Abstract
Extract: The change in levels of growth hormone in serum (SGH) following insulin-induced hypoglycemia was evaluated in 134 prepubertal children with growth retardation and 10 control subjects with normal growth patterns by radioimmunoassay, utilizing 131I-HGH and rabbit antiserum to human growth hormone. Mean maximum growth hormone concentration (mμg/ml) at any time during the test was:
Among factors found to affect the SGH response to insulin-induced hypoglycemia were: a) elevated fasting concentration of SGH which appeared to alter the responsiveness to stimulation; and b) age.
The mean maximum SGH concentration of the control subjects following insulin-induced hypoglycemia was 12.4 mμg/ml.
In 52/53 patients with hypopituitarism, the SGH concentration was 1 mμg or less at rest, with no increase or an increase to a maximum of 2.5 mμg/ml following hypoglycemia. One patient (CL) had a fasting serum growth hormone (FSCH) concentration of 3.4 mμg/ml with a rise to 5 mμg/ml at 60 minutes, as indicated in tables II and IIa.
Clinical data on 20 patients with constitutional shortness of stature are presented in table III. The mean maximum GH concentration following insulin-induced hypoglycemia of this group was 12.5 mμg/ml, a value comparable to that obtained in the control subjects (table IIIa).
Clinical data, including growth rate, of patients with primordial dwarfism are presented in table IV. The mean maximum SGH concentration following hypoglycemia of the 22 patients in this grop was 12.1 mμg/ml (table IVa). This response was not significantly different from that observed in the control group.
Nine patients with XO gonadal dysgenesis had a mean maximum SGH response to hypoglycemia of 13.4 mμg/ml, as indicated in tables VI and VIa.
Five children with delayed adolescence had a mean maximum SGH response of 11.8 mμg/ml, as shown in tables VI and VIa.
Four of five infants with maternal deprivation included in this study showed evidence of increased insulin sensitivity, but the mean maximum SGH response was not significantly different from that of the control group. Clinical data on nine patients with psychosocial drawfism are presented in table VI. The nine children in this group had a mean maximal rise of SGH concentration to 10.9 mμg/ml. Two of the children had an abnormal SGH response with concentrations of 1 mμg/ml in the fasting specimen and no rise following hypoglycemia. RS, who was retested after a hospitalization period of two months, had a maximum SGH rise to 7.4 mμg/ml (table VIa).
Clinical data on 11 patients with a variety of diseases associated with their growth retardation are presented in table VII. Included in this group are two males, NF and MB, with blunted responses to insulin-induced hypoglycemia and in whom the diagnosis of partial GH deficiency cannot be excluded.
As indicated on figures 1a and 1b, the mean maximum SGH response to insulin-induced hypoglycemia in children with growth retardation was not significantly different from the response observed in the control group. There was, however, a highly significant difference in the response of the control group when compared to the group of children with hypopituitarism.
In the presence of an elevated SGH concentration, eight of the eighteen subjects showed a decrease in FGH concentration or no response following insulin-induced hypoglycemia. This was observed in four control subjects and four children with growth retardation. The level of FSGH was significantly higher in children less than four years of age, but there was no significant correlation of the level of FSGH with age in children over four years, or according to sex. In this study, there was no correlation demonstrated between a decrease of 40% or greater in blood glucose and the maximum GH response achieved.
The response to therapy with human pituitary growth hormone (HGH) was evaluated in 8 children with growth retardation who had a mean maximum SGH concentration of 12.0 mμg/ml. In six of the patients, none of whom had the clinical features of pituitary dwarfism, there was no significant change in the rate of growth during the period of administration of HGH. Three of the six patients subsequently had low levels of antibodies to HGH and three had no detectable antibodies. ER, a primordial dwarf, had an acceleration in his growth rate during the first period of treatment with HGH which was not sustained. An increase in growth rate to 6.9cm/yr was observed in the second patient (NF) suspected of having mild or partial GH deficiency. In contrast, 20 patients with SGH levels of less than 2 mμg/ml had a growth rate of 9 to 15 cm/yr on treatment with HGH.
No evidence of a GH deficiency or a defect in the responsiveness of the hypothalamic GH-releasing mechanism was demonstrated in children with growth disorders, including constitutional shortness of stature, primordial dwarfism and gonadal dysgenesis. On the basis of our data, a FSGH of 7 mμg/ml or an increase to 7 mμg/ml or more folowing insulin-induced hypoglycemia has a high probability of association with normal pituitary acidophile function. A rigid definition of the normal range of SGH respons to hypoglycemia cannot be established because, in a small proportion of instances, there is overlap between the normal subject and some GH-deficient patients.
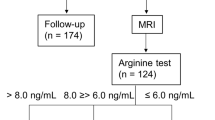
Problems in the interpretation of the rsponse to insulin-induced hypoglycemia have been observed. In children who have a borderline rise of SGH of 3 to 5 mμg/ml, the response may be indicative of GH deficiency. In a few children, however, individual differences in responsiveness to various stimuli have been observed. It is suggested that GH stimulation tests with arginine infusion or vasopressin administration be used in a child who exhibits a blunted GH response to hypoglycemia. In some children who have an apparently normal serum ‘immunoreactive’ GH response to a provocative test despite physical stigmata of hypopituitarism, short-term therapy with HGH may be necessary to establish the diagnosis of GH insufficiency.
Speculation: It has been observed that the changes in SGH induced by hypoglycemia are less in children than in adults. This difference between children and adults may be related to the concentration in serum of testosterone or estrogen, an altered sensitivity of the hypothalamus to stimuli in children, or to age related variations in the secretory rate of the pituitary. SGH response to insulin-induced hypoglycemia has been valuable in distinguishing children with GH deficiency, especially when this occurs as an isolated defect, from children with other forms of growth retardation. It has not been useful in identifying the etiology of growth retardation from other causes.
Similar content being viewed by others
Article PDF
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kaplan, S., Abrams, C., Bell, J. et al. Growth and Growth Hormone: I. changes in Serum Level of Growth Hormone Following Hypoglycemia in 134 children with Growth Rctardation. Pediatr Res 2, 43–63 (1968). https://doi.org/10.1203/00006450-196801000-00005
Issue Date:
DOI: https://doi.org/10.1203/00006450-196801000-00005
Keywords
This article is cited by
-
Die Reversibilität des idiopathischen, isolierten Wachstumshormonmangels
Journal für Klinische Endokrinologie und Stoffwechsel (2022)
-
Is the fear from insulin tolerance test in the evaluation of short stature justified?
European Journal of Pediatrics (2022)
-
Influence of biochemical diagnosis of growth hormone deficiency on replacement therapy response and retesting results at adult height
Scientific Reports (2021)
-
Growth hormone deficiency in children
Pituitary (2008)
-
Growth hormone treatment of Turner syndrome patients with insufficient growth hormone response to pharmacological stimulation tests
European Journal of Pediatrics (1991)