Abstract
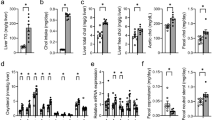
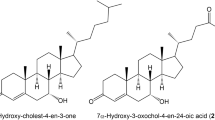
Summary: C6-C10-dicarboxylic and C6-C10-ω-1-hydroxy monocarboxylic acids were measured in postmitochondrial (10,000 g) fractions of rat liver after incubation with hexanoic, octanoic, and decanoic acids. In livers both from fed and starved rats, the proportion of decanoic acid converted to sebacic acid was high (approximately 25%) with only minor accumulation of the intermediate 10-hydroxy decanoic acid (1–2%). The conversion of octanoic and hexanoic acids to suberic and adipic acids, respectively, was low (<1%). The intermediate 8-hydroxy octanoic and 6-hydroxy hexanoic acids were also accumulated in very small amounts (<1%). It was concluded that cytochrome-P-450-mediated ω-hydroxylation was of decisive importance for the production rate of the dicarboxylic acids. Analysis of kinetic parameters of human and rat liver microsomal ω- and ω-1-hydroxylation of hexanoic, octanoic, decanoic, and dodecanoic acids gave the following results: in rats, the apparent Km values for the ω-hydroxylation for dodecanoic and decanoic acids are low, i.e., 171 and 3.1 μmole/liter, respectively, whereas they are high for octanoic and hexanoic acids (8211 and 8822 μmole/liter, respectively). In two different humans, the corresponding Km values for dodecanoic, decanoic, octanoic, and hexanoic acids are 3.6–186, 522–247, 4861–3892, and 6825–10400 μmole/liter, respectively. Based on these results, it is argued that adipic and suberic acids found in urine from rats and humans with acyl-CoA dehydrogenation deficiencies are not biosynthesized by direct ω-oxidation of hexanoic and octanoic acids, but most probably by means of β-oxidation of sebacic and dodecanedioic acids, produced by direct ω-oxidation.
The affinities of the ω-1-hydroxylation systems for the four monocarboxylic acids is similar in rats and humans. The apparent Km values for dodecanoic acid in rats and in the two human livers are low, i.e., 139 and 92–131 μmole/liter, respectively. The Km values for decanoic, octanoic, and hexanoic acids are all high in both rats and humans, i.e., in the range from 638 μmole/liter (for decanoic in one of the human livers) to more than 20,000 μmole/liter (for hexanoic acid in both species). It is argued that 5-hydroxy hexanoic, 7-hydroxy octanoic, and 9-hydroxy decanoic acids found in some patients with acyl-CoA dehydrogenation deficiencies are produced by β-oxidation of 11-hydroxy dodecanoic acid, which may be produced directly by ω-1-hydroxylationof dodecanoic acid.
Similar content being viewed by others
Article PDF
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Gregersen, N., Mortensen, P. & Kølvraa, S. On the Biologic Origin of C6-C10-Dicarboxylic and C6-C10-ω-1-Hydroxy Monocarboxylic Acids in Human and Rat with Acyl-CoA Dehydrogenation Deficiencies: in Vitro Studies on the ω- and ω-1-Oxidation of Medium-Chain (C6-C12) Fatty Acids in Human and Rat Liver. Pediatr Res 17, 828–834 (1983). https://doi.org/10.1203/00006450-198310000-00013
Issue Date:
DOI: https://doi.org/10.1203/00006450-198310000-00013
This article is cited by
-
Carnitine esters in metabolic disease
European Journal of Pediatrics (1994)
-
The inborn errors of mitochondrial fatty acid oxidation
Journal of Inherited Metabolic Disease (1987)
-
Fatty Acyl‐CoA dehydrogenase deficiency: Enzyme measurement and studies on alternative metabolism
Journal of Inherited Metabolic Disease (1984)