Abstract
Tyrosine is considered to be an indispensable dietary amino acid in the neonate, yet achieving adequate parenteral tyrosine intake is difficult due to its poor solubility. Increasing the supply of phenylalanine is the most common means of compensating for low tyrosine levels. Unfortunately, plasma phenylalanine concentrations are sometimes elevated in infants receiving high phenylalanine intake. This led us to study the phenylalanine and tyrosine metabolism in 16 neonates randomized to receive total parenteral nutrition with either a high or a moderate phenylalanine-containing amino acid solution. A primed, 24-h continuous stable isotope infusion of L-[1-13C]phenylalanine and L-[3,3-2H2]tyrosine was given to enable the measurement of phenylalanine and tyrosine kinetics. Results demonstrated that 1) phenylalanine hydroxylation was significantly greater in infants receiving high phenylalanine, 2) phenylalanine oxidation and percent dose oxidized was also significantly greater in infants receiving high phenylalanine, 3) apparent phenylalanine retention was greater in neonates receiving high phenylalanine, and 4) alternate catabolites of phenylalanine and tyrosine metabolism were significantly greater in infants receiving high phenylalanine compared with moderate phenylalanine. We conclude that neonates respond to increased parenteral phenylalanine intake by increasing their hydroxylation and oxidation rates. The greater oxidation of phenylalanine in infants receiving high phenylalanine in conjunction with the urinary excretion of alternate catabolites of phenylalanine and tyrosine suggests that the high phenylalanine intake may be in excess of needs. However, the lower apparent phenylalanine retention observed in infants receiving moderate phenylalanine suggests that the total aromatic amino acid level of moderate phenylalanine may be deficient for neonatal needs.
Similar content being viewed by others
Main
Tyrosine is considered to be an indispensable dietary amino acid during the neonatal period (1–3). Providing adequate tyrosine in the parenterally fed infant is particularly difficult due to its poor solubility, restricting maximum tyrosine intake to less than 1% of total amino acids. This level of intake is significantly lower than the estimated requirement of Snyderman (1) in neonates (2.0%), of House et al. (4) (2.7%) in the piglet, or the content of human milk (5.9%) (5). Increasing parenteral phenylalanine intake is the most common means of providing a precursor source of tyrosine. This approach relies on the neonate's ability to hydroxylate phenylalanine to tyrosine. Unfortunately, several investigations have observed that this approach may result in poor metabolic tolerance as indicated by elevated plasma phenylalanine (6–15) and urinary excretion of alternate catabolites of phenylalanine (phenylpyruvate, phenylacetate, phenyllactate, 2-hydroxyphenylacetate) and tyrosine (4-hydroxyphenylpyruvate, 4-hydroxyphenylacetate, 4-hydroxyphenyllactate) (6,14,15).
Early studies examining phenylalanine hydroxylase enzyme (EC 1.14.16.1) activity from fetal liver tissue demonstrated that the phenylalanine hydroxylase activity of the fetus is only 57% that of the adult (16). In contrast, recent studies investigating in vivo neonatal phenylalanine hydroxylation have found significant hydroxylation capacity in the neonate (2,17–20). These studies examined the rate of phenylalanine hydroxylation in response to parenteral nutrition (17,18,20,21), route of nutrient administration (19), maturity (2,21), and septic state (2). Although Shortland et al. (17) have demonstrated that phenylalanine hydroxylation increases in response to a supply of amino acid substrate, other studies have not shown such an increase (20,21). These latter studies (20,21) used amino acid solutions that had lower phenylalanine levels; the available phenylalanine may be an important factor in determining the phenylalanine hydroxylation response to parenteral nutrition.
Therefore, it is still unclear how the neonate responds to the differing phenylalanine intakes that are commonly provided by commercially available amino acids solutions. Of the solutions used in neonatal populations, "general purpose" amino acid solutions contain large amounts of phenylalanine to compensate for their low tyrosine levels. "Pediatric" amino acid solutions, in contrast, have been developed with particular concern for the immaturities of the neonate, and therefore contain only modest levels of phenylalanine. Infants receiving pediatric solutions generally demonstrate good metabolic tolerance of aromatic amino acid intake; however, evidence exists that these solutions may be inadequate in their content of total aromatic amino acids (15).
To better understand the utilization of phenylalanine as a parenteral precursor of tyrosine, this study examined the parenterally fed neonate's aromatic amino acid metabolism, using infusions of stable isotope labeled phenylalanine and tyrosine, while receiving amino acid solutions that differ in their content of phenylalanine. We hypothesized that the neonate responds to increased phenylalanine intake by increasing their rate of phenylalanine hydroxylation. Furthermore, we tested for urinary excretion of alternate catabolites of phenylalanine and tyrosine to assess tolerance of the aromatic amino acid intake.
METHODS
Patients. Sixteen parenterally fed infants (Table 1) who had been referred to the Neonatal Intensive Care Unit of The Hospital for Sick Children were enrolled into a randomized trial comparing two commercially available parenteral amino acid solutions currently used for neonates. All infants were clinically stable during the study period and were maintained in standard servo-controlled incubators or infant cribs. Gastroschisis, intestinal atresia, necrotizing enterocolitis, and respiratory distress syndrome were the most common diagnoses in each group (Table 1). The study protocol was reviewed and approved by the Human Subject Review Committee of The Hospital for Sick Children, and written, informed consent was obtained from one or both parents.
Infants were randomized to receive one of two parenteral feeding regimens differing only in amino acid profile. The amino acid solutions were: Vamin (Vamin N, Pharmacia and Upjohn, Stockholm, Sweden), the standard amino acid solution used at The Hospital for Sick Children and patterned after the amino acid profile of egg protein, and Primene (Baxter, Deerfield, IL), an amino acid solution patterned after human umbilical cord blood and designed for the pediatric population (Table 2). Intakes of parenteral nutrition were as prescribed by the attending physician (Table 3). Parenteral intake included a solution of amino acids and glucose with a complete vitamin and mineral supplement in combination with a 20% lipid emulsion (Intralipid, Pharmacia and Upjohn) to meet nutrient needs. Parenteral nutrition solutions were infused into a peripheral or central vein using an IMED pump (Gemini PC-4, IMED Corporation, San Diego, CA). Infants were enrolled when receiving adequate amino acid and energy intake (22). Minimal enteral feeds were consumed by six infants and represented no greater than 10% of the total amino acid requirement.
Study protocol. Isotopes used for the tracer study were L-[1-13C]phenylalanine (99%, 13C, Tracer Technologies, Sommerville, MA) and L-[3,3-2H2]tyrosine (98%, 2H2, Cambridge Isotope Laboratories, Andover, MA). Quality control tests were performed by the manufacturers. Chemical purity, specified isotopic enrichment, and position was confirmed by nuclear magnetic resonance, whereas isomeric purity and a second confirmation of isotopic enrichment was performed by gas chromatography/mass spectrometry. Solutions of each tracer amino acid in 3.3% dextrose and 0.3% NaCl were prepared by the Manufacturing Pharmacy at The Hospital for Sick Children. Isotope solutions were sterilized by passage through a 0.22-µm filter before dispensing into single-use vials for storage at 4°C until use. All solutions were demonstrated to be sterile and free of bacterial growth over 7 d in culture, and to be pyrogen-free by the Limulus amebocyte lysate test (23).
Two pilot studies were performed to determine the dose of phenylalanine required to achieve measurable expired 13CO2. The L-[3,3-2H2]tyrosine dose used was that of Castillo et al. (2). At least 24 h after the start of the infusion of TPN, phenylalanine flux, hydroxylation, oxidation, percent dose oxidized, and tyrosine flux were determined using a primed, 24-h constant infusion of L-[1-13C]phenylalanine (15.6 µmol·kg-1 and 13 µmol·kg-1·h-1, respectively), and L-[3,3-2H2]tyrosine (3.6 µmol·kg-1 and 3.0 µmol·kg-1·h-1, respectively). The priming dose was infused over the first 15 min. Urine samples were collected for the measurement of background and steady state tracer amino acid enrichment, creatinine, and alternate catabolites of phenylalanine and tyrosine. Urine was used for measuring amino acid enrichment, because it has been shown to be a valid representation of plasma enrichment (24,25). One to three baseline urine samples were collected before parenteral isotope infusion, followed by one sample every 3-4 h throughout the isotope infusion. Urine was stored at -20°C until analyses were performed. Expired CO2 was collected, and the production rate was measured before and after 17-20 h of constant isotope infusion. Breath measurements were possible in only 12 subjects, because four infants were on ventilators.
Analyses. Urine was analyzed for phenylalanine and tyrosine enrichment by the method of Patterson et al. (26). Briefly, urine samples (500 µL) were deproteinized with an equal volume of 20% trichloroacetic acid followed by centrifugation at 13 000 rpm × 5 min at 23°C. The supernatant was transferred to columns containing a cationic ion exchange resin (Dowex 50W-X8, 100-200 mesh, H+ form, Bio-Rad Laboratories, Hercules, CA) for amino acid separation. The effluent solution was freeze-dried (Freezone 12L, Labconco Corp., Kansas City, MO) before derivatization to its N,O-heptafluorobutyryl isopropyl esters. Phenylalanine and tyrosine amino acid enrichment was measured on a gas chromatograph (Hewlett Packard, model 5890 series 2, Mississauga, ON) attached to a quadrupole mass spectrometer (VG Trio-2, Cheshire, England). Derivatized amino acids were splitlessly introduced into the instrument by automatic injector (Hewlett Packard 7673 injector). Separation was performed on a 30 m × 0.32-mm (inner diameter) × 1.0-µm (film thickness) fused silica capillary column (HP-5, Hewlett Packard) with helium serving as a carrier gas, coupled directly to the ion source operated under conditions of negative chemical ionization. Ammonia was used as reactant gas. Selected ion chromatographs were obtained by monitoring the m/z ratio of 383, 384 and 417, 418, 419 for the M and M + 1, and M, M + 1, and M + 2 species of phenylalanine and tyrosine, respectively. The M + 1 peak of tyrosine represents L-[1-13C]tyrosine derived from the hydroxylation of L-[1-13C]phenylalanine. Areas under the peaks were integrated by a Digital DECp 450D2LP computer (Digital Instruments, Santa, Barbara, CA), using Lab-Base software (Vacuum Generator Biotech). Isotopomer corrections were performed based on the method of Rosenblatt et al. (27).
Urinary creatinine was measured by the Jaffe reaction (28) using a Kodak Ektachem 700 Analyzer (Johnson and Johnson, Rochester, NY). Urinary phenylalanine and tyrosine concentrations were analyzed by ion-exchange chromatography (Beckman 7300 amino acid analyzer). Phenylalanine and tyrosine alternate catabolites were measured by the method of Goodman and Markey (29). The organic acids in 1-3 mL of urine were first converted to oximes of ketoacids with hydroxylamine. α-Ketocaproate was used as internal standard for oxime formation and pentadecanoic acid as the analytical internal standard. Oxime formation was followed by acidification with 6 M HCl to pH < 2.0, saturated with NaCl, and extracted in successive equal volumes of ethyl ether and diethyl ether. Extracted organic acids were dried under N2, and subsequently derivatized to trimethylsilyl derivatives with bis(trimethylsilyl)-trifluoroacetamide containing 1% trimethylchlorosilane as a catalyst. Tetracosane was used as an external standard. The trimethylchlorosilane derivatives were separated on a fused silica capillary column (HP-5, Hewlett Packard) and analyzed by gas chromatography/mass spectrometry (model 5890, Hewlett Packard) using electron impact ionization. Quantification of the phenylalanine and tyrosine alternate catabolites was performed using standard curves prepared for each compound.
Expired CO2 production, collected from a ventilated hood system was measured indirectly using a portable CO2 analyzer (1400 series, Servomex, Westech Industrial Ltd., Mississauga, ON) and mass flowmeter (5860 series, Brooks, Trillium Measurement and Control, Stouffville, ON). Complete CO2 trapping was performed by bubbling the exhaust from the CO2 analyzer at a rate of 500 mL/min, into a condenser coil containing 10 mL of 1 M NaOH for 10 min (30). CO2 enrichment was measured on a dual inlet magnetic sector isotope-ratio mass-spectrometer (VG Micromass, model 602D, Cheshire, England). Enrichment measures were expressed as APE 13CO2 over a reference standard of compressed CO2 gas.
The model of amino acid metabolism used in this study is a modification of the Waterlow et al. (31) model: Q = B + I = S + PAH where Q is the rate of phenylalanine flux (µmol·kg-1·h-1); S is the rate of phenylalanine nonoxidative disposal, a measure of the rate of phenylalanine incorporation into body protein; PAH is the rate of phenylalanine hydroxylation to tyrosine; B is the rate of phenylalanine released from body protein; and I is the rate of exogenous phenylalanine intake.
Whole body phenylalanine and tyrosine turnover was calculated from the dilution of isotope in the body amino acid pool at isotopic steady state (24,25,32): Qphe = I[Ei/Ep - 1] where QPhe is the rate of phenylalanine or tyrosine (QTyr) flux; i is the isotope infusion rate (µmol·kg-1·h-1), Ei is the enrichment of the infused isotope (APE), and Ep is the enrichment of the amino acid in the urine at isotopic steady state (APE).
To measure the rate of phenylalanine hydroxylation, the model of Clarke and Bier (33) with the modification of Thompson et al. (34) was used: QPhe→Tyr = QTyr · [ETyr/EPhe][QPhe/ iPhe + QPhe] where ETyr and EPhe are the plasma enrichments of L-[1-13C]tyrosine and L-[1-13C]phenylalanine, respectively; and iPhe is the rate of infusion of the labeled phenylalanine.
The rate of phenylalanine oxidation was calculated as described by Matthews et al. (32): OPhe = F13CO2(1/EPhe - 1/Ei) · 100 where OPhe represents phenylalanine oxidation, and F13CO2 represents the rate of 13CO2 released by phenylalanine tracer oxidation (µmol·kg-1·h-1) calculated from the following equation: F13CO2 = (FCO2)(ECO2)(44.6)(60)/(W)(RF)(100) where FCO2 is the CO2 production rate (cm3·min-1); and ECO2 is the 13CO2 enrichment in expired breath at isotopic steady state (APE). The constants 44.6 µmol·(cm3)-1 and 60 min·h-1 convert FCO2 to micromoles/h, and the factor of 100 changes APE to a fraction. W is the weight (kg) of the infant. 13CO2 retained by the body as bicarbonate is corrected using a retention factor (RF) calculated from a regression equation using energy intake as the predictor variable (35). The percent phenylalanine isotope dose oxidized was calculated as: % dose oxidized = (F13CO2/iPhe)(100).
Statistical analysis. Results are expressed as mean ± SD. Data were tested for potential covariables (i.e. gestational age, postnatal age, postconceptual age, weight, protein, and energy intake) using analysis of covariance. None of these variables were found to be significant covariables, although an interaction response was identified between energy intake and percent phenylalanine isotope dose oxidized. Because covariables were not identified, we presented data analyzed by an unpaired two-tailed t test (36). Equality of variances was tested by the F test. Alternate metabolites of phenylalanine and tyrosine data were analyzed by the Wilcoxon signed rank test (36). A p value less than or equal to 0.05 was considered significant. Statistical analyses were performed using SAS (37).
RESULTS
Clinical characteristics were not significantly different between neonates receiving Vamin or Primene as the source of amino acids (Table 1). Body weights reflected the predominance of premature infants enrolled in the study. Infants were enrolled into the study if their nutrient intakes met amino acid and energy requirement levels (22). Actual dietary intakes were not significantly different between groups (Table 3).
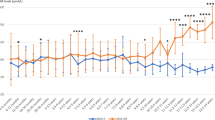
Urinary isotopic steady state was achieved for all neonates by 12 h, as defined by the absence of significant slope; therefore, urine samples collected after this point were used for calculation of the isotopic plateau (Fig. 1). Samples at each time point represent urine collected closest to that hour. The average coefficient of variation of individual plateaus was 12 (range, 2.6-24). The presence of isotope recycling was considered to be minimal as indicated by the lack of systematic increase in enrichment at plateau for individuals or group means (Fig. 1).
Phenylalanine and tyrosine metabolism. There was no difference in phenylalanine flux (Table 4) between groups. By design, phenylalanine intake was different between diet regimens (Table 4). Protein breakdown was 33% greater, and approached significance (p < 0.1) in the group receiving Primene (Table 4); however, percent flux from breakdown was found to be significantly greater in infants receiving Primene (p < 0.001) (Table 4). Phenylalanine hydroxylation was significantly greater (p < 0.004) in the neonates receiving Vamin (Table 4). Phenylalanine nonoxidative disposal (an estimate of phenylalanine incorporation into body protein) was not significantly different between groups (Table 4). No differences in tyrosine flux or intake were observed between the two groups (Table 4).
Expired 13CO2 enrichment was significantly greater in infants receiving Vamin compared with those receiving Primene, whereas the VCO2 production rate was not significantly different between groups (Table 5). Phenylalanine oxidation was significantly greater in infants receiving Vamin compared with infants receiving Primene (p < 0.02; Table 5). However, the actual precursor pool enrichment in the hepatocyte is unknown. Therefore, the percent phenylalanine isotope dose oxidized was measured and used as an index of phenylalanine oxidation that does not rely on precursor pool enrichment. Percent phenylalanine isotope dose oxidized was also found to be significantly greater (p < 0.01) in the infants receiving Vamin versus those receiving Primene. A significant (p < 0.02) interaction response was identified between energy intake and percent dose oxidized (Fig. 2). Neonates receiving Vamin experienced a reduction in the percent dose oxidized as energy intake increased, whereas those receiving Primene maintained a constant percent phenylalanine dose oxidized.
Urinary alternate catabolite excretion of phenylalanine and tyrosine was standardized to urinary creatinine. The concentration of these compounds was greater in infants receiving Vamin versus infants receiving Primene (Table 6). Phenylpyruvate and 4-hydroxyphenylpyruvate are known to degrade upon storage, and were therefore not measured (38). Excretion of the phenylalanine catabolite phenyllactate was significantly greater in Vamin compared with Primene-fed infants (p < 0.03), whereas differences in phenylacetate and 2-hydroxyphenylacetate levels were not significantly different between groups. The tyrosine metabolite 4-hydroxyphenylacetate was significantly greater in the Vamin compared with the Primene group (p < 0.01), whereas 4-hydroxyphenyllactate levels were not significantly different between groups. The sum of the alternate catabolites of phenylalanine and tyrosine metabolism was significantly greater in infants receiving Vamin versus infants receiving Primene (p < 0.003 and p < 0.03 for phenylalanine and tyrosine catabolites, respectively).
Urinary phenylalanine and tyrosine concentrations were significantly greater in infants receiving Vamin compared with those receiving Primene (phenylalanine, 130 ± 73 µmol·mmol creatinine-1, Vamin, 60 ± 52 µmol·mmol creatinine-1, Primene, p = 0.05; and tyrosine, 219 ± 132 µmol·mmol creatinine-1, Vamin, 86 ± 86 µmol·mmol creatinine-1, Primene, p < 0.05).
DISCUSSION
The present study examined phenylalanine and tyrosine metabolism in neonates receiving parenteral nutrition differing in source of amino acids. The difference in phenylalanine level in each amino acid solution, and therefore in total aromatic amino acid intake, is based on differing philosophies in the development of these solutions. Vamin, an amino acid solution designed for adults, has compensated for low tyrosine concentration by increasing the content of phenylalanine, the body's direct metabolic precursor of tyrosine. Although infants receiving Vamin exhibit good growth and nitrogen retention rates (39), some infants experience high plasma phenylalanine concentrations (7,9–11,13,14) and excrete alternate catabolites of phenylalanine and tyrosine metabolism (6,14,15). In neonates, these metabolites are normally found in low to trace amounts (40–42). Such findings imply overloading of neonatal catabolic pathways with high parenteral phenylalanine intake. Although we have not collected blood and therefore cannot provide plasma phenylalanine and tyrosine levels, we have measured urinary phenylalanine and tyrosine concentration, as an index of plasma concentration. We found that the level of urinary phenylalanine and tyrosine were elevated in infants receiving Vamin, but was within normal range for those receiving Primene. Similarly, piglets receiving Vamin experienced greater incidence of abnormal plasma phenylalanine and tyrosine levels with correspondingly higher levels of alternate catabolites of phenylalanine metabolism compared with piglets receiving the pediatric amino acid solution Vaminolact (15). These metabolic responses are of concern because of the knowledge that infants with high plasma phenylalanine due to phenylketonuria experience neurologic impairment. Lucas et al. (43) compared the neurologic outcome of 93 infants who experienced high plasma phenylalanine levels when receiving parenteral nutrition containing Vamin to (age-matched) controls. Although Lucas et al. (43) found no effect on these outcome measures at 18-mo corrected age, it would be preferable to avoid hyperphenylalaninemia as longer term consequences are unknown and research on the issue is limited.
In contrast, amino acid solutions for pediatric use have been developed with concern for the immaturities of the neonate. Due to the importance placed on the normalization of plasma amino acid levels, these solutions have not compensated for low tyrosine concentrations with additional phenylalanine. It has therefore been suggested that these solutions may be limiting in total aromatic amino acids (1,2,15). Although clinical studies comparing general purpose with pediatric amino acid solutions have found no differences in growth or nitrogen balance, this may be due to difficulties in nitrogen balance collections, small sample size, short study periods, and large variation in the patient population due to illness. Conversely, the piglet model, which grows at four times the rate of the human neonate, has revealed significant differences between groups receiving Vamin versus Vaminolact (a pediatric solution with an amino acid pattern similar to human milk protein), and provides further evidence that the pediatric solution is limiting in total aromatic amino acids (15).
Infants receiving high phenylalanine solutions are assumed to be able to hydroxylate sufficient phenylalanine to meet tyrosine requirements and dispose of excess phenylalanine that may have been infused. The neonates' ability to cope with a phenylalanine excess is dependent on the activity of the enzymes involved in the metabolism of phenylalanine and tyrosine. In the present study, infants receiving the greater intake of phenylalanine from Vamin had a significantly higher rate of hydroxylation compared with infants receiving Primene. Recent studies examining phenylalanine hydroxylation rates, using the Thompson et al. (34) modification of the Clarke and Bier (33) model, have found considerable hydroxylation in response to parenteral amino acid intake compared with a basal period where infants were receiving glucose alone (17,18). This response was not observed within a group of term infants (21) and recently in a group of 32-wk-old neonates (20) receiving an amino acid solution modest in phenylalanine supply. Although these results provide an indication that phenylalanine hydroxylase enzyme can be stimulated by substrate, the degree of response in relation to phenylalanine intake is unknown.
The hydroxylation rate observed in the neonates receiving Primene (22 µmol·kg-1·h-1) supports other studies where infants receiving pediatric amino acid solutions demonstrated phenylalanine hydroxylation rates ranging from 11 to 22 µmol·kg-1·h-1 (20,21). Similarly, in studies where neonates received parenteral nutrition with elevated phenylalanine, hydroxylation was found to be greater, and in the range of 37-48 µmol·kg-1·h-1 (present study, 42 µmol·kg-1·h-1) (2,17). These results support the rationale that the neonate does respond to increased phenylalanine intake by increasing the rate of phenylalanine hydroxylation to tyrosine. Nevertheless, the concern exists for whether the increased phenylalanine hydroxylation that occurs in these infants is sufficient to meet tyrosine needs and to dispose of possible excess phenylalanine intake.
Of interest was the finding that the percent of phenylalanine tracer dose oxidized deceased with increasing energy intake in infants receiving Vamin but not Primene. This suggests that increased energy intake supports improved utilization of the large supply of phenylalanine in the Vamin solution. Lucas et al. (43) demonstrated that poor tolerance of a high phenylalanine intake in neonates receiving Vamin did not occur when the total energy to protein energy intake ratio was 8.5:1 but occurred occasionally with a ratio of 6.5:1. In the present study the total energy to protein energy ratio was 8.1 ± 1.95 for infants receiving Vamin and 7.8 ± 0.82 for those receiving Primene. The present data suggests that at an energy intake of 350 kJ·kg-1·d-1, phenylalanine utilization was limited by energy intake. For infants receiving Primene, the low percent dose oxidized (6%, Table 5) and lack of significant change due to increasing energy intake indicates that phenylalanine oxidation is at its basal level.
The greater urinary excretion of alternate catabolites of phenylalanine and tyrosine in infants receiving Vamin supports the conclusion that the level of phenylalanine in Vamin is in excess of requirement. This said, although there is concern for the presence of these catabolites, there is no direct evidence that these compounds result in harm. Phenylalanine catabolites were also found in the urine of piglets receiving Vamin or a phenylalanine-supplemented pediatric amino acid solution, whereas piglets receiving the unsupplemented solution presented with only negligible amounts of the metabolites (15). Large concentrations of tyrosine catabolites were also found in infants (less than 33-wk gestational age) receiving Vamin-based TPN (14). The presence of these metabolites suggests an overload to the catabolic pathway of phenylalanine toward oxidation even in the presence of significant hydroxylation and oxidation. The parenteral route of administration of amino acids may also have a role in causing increased excretion of these catabolites. Studies comparing amino acid metabolism in subjects receiving nutrient intake parenterally versus enterally demonstrate significantly different responses in metabolism (44–46).
Apparent phenylalanine balance, estimated from the difference between phenylalanine nonoxidative disposal and phenylalanine appearance from tissue breakdown, gives an indication of the net body phenylalanine retention. Phenylalanine balance was 17 µmol·kg-1·h-1 for neonates receiving Vamin and 6 µmol·kg-1·h-1 for those receiving Primene. Assuming that there is 272 µmol of phenylalanine per g of mixed body protein (47), infants receiving Vamin experienced a net protein retention of 1.5 g, whereas those receiving Primene experienced a net retention of 0.5 g of protein. Phenylalanine and tyrosine requirements have recently been measured in the TPN piglet model (4,48). From this work we estimated that the optimal phenylalanine and tyrosine content of parenteral amino acid solutions for the neonate should be 3.2 and 2.7% total amino acids, respectively (4,48). Further, Vamin contains 7.9% phenylalanine, which our data, combined with the earlier piglet data (48), would seem to be in excess of that required.
This study specifically addresses phenylalanine and tyrosine metabolism in infants receiving Vamin or Primene, yet these solutions also differ in the level of other amino acids. Because phenylalanine has been shown to be the first limiting amino acid for protein synthesis in the piglet model of the parenterally fed neonate (15), the kinetic measurements within this report is largely responsive to differences in the level of phenylalanine versus differences in overall pattern of amino acids. Mitton and Garlick (49) performed a similar study where they examined leucine kinetics as an index of overall body protein turnover in premature infants receiving either Vamin or Vaminolact as the source of parenteral amino acid intake. These solutions differ similarly to the present study in pattern of amino acids. These investigators did not find any difference in leucine kinetics or overall protein metabolism in infants receiving either solutions. Comparable to this situation, is the study of Wykes et al. (15) in the piglet model of the parenterally fed neonate receiving Vamin, Vaminolact, or Vaminolact supplemented with phenylalanine to the level of Vamin. Piglets receiving Vamin had similar differences in phenylalanine kinetics as those found in the present study. The improved quality of the balance of the pediatric solution Vaminolact versus Vamin was demonstrated by the improved nitrogen balance that occurred when Vaminolact was supplemented with phenylalanine. From this evidence, it is suggested that it is most likely that the differences that exist in the pattern of amino acids would result in Primene supporting improved utilization (15), minimizing any differences observed in phenylalanine and tyrosine kinetics.
In summary, neonates receiving the high phenylalanine solution (Vamin) responded to increased intake of phenylalanine by increasing hydroxylation and oxidation rates compared with infants receiving the moderate phenylalanine solution (Primene). This is the first time, to our knowledge, that aromatic amino acid kinetics using stable isotope tracers were compared in infants receiving two commercially available amino acid solutions. These results suggest that current pediatric TPN amino acid mixtures may be limiting growth in the neonate. However, the high phenylalanine intake of Vamin results in urinary excretion of phenylalanine and tyrosine and their catabolites, suggesting that increased parenteral phenylalanine intake may not be the most efficient or effective means of meeting the total aromatic amino acid requirement in the neonate. There is therefore a need to determine the tyrosine requirements of parenterally fed human neonates.
Abbreviations
- TPN:
-
total parenteral nutrition
- APE:
-
atoms percent excess
References
Snyderman SE 1971 The protein and amino acid requirements of the premature infant. In: Jonxis JHP, Visser HKA, Troelstra JA (eds) Metabolic Processes in the Foetus and Newborn. Stenfert Kroese, Leiden, The Netherlands, 128–141.
Castillo L, Yu YM, Marchini JS, Chapman TE, Sanchez M, Young VR, Burke JF 1994 Phenylalanine and tyrosine kinetics in critically ill children with sepsis. Pediatr Res 35: 580–588
Uauy R, Green HL, Heird WC 1993 Conditionally essential nutrients: cysteine, taurine, tyrosine, arginine, glutamine, choline, inositol, and nucleotides. In: Tsang RC, Lucas A, Uauy R, Zlotkin S (eds) Nutritional Needs of the Preterm Infant. Williams & Wilkins, Baltimore, 267–280.
House JD, Pencharz PB, Ball RO 1997 Tyrosine kinetics and requirement during a total parenteral nutrition in the neonatal piglet: the effect of glycyl-L-tyrosine supplementation. Pediatr Res 41: 575–583
Rassin DK 1989 Amino acid metabolism in total parenteral nutrition during development. In: Friedman M (ed) Absorption and Utilization of Amino Acids. CRC Press, Boca Raton, FL, 71–85.
Chessex P, Zebiche H, Pineault M, Lepage D, Dallaire L 1985 Effect of amino acid composition of parenteral solutions on nitrogen retention and metabolic response in very-low-birth weight infants. J Pediatr 106: 111–117
Walker V, Hall MA, Bulusu S, Allan A 1986 Hyperphenylalaninaemia in parenterally fed newborn babies. Lancet 2: 1284
Evans SJ, Wynne-Williams TC, Russell CA, Fairbrother A 1986 Hyperphenylalaninaemia in parenterally fed newborn babies. Lancet 2: 1404–1405
Rigo J, Senterre J, Putet G, Salle B 1987 A new amino acid solution specially adapted to preterm infants. Clin Nutr 6: 105–109
Puntis JW, Edwards MA, Green A, Morgan I, Booth IW, Ball PA 1986 Hyperphenylalaninaemia in parenterally fed newborn babies. Lancet 2: 1105–1106
Mitton SG, Burston D, Brueton MJ 1988 Hyperphenylalaninaemia in parenterally fed newborn infants. Lancet 2: 1497–1498
McIntosh N, Mitchell V 1990 A clinical trial of two parenteral nutrition solutions in neonates. Arch Dis Child 65: 692–699
Puntis JW, Ball PA, Preece MA, Green A, Brown GA, Booth IW 1989 Egg and breast milk based nitrogen sources compared. Arch Dis Child 64: 1472–1477
Walker V, Mills GA 1990 Metabolism of intravenous phenylalanine by babies born before 33 wk of gestation. Biol Neonate 57: 155–166
Wykes LJ, House JD, Ball RO, Pencharz PB 1994 Amino acid profile and aromatic amino acid concentration in total parenteral nutrition: effect on growth, protein metabolism and aromatic amino acid metabolism in the neonatal piglet. Clin Sci 87: 75–84
Delvalle JA, Greengard O 1977 Phenylalanine hydroxylase and tyrosine aminotransferase in human fetal and adult liver. Pediatr Res 11: 2–5
Shortland GJ, Walter JH, Fleming PJ, Halliday D 1994 Phenylalanine kinetics in sick preterm neonates with respiratory distress syndrome. Pediatr Res 36: 713–718
Kilani RA, Cole FS, Bier DM 1995 Phenylalanine hydroxylase activity in preterm infants: is tyrosine a conditionally essential amino acid? Am J Clin Nutr 61: 1218–1223
van Toledo-Eppinga L, Kalhan SC, Kulik W, Jakobs C, Lafeber HN 1996 Relative kinetics of phenylalanine and leucine in low birth weight infants during nutrient administration. Pediatr Res 40: 41–46
Clark SE, Karn CA, Ahlrichs JA, Wang J, Leitch CA, Liechty EA, Denne SC 1997 Acute changes in leucine and phenylalanine kinetics produced by parenteral nutrition in premature infants. Pediatr Res 41: 568–574
Denne SC, Karn CA, Ahlrichs JA, Dorotheo AR, Wang J, Liechty EA 1996 Proteolysis and phenylalanine hydroxylation in response to parenteral nutrition in extremely premature and normal newborns. J Clin Invest 97: 746–754
Zlotkin SH, Stallings VA, Pencharz PB 1985 Total parenteral nutrition in children. Pediatr Clin North Am 32: 381–400
Pearson FC 1979 The Limulus amebocyte lysate endotoxin assay: current status. Am J Med Technol 45: 704–709
De Benoist B, Abdulrazzak Y, Brooke OG, Halliday D, Millward DJ 1984 The measurement of whole body protein turnover in the preterm infant with intragastric infusion of L-[1-13C]leucine and sampling of the urinary leucine pool. Clin Sci 66: 155–164
Wykes LJ, Ball RO, Menendez CE, Pencharz PB 1990 Urine collection as an alternative to blood sampling: a noninvasive means of determining isotopic enrichment to study amino acid flux in neonates. Eur J Clin Nutr 44: 605–608
Patterson BW, Hachey DL, Cook GL, Amann JM, Klein PD 1991 Incorporation of a stable isotopically labeled amino acid into multiple human apolipoproteins. J Lipid Res 32: 1063–1072
Rosenblatt J, Chinkes D, Wolfe M, Wolfe RR 1992 Stable isotope tracer analysis by GC-MS, including quantification of isotopomer effects. Am J Physiol 263: E584–E596
Butler AR 1975 The Jaffe reaction. Identification of the coloured species. Clin Chim Acta 59: 227–232
Goodman SI, Markey SP 1981 Diagnosis of organic acidemias by gas chromatography-mass spectrometry. Lab Res Methods Biol Med 6: 1–158
Jones PJ, Pencharz PB, Clandinin MT 1985 Whole body oxidation of dietary fatty acids: implications for energy utilization. Am J Clin Nutr 42: 769–777
Waterlow JC, Garlick PJ, Millward DJ 1978 Protein Turnover in Mammalian Tissues and in the Whole Body. Elsevier/North-Holland, Amsterdam, The Netherlands, 179–223.
Matthews DE, Motil KJ, Rohrbaugh DK, Burke JF, Young VR, Bier DM 1980 Measurement of leucine metabolism in man from a primed, continuous infusion of L-[1-13C]leucine. Am J Physiol 238: E473–E479
Clarke JT, Bier DM 1982 The conversion of phenylalanine to tyrosine in man. Direct measurement by continuous intravenous tracer infusions of L-[ring-2H5]phenylalanine and L-[1-13C]tyrosine in the postabsorptive state. Metabolism 31: 999–1005
Thompson GN, Pacy PJ, Merritt H, Ford GC, Read MA, Cheng KN, Halliday D 1989 Rapid measurement of whole body and forearm protein turnover using a [2H5]phenylalanine model. Am J Physiol 256: E631–E639
Van Aerde JE, Sauer PJ, Pencharz PB, Canagarayar U, Beesley J, Smith JM, Swyer PR 1985 The effect of energy intake and expenditure on the recovery of 13CO2 in the parenterally fed neonate during a 4-hour primed constant infusion of NaH13CO3 . Pediatr Res 19: 806–810
Rosner B 1990 Fundamentals of Biostatistics. Duxbury Press, Belmont, CA, 186–317.
SAS Institute 1991 SAS/STAT Guide for Personal Computers. SAS Institute, Inc., Cary, NC
Vavich JM, Howell RR 1971 Identification and quantification of urinary metabolites of phenylalanine in phenylketonuria. J Lab Clin Med 77: 159–167
Duffy B, Gunn T, Collinge J, Pencharz P 1981 The effect of varying protein quality and energy intake on the nitrogen metabolism of parenterally fed very low birthweight (less than 1600 g) infants. Pediatr Res 15: 1040–1044
Mitchell GA, Lambert M, Tanguay RM 1995 Hypertyrosinemia. In: Scriver CR, Beauder AL, Sly WS, Valle D (eds) The Metabolic and Molecular Bases of Inherited Disease. McGraw-Hill, Toronto, 1077–1106.
Bremer HJ, Duran M, Kamerling JP, Przyrembel H, Wadman SK 1981 Disturbances of Amino Acid Metabolism: Clinical Chemistry and Diagnosis. Urban & Schwarzenberg, Baltimore, 307–325.
Chalmers RA, Lawson AM 1998 Organic Acids in Man. Chapman & Hall, New York, 181–186.
Lucas A, Baker BA, Morley RM 1993 Hyperphenylalaninaemia and outcome in intravenously fed preterm neonates. Arch Dis Child 68: 579–583
Wykes LJ, Ball RO, Menendez CE, Ginther DM, Pencharz PB 1992 Glycine, leucine, and phenylalanine flux in low-birth-weight infants during parenteral and enteral feeding. Am J Clin Nutr 55: 971–975
Adeola O, Wykes LJ, Ball RO, Pencharz PB 1995 Comparison of oral milk feeding and total parenteral nutrition in neonatal pigs. Nutr Res 15: 245–265
Beaufrere B, Fournier V, Salle B, Putet G 1992 Leucine kinetics in fed low-birth-weight infants: importance of splanchnic tissues. Am J Physiol 263: E214–E220
Widdowson EM 1981 Changes in body composition during growth. In: David J, Dobbing J (eds) Scientific Foundations of Paediatrics. Heinemann, London, 330–342.
House JD, Pencharz PB, Ball RO 1997 Phenylalanine requirements determined by using L-[1-14C]phenylalanine in neonatal piglets receiving total parenteral nutrition supplemented with tyrosine. Am J Clin Nutr 65: 984–993
Mitton SG, Garlick PJ 1992 Changes in protein turnover after the introduction of parenteral nutrition in premature infants: comparison of breast milk and egg protein-based amino acid solutions. Pediatr Res 32: 447–454
Acknowledgements
The authors thank Larry Fisher for his assistance with mass spectrometric analysis, and Joan Brennan for support in the Neonatal Intensive Care Unit. We also appreciate the efforts of the staff in the Neonatal Intensive Care Unit and Pharmacy at The Hospital for Sick Children, and especially thank the families of the neonates participating in the study.
Author information
Authors and Affiliations
Additional information
Supported by the Medical Research Council, Grant MT 12928. S.A.R. was the recipient of a Fonds pour la Formation de Chercheurs et l'Aide a la Recherche studentship. Primene was generously donated by Baxter, Deerfield, IL.
Rights and permissions
About this article
Cite this article
Roberts, S., Ball, R., Filler, R. et al. Phenylalanine and Tyrosine Metabolism in Neonates Receiving Parenteral Nutrition Differing in Pattern of Amino Acids. Pediatr Res 44, 907–914 (1998). https://doi.org/10.1203/00006450-199812000-00014
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1203/00006450-199812000-00014
This article is cited by
-
The metabolome and bacterial composition of high-moisture Italian ryegrass silage inoculated with lactic acid bacteria during ensiling
Biotechnology for Biofuels and Bioproducts (2023)
-
Protein/Amino Acid Metabolism and Nutrition in Very Low Birth Weight Infants
Journal of Perinatology (2001)