiwata T, Kitamura H, Ohashi R, Ishizaki M, Asano G, Sugisaki Y, Yamanaka N 2001 Vascular endothelial growth factor enhances glomerular capillary repair and accelerates resolution of experimentally induced glomerulonephritis. Am J Pathol 159: 599–608
Ostendorf T, Kunter U, Eitner F, Loos A, Regele H, Kerjaschki D, Henninger DD, Janjic N, Floege J 1999 VEGF(165) mediates glomerular endothelial repair. J Clin Invest 104: 913–923
Kabbinavar F, Hurwitz HI, Fehrenbacher L, Meropol NJ, Novotny WF, Lieberman G, Griffing S, Bergsland E 2003 Phase II, randomized trial comparing bevacizumab plus fluorouracil (FU)/leucovorin (LV) with FU/LV alone in patients with metastatic colorectal cancer. J Clin Oncol 21: 60–65
Yang JC, Haworth L, Sherry RM, Hwu P, Schwartzentruber DJ, Topalian SL, Steinberg SM, Chen HX, Rosenberg SA 2003 A randomized trial of bevacizumab, an anti-vascular endothelial growth factor antibody, for metastatic renal cancer. N Engl J Med 349: 427–434
Esser S, Wolburg K, Wolburg H, Breier G, Kurzchalia T, Risau W 1998 Vascular endothelial growth factor induces endothelial fenestrations in vitro. J Cell Biol 140: 947–959
Lain KY, Wilson JW, Crombleholme WR, Ness RB, Roberts JM 2003 Smoking during pregnancy is associated with alterations in markers of endothelial function. Am J Obstet Gynecol 189: 1196–1201
Heeschen C, Jang JJ, Weis M, Pathak A, Kaji S, Hu RS, Tsao PS, Johnson FL, Cooke JP 2001 Nicotine stimulates angiogenesis and promotes tumor growth and atherosclerosis. Nat Med 7: 833–839
Eremina V, Sood M, Haigh J, Nagy A, Lajoie G, Ferrara N, Gerber HP, Kikkawa Y, Miner JH, Quaggin SE 2003 Glomerular-specific alterations of VEGF-A expression lead to distinct congenital and acquired renal diseases. J Clin Invest 111: 707–716
Tuohy JF, James DK 1992 Pre-eclampsia and trisomy 13. Br J Obstet Gynaecol 99: 891–894
Moses EK, Lade JA, Guo G, Wilton AN, Grehan M, Freed K, Borg A, Terwilliger JD, North R, Cooper DW, Brennecke SP 2000 A genome scan in families from Australia and New Zealand confirms the presence of a maternal susceptibility locus for pre-eclampsia, on chromosome 2. Am J Hum Genet 67: 1581–1585
Arngrimsson R, Sigurard ttir S, Frigge ML, Bjarnad ttir RI, Jonsson T, Stefansson H, Baldursdottir A, Einarsdottir AS, Palsson B, Snorradottir S, Lachmeijer AM, Nicolae D, Kong A, Bragason BT, Gulcher JR, Geirsson RT, Stefansson K 1999 A genome-wide scan reveals a maternal susceptibility locus for pre-eclampsia on chromosome 2p13. Hum Mol Genet 8: 1799–1805
Wolf M, Sandler L, Munoz K, Hsu K, Ecker JL, Thadhani R 2002 First trimester insulin resistance and subsequent preeclampsia: a prospective study. J Clin Endocrinol Metab 87: 1563–1568
Skjaerven R, Wilcox AJ, Lie RT 2002 The interval between pregnancies and the risk of preeclampsia. N Engl J Med 346: 33–38
Robillard PY, Hulsey TC, Perianin J, Janky E, Miri EH, Papiernik E 1994 Association of pregnancy-induced hypertension with duration of sexual cohabitation before conception. Lancet 344: 973–975
Thadhani R, Ecker JL, Mutter WP, Wolf M, Smirnakis KV, Sukhatme VP, Levine RJ, Karumanchi SA 2004 Insulin resistance and alterations in angiogenesis: additive insults that may lead to preeclampsia. Hypertension 43: 988–992
Ahmad S, Ahmed A 2004 Elevated placental soluble vascular endothelial growth factor receptor-1 inhibits angiogenesis in preeclampsia. Circ Res
Karumanchi SA, Lim KH, Sukhatme VP, August P 2004 Pathogenesis of preeclampsia. In: Rose BD (ed) UpToDate, Wellesley, MA, UpToDate Inc.
Author information
Authors and Affiliations
Corresponding author
Additional information
S.A.K was supported by National Institutes of Health (NIH) grants DK64255 and DK065997 and the Carl W. Gottschalk Research Scholar Award by the American Society of Nephrology. R.T. was supported by NIH grant HD39223 and the McGuirk Family Research Foundation.
Rights and permissions
About this article
Cite this article
Maynard, S., Venkatesha, S., Thadhani, R. et al. Soluble Fms-like Tyrosine Kinase 1 and Endothelial Dysfunction in the Pathogenesis of Preeclampsia. Pediatr Res 57, 1–7 (2005). https://doi.org/10.1203/01.PDR.0000159567.85157.B7
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1203/01.PDR.0000159567.85157.B7
This article is cited by
-
Sex and fetal genome influence gene expression in pig endometrium at the end of gestation
BMC Genomics (2024)
-
Expression profiles and functions of ferroptosis-related genes in the placental tissue samples of early- and late-onset preeclampsia patients
BMC Pregnancy and Childbirth (2022)
-
Biochemical markers in the pathogenesis of preeclampsia: novel link between placental growth factor and interleukin-6
Molecular and Cellular Biochemistry (2022)
-
The Role of Biomarkers and Imaging to Predict Preeclampsia and Subsequent Cardiovascular Dysfunction
Current Treatment Options in Cardiovascular Medicine (2021)
-
Bioenergetic effects of hydrogen sulfide suppress soluble Flt-1 and soluble endoglin in cystathionine gamma-lyase compromised endothelial cells
Scientific Reports (2020)