Abstract
The kidney provides an important contribution to permit the fetus to successfully transition to an independent existence by production of urine with significantly different osmolality compared with plasma. Although recent work has uncovered many aspects of the maturation and regulation of the renal concentrating and diluting mechanism, understanding of how alterations in the expression of aquaporin (AQP) water channels contribute to the formation of urine in the perinatal period is incomplete. Here, we report that both AQP-2 and -3 are expressed during fetal life as early as embryonic d 18 in ureteric buds of rat kidneys, where each is localized to the apical and basolateral membranes of epithelial cells, respectively. Northern analyses demonstrate that the 1.9-kb AQP-2 transcript is present in fetal and postnatal rat kidneys similar to that observed in adults. AQP-2 mRNA expression increases after d 3 of postnatal life. Immunoblotting reveals an increase in total kidney AQP-2 protein particularly with respect to its glycosylated form after postnatal d 3. AQP-3 protein also exhibits a similar alteration likely due to a similar increase in its glycosylation state. Both AQP-2 and AQP-3 display a distribution in the collecting ducts of human postnatal infants and adults identical to that exhibited in rat kidneys. These data show that both AQP-2 and -3 are present in collecting duct epithelia of fetal and postnatal kidneys. Thus, the reduced AVP-responsiveness and decreased urinary concentrating ability of the kidney during the fetal and immediate postnatal period does not appear to be caused by lack of AQP-2 or AQP-3 proteins.
Similar content being viewed by others
Main
During development, epithelial cells of the kidney undergo a complex series of maturational processes resulting in the formation of tubular and glomerular elements. The epithelial cells of the ureteric bud play a critical inductive role in normal fetal nephron development and ultimately form collecting ducts in the adult kidney. Collecting duct epithelial cells in the adult kidney are highly polarized and capable of targeting proteins to apical and basolateral locations in the cells, resulting in vectorial transport of ions and water(1). In contrast, limited data are available regarding how ureteric bud epithelial cells acquire the polarity, protein targeting, and transport functions as the kidney develops during intervals of fetal and PN life.
After birth, the newborn must adapt to varying oral solute loads. However, nonstressed fetal and newborn animals generally produce urine that is hypoosmotic compared with plasma(2–4) and are unable to achieve a urinary osmolality similar to that present in adult animals until a PN age of 3 wk in both rats and rabbits or 2-12 mo in humans(5–7). Production of a hypertonic urine is dependent on a combination of factors including proximal tubule solute reabsorption, thick ascending limb of Henle NaCl transport, the formation of a hypertonic medullary interstitium, and vasopressin (AVP)-elicited increases in both water and urea permeabilities in the kidney collecting ducts(3, 4).
Previous work has suggested that multiple factors may contribute to the inability of the perinatal kidney to produce hyperosmotic urine. These include a smaller loop of Henle, decreased medullary tonicity, increased production of prostaglandins, and a reduced AVP-elicited water permeability(Pf) response of the collecting duct(2, 5). Although AVP synthesis and release by the fetal neurohypophysis is intact(8, 9) and collecting duct V2 receptors are present(10), data from PN rats(11, 12) and rabbits(12) have suggested that the capacity of the fetal collecting duct to respond to AVP is “immature,” and may result from both a reduction in collecting duct adenylate cyclase activity and AVP receptor-binding capacity.
Recent work has focused on how AQP water channels function in the development of the renal concentrating mechanism(13). AQP-1 is present in both the proximal tubules and thin descending limbs of Henle of kidney(14). Although AQP-1 significantly increases membrane Pf, facilitates the constitutive water transport in these cells(15), and is expressed in human fetal kidney(16), humans lacking AQP-1 protein do not appear to exhibit major alterations in fluid and electrolyte homeostasis(17). In contrast, significant AQP-1 expression does not occur in rats until after birth(18, 19).
APQ-2 is the AVP-elicited water channel present in both cortical and medullary collecting ducts(20–23) and mutations in the human AQP-2 gene produce nephrogenic diabetes insipidus(24). AVP stimulation of rat collecting duct not only produces rapid increases in its Pf by insertion of AQP-2 into the apical membrane(20–22) but augments Pf on a long-term basis by promoting increases in the cellular content of AQP-2 protein(22, 25). Recent studies in rats(7, 19, 26) have shown that AQP-2 is expressed by the fetal kidney and its expression increases after 1 wk of PN life. More limited studies in human kidney have reported similar data together with an alteration in the pattern of AQP-2 glycosylation in fetal kidney(16).
AQP-3 is also present on the basolateral membrane of medullary collecting ducts that express AQP-2(27). Although AQP-3 is expressed in fetal rat(19) and adult human(28) kidneys, its PN expression has been studied exclusively by quantitative PCR and immunohistochemistry(19). Thus, alterations in the PN expression of AQP-2 and -3 proteins in rat kidneys have not been carefully studied, particularly in the immediate (0-72 h) postpartum interval.
In this report, we have used a combination of immunohistochemistry, immunoblotting, and Northern analyses to determine AQP-2 and -3 expression in both fetal and PN rats. These data confirm and extend recent demonstrations of AQP-2 and -3 expression in fetal rat kidneys and demonstrate that the pattern of glycosylation of both AQP-2 and -3 is altered in PN rats compared with that displayed in kidneys from adult animals. Taken together, these data provide additional evidence suggesting that PN alterations in the expression of AQP water channels may contribute to development of an intact renal concentrating mechanism in adult animals.
METHODS
Preparation of antibodies. Rabbit anti-AQP-3 antiserum (peptide sequence: EAENVKLAHMKHKEQ, amino acids 263-277 of AQP-3) was prepared in manner identical to that described previously for AQP-2(22). Both were affinity-purified before their use via peptide linkage to SulfoLink Coupling gel (Pierce, Rockford, IL) as previously described(22).
Experimental animals. All animal study protocols were approved by the Animal Care and Use Committee at Children's Hospital and conform to guidelines prescribed by the National Institutes of Health. Male Sprague-Dawley adult rats (250 g), timed-pregnancy female Sprague-Dawley rats, and fetuses of gestational ages E15-21, as well as PN rats were used in this study. Adult and perinatal rats were killed after inducing anesthesia with intraperitoneal pentobarbital or CO2 inhalation, respectively. Kidneys were then harvested from the adult, fetal, and newborn rats and prepared for either immunohistochemistry, immunoblotting, or Northern analyses.
Human tissue. Human tissue was obtained from either normal adult kidney specimens after nephrectomy (secondary to renal cell carcinoma) or autopsy of a 3-d-old infant product of a 37½-wk gestation who died of respiratory failure secondary to a diaphragmatic hernia.
Immunohistochemistry. E15 (n = 8), E18 (n = 4), E19 (n = 3), E20 (n = 6), E21 (n = 2), and PN1 (n = 3) as well as adult rats were studied using immunohistochemistry. After induction of anesthesia as outlined above, rat kidneys were fixed by either immersion (E15, E18, and E19) or perfusion (E20, E21, PN1, and adult) fixation in situ using freshly prepared 4% paraformaldehyde in isotonic PBS, and then harvested and prepared for sectioning as previously described(29). Immunohistochemistry was performed as described previously(29) with slight modifications. Briefly, 4-μm sections were fixed in acetone and incubated with either primary antibody-AQP-2 (1:3000) or AQP-3 (1:750) antisera, or rabbit anti-bovine keratin antisera (1:300) (DAKO, Carpenteria, CA) as a positive control-or with anti-AQP-2 or -3 preincubated with peptide overnight. Sections were rinsed, incubated with peroxidase-conjugated, donkey anti-rabbit secondary antibody(Jackson Immunoresearch, Westgrove, PA), and developed using the immunoperoxidase/amino-ethylcarbazole technique and counterstained with Gill's hematoxylin #3 or methyl green. Specimens were then mounted in glycerol gelatin (Sigma Chemical Co., St. Louis, MO) and examined by light microscopy, and selected sections were photographed, scanned, and printed.
RNA isolation and analysis of AQP-2 expression. Northern analyses were performed on specimens of kidneys, brain, and lung collected and pooled separately from both adult and perinatal rats (E18, E20, E21, and PN1-14). All PN rats received normal maternal care. All tissues were either processed immediately or snap frozen in liquid N2 and stored at -70°C for further use. Total RNA was isolated using STAT 60 Kit (Tel-Test B, Friendswood, TX) and subjected to Northern analyses as described previously(30). Blots were hybridized with full-length AQP-2 probe(obtained from K. Fushimi, Tokyo, Japan)(20) and32 P-labeled with New England Biolabs (Beverly, MA) random priming kit(5 × 105 cpm/mL). Blots were washed under high stringency conditions (0.1 × SSC + 0.1% SDS at 55 °C), then subjected to autoradiography at -70 °C using Lightening Plus intensifier screens and XAR-5 film (Eastman Kodak, Rochester, NY). Individual lanes of autoradiograms were quantified using photoanalytic image processing with National Institutes of Health Image (Bethesda, MD) after normalizing the RNA content toβ-actin.
Immunoblotting and N-glycanase digestions. Immunoblotting analyses of protein fractions containing AQP-2 and -3 were performed on pooled specimens of adult and perinatal (E21 and PN1-14) rat kidney homogenates using anti-AQP-2 and -3 antisera as described previously(30). All tissues were either processed immediately or snap frozen in liquid N2 and stored at -70 °C for further use.
Rat AQP-2 was deglycosylated with N-glycanase (Genzyme Corp., Cambridge, MA) using a protocol reported previously for the deglycosylation of human AQP-2 by Sasaki et al.(23) with modifications. When our pilot experiments demonstrated that inclusion of 1,10-o-phenanthroline as a recommended protease inhibitor in the deglycosylation reaction mixture destroyed AQP-2 immunoreactivity in immunoblots of both native and N-glycanase-digested AQP-2 proteins, we replaced it with a mixture of 2 mM EDTA, phenylmethsulfonyl fluoride, and leupeptin (final concentrations of 1 mM, 5 μg/mL, and 5μg/mL, respectively). After overnight incubation with N-glycanase, the reaction mixture was denatured in SDS and analyzed by Western blotting as described above.
RESULTS
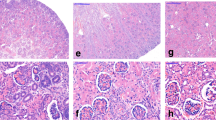
AQP-2 and -3 proteins are expressed by collecting duct epithelial cells in adult, PN, and fetal rats as well as PN and adult humans. We(22) and others(23) have shown that AQP-2 staining is most prominent in the apical membrane of IMCD epithelial cells in adult rats (Fig. 1,a and b). In a pattern similar to that displayed by IMCD from adult rats, abundant apical membrane AQP-2 staining was observed in the IMCD of PN1 rats (Fig. 1c) as well as in large ureteric buds in E21(Fig. 1d) and E20 (Fig. 1e) fetal rats. However, only moderate apical AQP-2 staining was observed in the ureteric buds of kidneys from E19 rat kidneys (Fig. 2a), and only faint staining was visualized in tubules of E18 kidneys (Fig. 2b). No AQP-2 staining was evident in the branching ureteric buds of E15 kidneys (Fig. 2c).
Immunolocalization of AQP-2 protein in rat kidneys using affinity purified rabbit anti-AQP-2 antiserum. Specific binding of AQP-2 antiserum is indicated by the rose colored reaction product. In adult kidney, AQP-2 staining is present predominantly on the apical membrane of collecting duct epithelial cells (a, ×2000) where its staining is ablated by the addition of excess immunizing AQP-2 peptide (b, ×2000). AQP-2 is also present in the apical membrane of epithelia in both PN1 kidneys(c, ×200), as well as fetal E21 (d, ×800) and E20 kidneys (e, ×800). In contrast to smaller tubules in the E20 fetal kidney (arrowheads), only tubules which possess the morphology of large ureteric buds stain for AQP-2.
Immunolocalization of AQP-2 protein in kidneys from fetal E19, E18, and E15 rats. In contrast to Figure 1, only moderate apical membrane AQP-2 staining is observed in E19 kidneys(a, ×950) and faint AQP-2 staining is present in E18 kidneys(b, ×950). No AQP-2 staining is detected in branching ureteric buds of E15 kidneys (c, ×950).
The pattern of AQP-3 protein expression in sections derived from identical groups of rats in Figures 1 and 2 is displayed in Figure 3. AQP-3 protein was confined to IMCD basolateral membranes in both adult (Fig. 3a) and PN1 rats (Fig. 3,b and c). In kidneys from E20 fetal rats, intense basolateral AQP-3 staining was routinely observed (Fig. 3d). In a manner similar to that observed for AQP-2, AQP-3 staining was much less intense in kidneys of E18 rats (Fig. 3e), and no staining was detectable in earlier gestational ages (data not shown). These data confirm and extend the recent report of AQP-2 and -3 immunocytochemistry in rats by Yamamoto et al.(19).
Immunolocalization of AQP-3 protein in adult, PN, and fetal rat kidneys. AQP-3 staining is localized to the basolateral membranes of collecting duct epithelial cells in both adult rat kidney (a,×2000) as well as PN1 kidneys (b and c, ×200 and ×800, respectively). In E20 fetal rat kidneys, the basolateral membranes of ureteric buds possess AQP-3 (d, ×800). In contrast, AQP-3 staining of the ureteric buds of E18 kidneys (e,×800) is less much intense compared with that displayed in a-d.
Although AQP-1 and -2 have been recently localized in kidney tissue derived from human fetuses and infants(16), no data are available comparing the distribution of AQP-2 versus AQP-3 in human infants. As shown in Figure 4, immunocytochemistry using the combination of anti-AQP-2 and -3 antisera shown in Figures 1–3 demonstrates that corresponding human AQP-2 and -3 proteins are present in pathologic kidney specimens from adult (Fig. 4,a and c) and a 3-d-old child (Fig. 5,b and d). These data confirm and extend an earlier report(16) showing that human AQP-2 displays an apical membrane distribution in adult human (Fig. 4a) kidney. The combination of data displayed in Figures 3 and 4 localizes AQP-3 protein to the basolateral membranes of collecting duct epithelial cells in adult, fetal, and PN rats as well as adult and neonatal humans.
Immunolocalization of AQP-2 and -3 proteins in human adult and infant kidney using anti-AQP-2 and -3 antisera. Sections obtained from pathologic specimens of either a human adult nephrectomy (a and c) or autopsy of a 3-d-old infant (b and d) were stained using either anti-AQP-2 (a and b) or AQP-3(c and d) antisera. Note that AQP-2 localizes predominantly to the apical membranes of collecting duct epithelial cells in both adult (a, ×600), as well as neonatal kidney (b, 1500). In contrast, AQP-3 localizes to the corresponding basolateral membranes of the collecting duct in adult (c, ×600) and neonatal kidney(d, ×1500).
Expression of AQP-2 in kidney but not lung or brain of fetal, PN, and adult rats. Northern analyses was performed on twenty micrograms of total RNA per lane from the kidney, lung, and brain of E18, E20, PN1, and adult rats using a 32P-labeled AQP-2 cDNA probe. The 1.9-kb AQP-2 transcript is expressed exclusively in kidney (K), and not brain (B) or lung (L) of adult, PN1, and E20 rats. No AQP-2 transcript is detected in the E18 kidneys after standard autoradiograph exposure times.
Comparative Northern analyses of total RNA from fetal, PN, and adult rat kidneys. To determine when AQP-2 mRNA expression is altered in the kidneys of rats during fetal and PN development, Northern analyses of total RNA prepared from kidneys of various ages was performed as shown in Figures 5 and 6. The identical 1.9-kb AQP-2 transcript was expressed exclusively by the kidney and not lung or brain of fetal, PN, and adult rats (Fig. 5). However, no corresponding AQP-2 transcript was detectable in E18 total RNA from whole kidneys after a 72 h autoradiographic exposure despite the presence of AQP-2 protein by immunocytochemistry as shown in Figure 2. This apparent discrepancy is likely due to the greater sensitivity of immunohistochemistry compared with standard northern analyses. Consistent with these data are the recent report(19) of detection of AQP-2 mRNA using a sensitive ribonuclease protection assay in E18 rat kidney. Taken together, these data demonstrate that both AQP-2 protein and mRNA are expressed by epithelial cells of the ureteric bud and collecting duct in both fetal and PN rats.
Alterations in AQP-2 expression in PN rat pups. Total kidney RNA (30 μg/lane) prepared from kidneys pooled from five pups harvested at PN1, 2, 3, 6, 10, and 14 (total of 30 animals) was subjected to Northern blotting analyses using 32P-labeled AQP-2 and β-actin cDNA probes. Quantitation of relative AQP-2 expression (upper panel) corrected for differences in loading with β-actin expression(middle panel) revealed no alterations in AQP-2 expression during the initial 3 d of PN life (bottom panel). In contrast, a 2.5-fold increase in AQP-2 mRNA expression was observed during the interval between 6 and 14 d of PN life.
To determine whether AQP-2 expression is altered significantly during the immediate PN period, AQP-2 mRNA was quantified in groups of rat pups from multiple litters at various intervals during the initial 14 d of PN life (Fig. 6). No change in AQP-2 expression occurs during the initial 3 d interval after birth after which there is a corresponding increase of approximately 2.5-fold after 6-14 d of PN life. This 2.5-fold increase in AQP-2 mRNA observed here is consistent with previous reports using ribonuclease protection assays to demonstrate that an approximately 2-fold increase in AQP-2 occurs during the initial 2 wk after birth in rats(19). To further examine as to whether variations in AQP-2 expression occur within a single litter of rat pups, Northern analyses was performed in groups of littermates after 2, 14, 22, and 72 h of PN life and no significant changes in AQP-2 expression was observed (Fig. 7).
Lack of significant alterations in AQP-2 expression in littermates of rat pups during the first 72 h of PN life. Total kidney RNA (30μg/lane) was prepared from individual kidneys of single rats selected from a single litter of 13 animals harvested at intervals of 2 h (n = 3), 14 h (n = 3), 22 h (n = 3), and 72 h (n = 4) after birth while receiving normal maternal care. Laser densitometry quantification of the resulting AQP-2 bands from autoradiography demonstrate no differences in AQP-2 mRNA expression during the first 72 h of PN life.
Immunoblotting analyses of AQP-2 and -3 proteins in fetal, PN, and adult rat kidney. Previous immunoblotting studies of AQP-2 protein in PN rat kidneys have been limited to studies of rat pups greater than 10 d of age(7). Immunoblotting of AQP-1 and -2 from human fetal kidneys(16) suggest that both AQPs exhibit overall decreases in the proportion of AQP protein possessing covalently linked carbohydrate as detected by alterations in their SDS-PAGE electrophoretic mobilities. To determine whether AQP-2 protein expressed in fetal rat kidney also exhibited alterations in its electrophoretic mobility due to differences in its glycosylation, crude membrane fractions prepared from E21 fetal and adult rat kidneys were immunoblotted before and after digestion with N- glycanase. As shown in Figure 8 membranes from E21 kidneys (lane 1) consistently displayed (n = 7) a reduction in the intensity of the glycosylated 35-45-kD AQP-2 band compared with that present in the adult kidney (lane 3). Thus, in contrast to AQP-2 protein from adult kidney that display a 4.2 to 1 ratio of the 35-45-kD glycosylated AQP-2 reactive versus its corresponding 29-kD nonglycosylated counterpart, the ratio of glycosylated to nonglycosylated AQP-2 in E21 membranes was approximately 1.2 to 1.
Immunoblot and N-glycanase digestion analyses of AQP-2 protein expression in adult and fetal rat kidneys. Crude membrane fractions prepared from E21 (lanes 1 and 2) and adult(lanes 3 and 4) rat kidneys were subjected to immunoblot analyses using anti-AQP-2 antiserum before (lanes 1 and 3) or after (lanes 2 and 4) N-glycanase digestion. In contrast to the adult kidney that displays AQP-2 bands of 29 and 35-45 kD(lane 3), E21 rat kidney membranes show a diminished glycosylated 35-45-kD AQP-2 band (lane 1). N-Glycanase digestion of duplicate samples displayed in lanes 1 and 3 demonstrates complete conversion of the 35-45-kD bands of both adult (lane 4) and E21 (lane 2) into corresponding bands of approximately 30 kD.
N-Glycanase removes Asn-linked complex oligosaccharides from proteins by hydrolysis of the β-aspartyl-glycosylamine bond between Asn and the carbohydrate moiety(31). As demonstrated in experiments with human AQP-2(23), removal of Asn-linked carbohydrate from AQP-2 protein should result in the disappearance of the broad 35-45-kD immunoreactive AQP-2 band and the appearance of new band of approximately 28-30 kD, corresponding to deglycosylated AQP-2 polypeptide. As shown in Figure 8, N-glycanase treatment of AQP-2 from either E-21 fetal (lane 2) or adult (lane 4) rat kidney medulla converts the respective 35-45-kD glycosylated AQP-2 bands to corresponding 30-kD deglycosylated AQP-2 protein products. Taken together, these data demonstrate that the AQP-2 protein present in fetal rat kidneys possesses a lower Asn-linked complex carbohydrate content compared with AQP-2 protein present in adult rat kidney.
To determine whether the content or glycoslylation state of AQP-2 is altered in kidneys of PN rats during the initial 14 d of PN life, identical aliquots of kidney membranes from rat pups were immunoblotted as shown in the top panel of Figure 9. These data show that PN rats display a progressive increase in both 35-45-kD glycosylated (4.5-fold increase) and 29-kD nonglycosylated (1.5-fold increase) immunoreactive bands of AQP-2 as a function of increasing PN age. The greater relative increase in the immunoreactive 35-45-kD band compared with the 29-kD AQP-2 band is consistent with an increase in the net content of glycosylated AQP-2 during this interval of PN life.
Immunoblot analyses of AQP-2 and -3 protein expression in PN rat kidneys. Duplicate filters of SDS-PAGE gels containing crude membrane fractions (50 μg/lane) prepared from pooled whole kidneys of five animals at each time point (PN1, 2, 3, 6, 10, and 14 d) were each subjected to immunoblot analyses using either anti-AQP-2 or -3 antisera. (Upper panel) AQP-2 immunoblot: the total AQP-2 immunoreactivity in kidneys from PN1-14 rats increases as a function of increasing PN age when compared with membranes from adult inner medulla (A-IM). Note that both the nonglycosylated 29-kD band of AQP-2 and its glycosylated counterpart (35-45-kD band) increase as a function of PN age. (Lower panel) AQP-3 immunoblot: a duplicate filter of that shown in the upper panel reveals that the AQP-3 bands of approximately 25 and 30-35 kD that are present in all lanes decrease in intensity as a function of increasing PN age. Note the more prominent 25-kD nonglycosylated AQP-3 band in membranes from PN1 kidneys compared with adult (A-IM).
To determine whether AQP-3 exhibited alterations in its protein content and glycosylation similar to that displayed by AQP-2 above, identical immunoblotting analyses were performed on the same samples using anti-AQP-3 antiserum (Fig. 9,bottom panel). Previous work(32) has determined that AQP-3 immunoblots display a 25-27-kD nonglycosylated protein as well as a 30-35-kD glycosylated band in membrane prepared from adult rat kidneys. Comparison of AQP-3 bands present in kidney membranes of PN rats (PN1-3 and PN6) versus adults(A-IM) reveals that a greater proportion of their total AQP-3 immunoreactivity is present as a nonglycosylated 25-kD band compared with its counterpart in adult kidney. These data suggest that AQP-3 may also undergo alterations in its pattern of glycosylation similar to that displayed by AQP-2 in fetal and PN rat kidney.
In contrast to the apparent increase in AQP-2 protein in kidney membranes as shown in Figure 9, top panel, the total AQP-3 content of these same samples appears to decrease (Fig. 9,bottom panel). Although multiple explanations for this decrease are possible (see “Discussion”), the greater proportional growth and development of the renal cortex (lacking significant AQP-3 but possessing AQP-2) compared with the kidney medulla (rich in AQP-2 and -3) may account for this observation.
DISCUSSION
The combination of data contained in this report and recent publications provide important details of the expression, localization, and modulation of AQP-2 and -3 water channel proteins during fetal and PN development in rats. Although previous studies have examined developmental alterations in either AQP-2 and -3 proteins(16) or mRNA expression(19) in the fetal period, investigation of changes in AQP-2 and -3 immediately after birth have been limited to only PN1 as well as intervals greater than PN7(7, 19). Furthermore, analyses of whether alterations in the glycosylation pattern of AQP-2 protein occur in fetal and PN rat kidney in an manner similar to that reported for fetal human kidney(16) have not been performed.
Immunocytochemistry data shown in Figures 1–4 together with previous reports(16, 19, 26) demonstrate that expression of AQP-2 and -3 occur during fetal development in epithelial cells present in large ureteric buds in both rat and human kidneys. The respective apical and basolateral localizations of AQP-2 and -3, respectively provide evidence that epithelial cells of the branching ureteric bud in the fetal period are capable of targeting proteins to apical and basolateral locations in the cell. These markers of epithelial cell polarization suggest that the cells of the developing fetal ureteric bud possess the cellular machinery necessary to allow for vectorial salt and water transport. However, we have not performed ultrastructural analyses to determine whether fetal AQP-2 is present in subapical vesicles in a manner identical to that in adult rats(22).
The combination of immunocytochemistry data (Figs. 1–3)(19) and ribonuclease protection assay data(19) demonstrate that expression of both AQP-2 and -3 are detectable as early as E16 (AQP-2) and E18(AQP-3) in fetal rat kidney where their expression is confined to ureteric bud or collecting duct epithelial cells. Standard Northern analyses reveal expression of a 1.9-kb AQP-2 transcript that is indistinquishable from that present in adult kidney as early E20 (Fig. 5). Analysis of AQP-2 mRNA (Figs. 6 and 7) expression reveal no significant alterations in the kidneys of rat pups during the initial 72 h of PN life. Similar analyses of AQP-2 protein (Fig. 9) show no change until 72 h of PN life. These data extend previous reports of AQP-2 expression in PN rat kidneys(7) and demonstrate only modest increases in AQP-2 occur during the initial 7 d of life. This is then followed by nearly 3-fold increases after an interval of 1-2 mo concomitant with the development of maximal urinary concentrating ability in rats(7).
Vasopressin receptor (V2R) mRNA is present as early as E16 in rat collecting ducts with increasing expression of V2R mRNA in the first PN weeks(10). These data suggest a possible coordinated expression of the V2R and AQP-2 in the developing collecting duct. In adult rats, chronic AVP stimulation (and thus chronic V2R activation) produces increases in AQP-2 expression(22) possibly through a cAMP responsive element present in the promoter region of the AQP-2 gene(33). Furthermore, these data may also provide an explanation for the observations that the fetal kidney may be capable of production of a hypertonic urine during intervals where pharmacologic doses of exogenous AVP are administered(34) or when endogenous AVP levels are increased significantly under stressful conditions such as maternal bleeding, hypoxia or immediately before birth(2, 35). Further increases in AQP-2 protein expression may also be mediated by increases in dietary protein intake that have been reported to modulate AQP-2 expression in adult rats(36).
Previous reports suggest that although expression of AQP-3 protein may be regulated in a manner similar to that of AQP-2 in adult rats(37–39), data from fetal and PN rats show rather modest changes in AQP-3 mRNA that do not correlate with development of maximal urinary concentrating ability(19).
Immunoblotting data shown in Figures 8 and 9 suggest that AQP-2 protein present in fetal and early PN rat kidney exhibits a reduction in its glycosylation state compared with AQP-2 protein present in kidneys from adult rats. Data shown in Figure 9 also suggest that AQP-3 may also display a similar reduction in its glycosylation state in kidneys from early PN animals. These data are similar to those reported for AQP-1 and -2 by Devuyst et al.(16) in human kidney. The significance of these differences in glycosylation ratios is unclear. Studies of AQPs in Xenopus oocytes(40–42) and Madin-Darby canine kidney cells transfected with AQP-2(43) have suggested that mutations altering the Asn-linked glycosylation site do not dramatically alter AQP-2 function or trafficking in response to cAMP. Further studies are needed to elucidate both the functional and structural differences between AQP-2 present in fetal and neonatal kidneys versus adult kidneys.
Comparison of alterations in the immunoreactive AQP-2 and -3 protein contents of crude membranes prepared from the kidneys of rats of various PN ages shows that although AQP-2 immunoreactivity increases, AQP-3 immunreactive protein appears to undergo a net reduction (Fig. 9). Previous reports(19) using ribonuclease protection assays have suggested that both AQP-2 and -3 mRNAs are increased approximately 2- and 5-fold, respectively, over the first 2 wk of PN life. However, the relative abundance of AQP-2 is at least 5-7-fold larger compared with AQP-3 mRNA. These apparently conflicting reports will require more detailed studies of both the recoveries of AQP-2 and -3 from PN rat kidneys as well as a single study correlating AQP-3 mRNA expression with APQ-3 protein levels in PN kidneys of various ages. It is noteworthy in this regard that during this interval of PN life there is differential growth of the renal cortex(containing significant AQP-2 in cortical collecting ducts but not AQP-3) compared with the renal medulla where the bulk of the AQP-3 is expressed. Such differential cortical growth in PN kidney development may have the effect of producing a net reduction in the overall AQP-3 content of crude membranes. Taken together, these data provide further evidence supporting a key role for AQP water channels in both developmental renal physiology as well as maturation of the renal concentrating mechanism.
Abbreviations
- IMCD:
-
inner medullary collecting duct
- P f :
-
water permeability
- E:
-
embryonic
- PN:
-
postnatal
- AQP:
-
aquaporin
References
Robillard JE, Porter CC, Jose PA 1994 Structure and function of the developing kidney. In: Holliday M, Barratt TM, Avner ED (eds) Pediatric Nephrology. Williams & Wilkins, Baltimore, pp 21–39.
Robillard JE, Matson J, Sessions C, Smith F 1979 Developmental aspects of renal tubular reabsorption of water in the lamb fetus. Pediatr Res 13: 1172–1176.
Alexander DP, Nixon DA, Widdas WF, Wohlzogen FX 1958 Gestational variations in the composition of the foetal fluids and foetal urine in the sheep. J Physiol 140: 1–13.
McCance RA, Young WF 1941 The secretion of urine by newborn infants. J Physiol 99: 265–282.
Spitzer A, Schwartz G 1992 The kidney during development. In: Windhager E (ed) Handbook of Physiology: Renal Physiology, Vol 1. Oxford University Press, New York, pp 475–544.
Heller H 1952 The action and fate of vasopressin in newborn and infant rats. J Endocrinol 8: 214–223.
Yasui M, Marples D, Belusa R, Ecklof AC, Celsi G, Nielsen S, Aperia A 1996 Development of urinary concentrating capacity: role of aquaporin-2. Am J Physiol 271:F461–F468.
Skowsky WR, Fisher D 1977 Fetal neurohypophyseal arginine vasopressin and arginine vasotocin in man and sheep. Pediatr Res 11: 627
Schubert F, George J, Rao MB 1981 Vasopressin and oxytocin content of human fetal brain at different stages of gestation. Brain Res 213: 111–117.
Ostrowski N, Young WS, Knepper M, Lolait S 1993 Expression of vasopressin V1a and V2 receptor messenger ribonucleic acid in the liver and kidney of embryonic, developing and adult rats. Endocrinology 133: 1849–1859.
Rajerison R, Butlen D, Jard S 1976 Ontogenic development of antidiuretic hormone receptors in rat kidney: comparison of hormonal binding and adenylate cyclase activation. Mol Cell Endocrinol 4: 271–285.
Schlondorff D, Weber H, Trizna W, Fine L 1978 Vasopressin responsiveness of renal adenylate cyclase in newborn rats and rabbits. Am J Physiol 3:F16–F21.
Harris HW, Zeidel M 1993 Water channels. Curr Opin Nephrol Hypertens 2: 699–707.
Denker BM, Smith BL, Kuhajda FP, Agre P 1988 Identification, purification, and partial characterization of a novel Mr 28,000 integral membrane protein from erythrocytes and renal tubules. J Biol Chem 263: 15634–15642.
Sabolic I, Valenti G, Verbatz JM, Hoek A, Verkman AS, Ausiello D, Brown D 1992 Localization of the CHIP-28 water channel in rat kidney. Am J Physiol 263:C1225–C1233.
Devuyst O, Burrow CR, Smith BL, Agre P, Knepper MA, Wilson PD 1996 Expression of aquaporins-1 and -2 during nephrogenesis and in autosomal dominant polycystic kidney disease. Am J Physiol 271:F169–F183.
Preston GM, Smith BL, Zeidel ML, Mounds JJ, Agre P 1994 Mutations in aquaporin 1 in phenotypically normal humans without functional CHIP water channels. Science 265: 1585–1587.
Smith B, Baumgarten R, Nielsen S, Raben D, Zeidel M, Agre P 1993 Concurrent expression of erythroid and renal aquaporin CHIP and appearance of water channel activity in perinatal rats. J Clin Invest 92: 2035–2041.
Yamamoto T, Sasaki S, Fushimi K, Ishibashi K, Yaoit E, Kawasaki K, Fujinaka H, Marumo F, Kihara I 1996 Expression of AQP family in rat kidneys during development and maturation. Am J Physiol 272:F198–F204.
Fushimi K, Uchida S, Hara Y, Hirata Y, Marumo F, Sasaki S 1993 Cloning and expression of apical membrane water channel of rat kidney collecting tubule N. ature 361: 549–552.
Hayashi M, Sasaki S, Tsuganezawa H, Monkawa T, Kitajima W, Konishi K, Fushimi K, Marumo F, Saruta T 1994 Expression and distribution of aquaporin of collecting duct are regulated by vasopressin V2 receptor in rat kidney. J Clin Invest 4: 1778–1783.
Nielsen S, DiGiovanni S, Christensen E, Knepper M, Harris HW 1993 Cellular and subcellular immunolocalization of vasopressin-regulated water channel in rat kidney. Proc Natl Acad Sci USA 90: 11663–11667.
Sasaki S, Fushimi K, Saito H, Saito F, Uchida S, Ishibashi K, Kuwahara M, Ikeuchi T, Inui K, Nakajima K, Watanabe T, Marumo F 1994 Cloning, characterization and chromosomal mapping of human aquaporin of collecting duct. J Clin Invest 93: 1250–1256.
Deen P, Croes H, van Aubel R, Ginsel L, van Os C 1995 Water channels encoded by mutant aquaporin-2 genes in nephrogenic diabetes insipidus are impaired in their cellular routing. J Clin Invest 95: 2291–2296.
Hayashi M, Sasaki S, Tsuganezawa H, Monkawa T, Kitajima W, Konishi K, Fushimi K, Marumo F, Saruta T 1994 Expression and distribution of aquaporin collecting duct are regulated by vasopressin V2 receptor in rat kidney. J Clin Invest 94: 1778–1783.
Sabolic I, Katsura T, Verbavatz JM, Brown D 1995 The AQP-2 water channel: Effect of vasopressin treatment, microtubule disruption and distribution in neonatal rats. J Membr Biol 143: 165–175.
Ishibashi K, Sasaki S, Fushimi K, Uchida S, Kuwahara M, Saito H, Furukawa T, Nakajima K, Yamaguchi Y, Gojobori T, Marumo F 1994 Molecular cloning and expression of a member of the aquaporin family with permeability to glycerol and urea in addition to water expressed at the basolateral membrane of kidney collecting duct cells. Proc Natl Acad Sci USA 91: 6269–6273.
Christensen BM, Olsson B, Knepper MA, Maunsbach AB, Nielsen S 1997 Cellular and subcellular localization of aquaporin-2 and aquaporin-3 in human kidney. J Am Soc Nephrol 8: 16A
Chattapadhyay N, Baum M, Bai M, Riccardi D, Hebert S, Harris HW, Brown EM 1996 Ontogeny of the extracellular calcium-sensing receptor in rat kidney. Am J Physiol 271:F736–F743.
Jo I, Harris HW, Amendt-Raduege AM, Majewski RR, Hammond TG 1995 Rat kidney papilla contains abundant synaptobrevin protein that participates in the fusion of antidiuretic hormone-regulated water channel-containing endosomes in vitro. Proc Nat Acad Sci USA 92: 1876–1880.
Elder JH, Alexander S 1982 Endo-β-N-acetylglucosaminidase F: endoglycosidase from Flavobacterium menigosepticam that cleaves both high- mannose and complex glycoproteins. Proc Natl Acad Sci USA 79: 4940–4544.
Ecelbarger C, Terris J, Frindt G, Echevarria M, Marples D, Nielsen S, Knepper M 1995 Aquaporin-3 water channel localization and regulation in rat kidney. Am J Physiol 269:F663–F672.
Uchida S, Sasaki S, Fushimi K, Marumo F 1994 Isolation of human aquaporin-CD gene. J Biol Chem 269: 23451–23455.
Robillard, J Weitzman R 1980 Developmental aspects of the fetal renal response exogenous arginine vasopressin. Am J Physiol 238:F407–F414.
Robillard J, Weitzman R, Fisher D, Smith F 1982 Developmental aspects of renal tubular reabsorption of water and fetal renal response to arginine vasopressin. In: Spitzer A (eds) The Kidney during Development: Morphology and Function. Masson, New York, pp 205–214.
Sands JM, Naruse M, Jacobs JD, Wilcox JN, Klein JD 1996 Changes in aquaporin-2 protein contribute to the urine concentrating defect in rats fed a low-protein diet. J Clin Invest 97: 2807–2814.
Terris J, Ecelbarger C, Nielsen S, Knepper M 1996 Long-term regulation of four renal aquaporins in rats. Am J Physiol 271:F414–F422.
Yamamoto T, Sasaki S, Fushimi K, Ishibashi K, Marumo F, Kihara I 1994 Subcellular localization of two types of water channels (AQP-CD and AQP-3) in the collecting duct cells of AVP-stimulated Brattelboro rats. J Am Soc Nephrol 5: 281A
Ishibashi K, Sasaki S, Fushimi K, Yamamoto T, Kuwahara M, Marumo F 1997 Immunolocalization and effect of dehydration on AQP-3, a basolateral water channel of kidney collecting ducts. Am J Physiol 272:F235–F241.
Jung JS, Preston GM, Smith BL, Guggino WP, Agre P 1994 Molecular structure of the water channel through aquaporin CHIP. The hourglass model. J Biol Chem 259: 14648–14654.
Preston GM, Jung JS, Guggino WP, Agre P 1993 The mercurial-sensitive residue at cysteine 189 in the CHIP28 water channel. J Biol Chem 268: 17–20.
Bai L, Fushimi K, Sasaki S, Marumo F 1996 Structure of aquaporin-2 vasopressin water channel. J Biol Chem 271: 5171–5176.
Baumgarten R, Wetzels J, Van Os C, Deen P 1997 Glycosylation of aquaporin-2 is not essential for routing and functioning in mammalian cells. J Am Soc Nephrol 8: 15A
Acknowledgements
The authors thank Dr. D. Ward and J. Crouse for assistance in tissue collection and sample preparation.
Author information
Authors and Affiliations
Additional information
Supported by National Institutes of Health Grants DK-38874 (H.W.H.) and DK-7268 (H.W.H.). M.B received support from a research fellowship grant from the Massachusetts Affiliate American Heart Association.
Rights and permissions
About this article
Cite this article
Baum, M., Ruddy, M., Hosselet, C. et al. The Perinatal Expression of Aquaporin-2 and Aquaporin-3 in Developing Kidney. Pediatr Res 43, 783–790 (1998). https://doi.org/10.1203/00006450-199806000-00011
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1203/00006450-199806000-00011
This article is cited by
-
Aquaporin1–3 expression in normal and hydronephrotic kidneys in the human fetus
Pediatric Research (2019)
-
Influence of sex on aquaporin1–4 and vasopressin V2 receptor expression in the pig kidney during development
Pediatric Research (2016)
-
Ontogeny of the mammalian kidney: expression of aquaporins 1, 2, 3, and 4
World Journal of Pediatrics (2014)