Abstract
An increasing series of pediatric endocrinopathies and metabolic anomalies has been recognized as related to reduced prenatal growth. We have tested whether the association of precocious pubarche (PP), dyslipidemia, and low serum IGF binding protein-1 in girls is also related to reduced prenatal growth. Fasting serum lipids, lipoproteins, and IGFBP-1 concentrations were measured in 187 girls (83 without PP and 104 with PP; mean age, 11.8 y; range, 5-18 y) with known birthweight and gestational age, the latter being transformed into birthweight SD scores. Birthweight SD scores of girls with PP were lower than those of girls without PP. Within the group of PP girls, those with dyslipidemia and low IGFBP-1 had lower (p < 0.0001) birthweight SD scores (-2.02 ± 0.23; mean ± SEM) than those with normal lipids, lipoproteins, and IGFBP-1 (-0.37 ± 0.15), whereas girls with an intermediate number of abnormalities had intermediate birthweight SD scores (-0.80 ± 0.18). In conclusion, dyslipidemia and low serum IGFBP-1 in girls with PP were found to be related to reduced prenatal growth, an observation pointing to the prenatal origin of these metabolic abnormalities.
Similar content being viewed by others
Main
An increasing body of evidence suggests that the spectrum of variation for numerous endocrine and metabolic variables in adult life is related to fetal growth(1). Pronounced adrenarche and PP in girls, defined as the appearance of pubic hair before the age of 8 y(2), are among the endocrine variants related to reduced fetal growth(3,4). In turn, PP in girls was recently found to be associated with dyslipidemia and low serum IGFBP-1 concentrations(5,6). Accordingly, we have now studied whether the presence of dyslipidemia and low serum IGFBP-1 in PP girls is also linked to reduced prenatal growth and may therefore have a prenatal origin.
METHODS
Study population. The study population consisted of 187 girls, 83 of whom had no history of PP (age, 12.1 ± 2.7 y; range, 6-17 y) and 104 of whom had PP (age, 11.5 ± 3.4 y; range, 5-18 y). Clinical characteristics of prepubertal, pubertal, and postmenarchal subgroups(7) are displayed in Table 1.
Girls with PP were only included in the study when PP was secondary to pronounced adrenarche, i.e. after exclusion of nonclassic adrenal hyperplasia(8–11). None of the girls presented evidence for thyroid dysfunction, Cushing syndrome, hyperprolactinemia, diabetes mellitus, or hypertension, nor received medication affecting lipid metabolism. All subjects had normal glucose tolerance(12); insulin responses to an oral glucose load, the lipid profile, and the IGFBP-1 levels in 185 of these girls have been reported within a different context(5,6).
The Institutional Review Board of the Barcelona Hospital approved the study protocol. Informed consent was obtained from the parents and assent from the girls.
Experimental methods. Birth weight and gestational age data were obtained from hospital records and transformed into SD scores, as previously described(4).
Serum lipids and lipoproteins were measured from blood samples after an overnight fast. Samples were centrifuged, and serum was separated and frozen at -20°C until assay. LDL-cholesterol was calculated by the Friedewald formula. Lipoprotein fractionation was performed using ultracentrifugation with a Centrikon T-1035, rotor type TFT 45.6 ultracentrifuge (Kontron International, Zurich, Switzerland). Total HDL was isolated by selective precipitation with phosphotungstic acid-magnesium chloride and measured by the CHOD-PAP method(13). Serum triglycerides were measured using CHOD-PAP- and GPO-PAP-based methods in a Hitachi 917 autoanalyzer (Boehringer Mannheim, Mannheim, Germany). The mean intra- and interassay coefficients of variation were 2.2% and 2.0%, respectively. Serum triglycerides were considered high when >95 mg/dL in girls aged 5-10 y and >110 mg/dL in girls aged 11-18 y(14). LDL/HDL ratios >1.8 were considered high(6).
Serum IGFBP-1 was measured by a quantitative immunometric assay (Medix-Biochemma, Oulu, Finland), as described previously(5). Values were considered low if they were <2.8 µg/L before puberty and <1.5 µg/L in pubertal or postmenarchal girls(5).
Results are expressed as mean ± SEM. Mann-Whitney U test was used for statistical comparisons, with significance at p < 0.05.
RESULTS
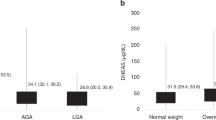
Birthweight SD scores of girls with PP (-0.76 ± 0.12) were lower (p < 0.0001) than those of girls without PP (0.38 ± 0.08); these scores correspond to respective birthweights of 3024 ± 60 g (after 39.08 ± 0.17 wk) and 3501 ± 39 g (after 39.06 ± 0.12 wk).
Among girls who had PP (Fig. 1), those with dyslipidemia and low IGFBP-1 had strikingly lower (p < 0.0001) birthweight SD scores (-2.02 ± 0.23 corresponding to 2485 ± 129 g) than those with normal lipids, lipoproteins, and IGFBP-1 (-0.37 ± 0.15 for 3284 ± 57 g), whereas girls with an intermediate number of abnormalities had intermediate birthweight SD scores (-0.80 ± 0.18 for 2914 ± 105 g).
DISCUSSION
The presence of dyslipidemia or low serum IGFBP-1 in girls who had PP was found to depend on weight gain before birth. Previous associations are herewith extended in two directions. First, the link between dyslipidemia and reduced fetal growth, initially recognized in 64-year-old men(15), appears to be already detectable in girls with PP. Second, the spectrum of pediatric endocrinopathies and metabolic anomalies that are somehow related to reduce fetal growth is further broadened to include dyslipidemia and low circulating IGFBP-1 levels, as well as insulin resistance and hyperinsulinism(4,16), growth failure(17,18), exaggerated adrenarche with PP(3,4), ovarian hyperandrogenism and anovulation(4,19), and male subfertility(20). However, the distinct or common mechanisms underlying each of these associations remain poorly understood.
Circulating IGFBP-1 levels correlate inversely with plasma insulin levels, and are considered to be a useful marker for hyperinsulinemia or insulin resistance in subjects with intact endogenous insulin secretion(21–23). Hyperinsulinemia and suppressed IGFBP-1 levels may enhance the availability and insulin-like actions of free IGF-I(24). Increased plasma IGF-I concentrations in low birthweight children showing postnatal catch-up growth has been suggested, together with hyperinsulinemia, as one of the mechanisms linking reduced fetal growth to cardiovascular disease in adult life(25,26).
Increased fasting triglyceride, high LDL, and decreased HDL concentrations are independent risk factors for the early development of cardiovascular disease(27,28). Hyperinsulinemia is the presumed link between dyslipidemia and the other components of syndrome X(29–31). The present results support previous studies in children indicating that this cluster of metabolic abnormalities can already be detected at a young age(6,32,33) and may indeed have a prenatal origin.
In conclusion, the presence of dyslipidemia and low serum IGFBP-1 in girls who had PP was found to be related to prenatal weight gain. The pathophysiologic mechanisms underlying these relationships remain to be elucidated.
Abbreviations
- PP:
-
precocious pubarche
- IGFBP-1:
-
IGF binding protein-1
References
Barker DJ 1998 Mothers, Babies and Diseases in Later Life. Churchill Livingstone, London
Ibáñez L, Potau N, Carrascosa A 1997 Androgens in adrenarche and pubarche. In: Azziz R, Nestler JE, Dewailly D (eds) Androgen Excess in Women. Lippincott-Raven Publishers, Philadelphia, 73–84.
Francois I, de Zegher F 1997 Adrenarche and fetal growth. Pediatr Res 41: 440–442.
Ibáñez L, Potau N, Francois I, de Zegher F 1998 Precocious pubarche, hyperinsulinism and ovarian hyperandrogenism in girls: relation to reduced fetal growth. J Clin Endocrinol Metab 83: 3558–3662.
Ibáñez L, Potau N, Zampolli M, Rique S, Saenger P, Carrascosa A 1997 Hyperinsulinemia and decreased insulin-like growth factor binding protein-1 are common features in prepubertal and pubertal girls with a history of premature pubarche. J Clin Endocrinol Metab 82: 2283–2288.
Ibáñez L, Potau N, Chacon P, Pascual C, Carrascosa A 1998 Hyperinsulinemia, dyslipaemia and cardiovascular risk in girls with a history of premature pubarche. Diabetologia 41: 1057–1063.
Marshall WA, Tanner JM 1969 Variations in the pattern of pubertal changes in girls. Arch Dis Child 44: 291–303.
Rosenfield RL 1971 Plasma 17-ketosteroids and 17β-ketosteroids in girls with premature development of sexual hair. J Pediatr 79: 260–266.
Rosenfield RL 1994 Normal and almost normal precocious variations in pubertal development: premature pubarche and premature thelarche revisited. Horm Res 41( suppl 2): 7–13.
New MI, Lorenzen F, Lerner AJ, Kohn B, Oberfield SE, Pollack MS, Dupont B, Stoner E, Levy DJ, Pang S, Levine LS 1983 Genotyping steroid 21-hydroxylase deficiency: hormonal reference data. J Clin Endocrinol Metab 56: 320–325.
Sakkal-Alkaddour H, Zhang L, Yang X, Chang YT, Kappy M, Slover RS, Jorgensen V, Pang S 1996 Studies of 3β-hydroxysteroid dehydrogenase genes in infants and children manifesting premature pubarche and increased adrenocorticotropin-stimulated Δ5-steroid levels. J Clin Endocrinol Metab 81: 3961–3965.
The Expert Committee on the Diagnosis and Classification of Diabetes Mellitus 1997 Report of the Expert Committee on the Diagnosis and Classification of Diabetes Mellitus. Diabetes Care 20: 1183–1197.
Assman G, Schiewr H, Schnitz G, Haegle D 1983 Quantification of high density lipoprotein cholesterol by precipitation with phosphotungstic acid-Mg-Cl2 . Clin Chem 29: 2026–2030.
Christensen B, Glueck C, Kwiterovich P, Degroot I, Chase G, Heiss G, Mowery R, Tamir I, Rifkind B 1980 Plasma cholesterol and triglyceride distributions in 13665 children and adolescents: the prevalence study of the Lipid Research Clinics Program. Pediatr Res 14: 194–202.
Barker DJP, Hales CN, Fall CHD, Osmond C, Phipps K, Clark PMS 1993 Type 2 (non-insulin-dependent) diabetes mellitus, hypertension and hyperlipidaemia (syndrome X): relation to reduced fetal growth. Diabetologia 36: 62–67.
Hofman PL, Cutfield WS, Robinson EM, Bergman RN, Menon RK, Sperling MA, Gluckman PD 1997 Insulin resistance in short children with intrauterine growth retardation. J Clin Endocrinol Metab 82: 402–406.
de Zegher F, Francois I, van Helvoirt M, Van de Berghe G 1997 Small as a fetus and short as a child: from endogenous to exogenous growth hormone. J Clin Endocrinol Metab 82: 2021–2026.
de Zegher F, Francois I, Ibáñez L 1999 Pediatric endocrinopathies related to reduced fetal growth. Growth Genet Horm 15: 1–5.
Ibáñez L, Potau N, de Zegher F 1999 Anovulation after precocious pubarche: early markers and time course in adolescence. J Clin Endocrinol Metab ( in press)
Francois I, de Zegher F, Spiessens C, D'Hooghe T, Vanderschueren D 1997 Low birth weight and subsequent male subfertility. Pediatr Res 42: 899–901.
Lee PDK, Jensen MD, Divertie GD, Heiling VJ, Katz HH, Conover CA 1993 Insulin-like growth factor binding protein-1 response to insulin during suppression of endogenous insulin secretion. Metabolism 42: 409–414.
Ibáñez L, Potau N, Carrascosa A 1998 Insulin resistance, premature adrenarche, and a risk of the polycystic ovary syndrome (POS). Trends Endocrinol Metab 9: 72–77.
Yki-Järvinen H, Makimattila S, Utrianen T, Rutanen E 1995 Portal insulin concentrations rather than insulin sensitivity regulate serum sex hormone binding-globulin and insulin-like growth factor binding protein 1 in vivo . J Clin Endocrinol Metab 80: 3227–3232.
Travers S, Labarta JI, Gargosky SE, Rosenfeld RG, Jeffers BW, Eckel RH 1998 Insulin-like growth factor binding protein-I levels are strongly associated with insulin sensitivity and obesity in early prepubertal children. J Clin Endocrinol Metab 83: 1935–1939.
Fall CHD, Pandit AN, Law CM, Yajnik CS, Clark PM, Breier B, Osmond C, Shiell AW, Gluckman PD, Barker DJ 1995 Size at birth and plasma insulin-like growth factor-1 concentrations. Arch Dis Child 73: 287–293.
Langford K, Blum W, Nicolaides K, Jones J, McGregor A, Miell J 1994 The pathophysiology of the insulin-like growth factor axis in fetal growth failure: a basis for programming by undernutrition?. Eur J Clin Invest 24: 851–856.
Newman WP, Freedman DS, Voors AW, Gard PD, Srinivasan SR, Cresanta JL, Williamson GD, Webber LS, Berenson GS 1986 Relation of serum lipoprotein levels and systolic blood pressure to early atherosclerosis. N Engl J Med 314: 138–143.
Patsch JR, Miesenböck G, Hopferwieser T, Muhlberger V, Knapp E, Dunn JK, Gotto AM Jr, Patsch W 1992 Relation of triglyceride metabolism and coronary artery disease: studies in the postprandial state. Arterioscler Thromb 12: 1336–1345.
Ferrannini E, Haffner SM, Mitchell BD, Stern MP 1991 Hyperinsulinemia: the key feature of a cardiovascular and metabolic syndrome. Diabetologia 34: 416–422.
Orchard TJ, Becker DJ, Bates M, Kuller LH, Drash AL 1983 Plasma insulin and lipoprotein concentrations: an atherogenic association?. Am J Epidemiol 118: 326–337.
Després JP 1993 The insulin resistance-dyslipidemia syndrome: the most prevalent cause of coronary artery disease?. Can Med Assoc J 148: 1339–1340.
Rönnemaa T, Knip M, Lautala P, Viikari J, Uhari M, Leino A, Kaprio EA, Salo MK, Dahl M, Nuutinen EM 1991 Serum insulin and other cardiovascular risk indicators in children, adolescents and young adults. Ann Med 23: 67–72.
Arslanian S, Suprasongsin C 1996 Insulin sensitivity, lipids and body composition in childhood: is "syndrome X" present?. J Clin Endocrinol Metab 81: 1058–1062.
Acknowledgements
The authors thank Karin Vanweser, RN, for her editorial assistance.
Author information
Authors and Affiliations
Additional information
Supported by a scholarship from the European Society for Paediatric Endocrinology. F.d.Z. is a Clinical Research Investigator of the Fund for Scientific Research, Flanders, Belgium.
Rights and permissions
About this article
Cite this article
Ibáñez, L., Potau, N. & de Zegher, F. Precocious Pubarche, Dyslipidemia, and Low IGF Binding Protein-1 in Girls: Relation to Reduced Prenatal Growth. Pediatr Res 46, 320–322 (1999). https://doi.org/10.1203/00006450-199909000-00012
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1203/00006450-199909000-00012
This article is cited by
-
Clinical spectrum of premature pubarche: Links to metabolic syndrome and ovarian hyperandrogenism
Reviews in Endocrine and Metabolic Disorders (2009)