Abstract
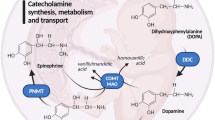
Summary: Phenylethanolamine N-methyl transferase (PNMT), the terminal enzyme in epinephrine biosynthesis, was identified in fetal and newborn ovine lung. The ovine lung PNMT demonstrated the appropriate substrate specificity and affinity (Km, 9 × 10-6 M). Although some homology between adrenal and lung PNMT was observed on polyacrylamide gel electrophoresis, the lung PNMT differed in its migration on ion exchange chromatography and was not inhibited by pharmacologic inhibitors active against the adrenal enzyme. Activity increased from a mean of 132 pmole/(mg protein · h) × 10-3 at 0 days gestation to 326 pmole/mg protein · h) × 10-3 in newborn animals between 1–4 days of age (r = 0.571, P < 0.05). The levels of N-methylating activity in extraadrenal tissues were relatively low; lesser but significant N-methylating activities were demonstrated in brown adipose tissue and myometrial tissues. Adrenal gland activity was 1000-fold greater than lung activity expressed on a per mg protein basis and 250-fold greater expressed per mg wet weight.
Similar content being viewed by others
Article PDF
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Padbury, J., Lam, R., Hobel, C. et al. Identification and Partial Purification of Phenylethanolamine N-Methyl Transferase in the Developing Ovine Lung. Pediatr Res 17, 362–367 (1983). https://doi.org/10.1203/00006450-198305000-00011
Issue Date:
DOI: https://doi.org/10.1203/00006450-198305000-00011