Abstract
In recent years, significant research effort has focused on creating supramolecular polymers that can be attained by specific host–guest interactions of the repeating units. During the supramolecular polymerization process, molecular recognition events, which are predetermined by the molecular building blocks, are highly selective and directional for defining the size, direction and dimension of the resulting supramolecular polymers. The diversity of the supramolecular building blocks ranges from small aromatic units to macrocycles. Recently, the interplay of supramolecular and polymer chemistry has led to the creation of novel supramolecular materials, which display fascinating functions such as self-healing, stimuli-responsiveness and rubber-like elastomeric properties. Supramolecular cross-linking and supramolecular block copolymerization are the methods that have been used to install fascinating and functional moieties onto polymer backbones. Currently, the development of practical supramolecular polymeric materials is an ongoing challenge for supramolecular chemists. This review will focus on the recent developments in supramolecular polymers composed of discrete repeating units, as well as novel supramolecular materials produced by the interplay of supramolecular and polymer chemistry.
Similar content being viewed by others
Introduction
Synthetic polymers with tailored properties offer a wide range of applications for plastic products and advanced photonic and electronic functional materials. Polymers with a high melt viscosity are often tedious to process, which restricts the number of applications for advanced functional materials. Therefore, the facile fabrication and construction of conventional polymers remain in demand. Although improved methods for the synthesis of bulk materials have been actively studied, supramolecular polymerizations have emerged as an attractive alternative for the synthetic method of polymer materials. Supramolecular polymers hold promise as a unique class of innovative materials because the attractive features of conventional polymers are integrated with reversibility. Structural and dynamic properties of supramolecular polymers, including the DP, chain lifetime and conformational flexibility, are influenced by external stimuli; therefore, these result in supramolecular ‘smart’ materials, which offer adaptivity that leads to easy fabrication, stimuli responsiveness and self-healing at the forefront of science and engineering.1
The term supramolecular polymer2, 3 is broadly defined as any type of an assembly formed from one or more molecular components via reversible bonds; therefore, monomeric and polymeric states are in equilibrium over the relevant experimental timescale. One could imagine that liquid crystals, molecular crystals, micelles, vesicles, lipid bilayers and others are in the class of supramolecular polymers. Liquid and molecular crystals lose polymeric structures and properties in the isotropic state and in solution. Micelles, vesicles and lipid bilayers are driven by the hydrophobic effect: amphiphilic molecular constituents interact with each other to coalesce through non-specific and non-directional intermolecular interactions. However, these molecular assemblies are not commonly considered to be in the class of supramolecular polymers, even though they are constitutionally dynamic and composed of repeating monomers. Meijer et al.4 proposed the most limiting definition regarding supramolecular polymers as polymeric arrays of monomeric units that are brought together by reversible and directional supramolecular interactions and exhibit chain-like behaviors in dilute solutions, concentrated solutions and in bulk. A monomeric unit has two or more interaction sites that find their complements on other units, which generate strong supramolecular interactions through molecular recognition. The predetermined supramolecular interactions define the size, direction and dimension of supramolecular polymers. A variety of noncovalent interactions (for example, hydrogen bonding, van der Waals, coulombic, solvophobic, aromatic stacking, donor–acceptor and ionic interactions) must be incorporated to generate highly specific, directional and strong supramolecular interactions.5
Figure 1 displays the schematic representation of a supramolecular polymerization. Initially, molecular associations between the monomers bearing complementary binding sites form oligomers. This process is thermodynamically reversible, and an average DP can be estimated based on the association constant of each connection site. Further growth of the oligomers leads to sizable polymers that are entangled, which form three-dimensional networked structures. The interchain interactions among supramolecular polymeric chains may become prominent. In such a case, supramolecular polymers may possibly show physical properties similar to conventional polymers, even though supramolecular bonds are in equilibrium.6
The synthetic strategy of linear supramolecular polymers could be classified into four major types with respect to the structural motif that leads to assembly (Figure 2). Figure 2a shows the homoditopic monomer that possesses two A-type recognition sites. A self-complementary dimer A–A is formed at each terminus, and the iterative dimerization results in supramolecular homopolymers. Another type of homopolymerization is accomplished using a specific complementary heterodimerization module B–C that connects each monomer in a head-to-tail manner, resulting in a supramolecular homopolymer (Figure 2b). The heterodimerization module B–C can also be useful for the synthesis of AB-type alternating block copolymers. The homoditopic monomers B–B and C–C can polymerize via the intermolecular heterodimerization in an alternating manner (Figure 2c). It may appear that an aromatic-stacking interaction might be too weak and less directional to drive supramolecular polymerization. However, the introduction of directing groups on the periphery of an aromatic core D can enhance the formation of supramolecular polymers (Figure 2d). Solvophobic, hydrogen bonding, dipole–dipole and donor–acceptor interactions introduced to the periphery of aromatic cores have previously led to the construction of supramolecular stacks. Key reviews dealing with the construction of molecular stacks have been previously published.7, 8, 9, 10, 11, 12, 13
Schematic representation of the formation of linear main-chain supramolecular polymers from complementary monomers. Supramolecular homopolymers (a) from homoditopic monomers and (b) from heteroditopic monomers. (c) Supramolecular alternating block copolymer from two different homoditopic monomers. (d) Supramolecular polymer driven by molecular-stacking interactions.
Recent trends in this field are directed toward the more sophisticated supramolecular polymers in terms of material properties as well as structures. The focus of this review article is on main-chain supramolecular polymers formed via molecular-recognition-directed self-assembly of small monomers and selected recent advances of polymer-based supramolecular polymers.
Hydrogen-bonded supramolecular polymers
Supramolecular polymerizations directed by complementary hydrogen bonding
Hydrogen bonds are a well-known noncovalent interaction.14 Their directionality and versatility have been providing a leading place in supramolecular chemistry. The major drawback of hydrogen bonds is the weak interaction energy of a single donor–acceptor hydrogen bond, particularly in polar solvents. Multiple hydrogen bonds with a specific donor–acceptor arrangement per binding unit can greatly enhance the stability and directionality of a hydrogen-bonding interaction in a supramolecular structure. Therefore, great efforts have been devoted toward developing complementary multiple hydrogen-bonding units.
Lehn et al.15, 16 reported the pioneering idea of using linear-chain noncovalent polymers driven by multiple hydrogen-bonding interactions as a starting point of supramolecular polymer chemistry for developing functional materials (Figure 3). Two homoditopic monomers 1 and 2 were developed: 2 was composed of two diaminopyridines connected by a tartrate linker, and two uracil derivatives were used in 1. Complementary triple hydrogen bonds are formed between the diaminopyridine and uracil moieties. The polymerization between 1 and 2 produced hydrogen-bonded supramolecular polymers with liquid crystallinity over a wide range of temperatures, although they were not liquid crystalline compounds. Additionally, the molecular-recognition-directed polymerization and subsequent self-organization resulted in helical supramolecular entities through the translation of the molecular chirality into supramolecular level at nanometric dimension.17
Their subsequent research demonstrated hydrogen-bonded supramolecular polymers of 3 and 4 in solution (Figure 3).18 The diaminopyridine-substituted isophthalimide and cyanuric acid moieties form complementary six hydrogen bonds with a large association constant (Ka>104 l mol−1) in chloroform. As a solution of 3 and 4 was cooled, the amide protons shifted downfield in the 1H NMR, which is indicative of supramolecular polymerization via hydrogen-bonding interactions. In a hydrocarbon solvent, a stoichiometric mixture of 3 and 4 resulted in a highly viscous solution due to the formation of long, entangled fibers. Observation of the fibers with an electron microscope provided evidence for the formation of helical fibers.
Ghadiri et al.19 reported the synthesis of peptide nanotubes using stacked cyclic peptides composed of alternating D- and L-amino acids (Figure 4). Cyclic peptide 5 assembled in membranes to form trans-membrane channels. The hollow nanotubes with a diameter of approximately 7.5 Å, were retained in a lipid bilayer, and displayed transport activity for potassium and sodium cations. Expanded diameters can be accomplished using larger cyclic peptide oligomers. For example, the cyclic peptide, cyclo[Gln-(D-Leu-Trp)4-D-Leu], produced a tubular ensemble bearing a 10 Å internal diameter. When this tubular ensemble was placed in a lipid bilayer, the channel mediated the transport of glucose.20 Further extension of the ring size can be achieved using a 12-residue cyclic peptide. The nanotube ensembles formed with a uniform 13 Å pore diameter. Electron diffraction analysis revealed that the peptide formed a tight network of hydrogen-bonded β-sheet-type structures, and a hexagonal body-centered packing of nanotubes was confirmed.21
Meijer et al.22, 23, 24 developed a complementary hydrogen-bonding motif. Ureidopyrimidone (UPy) formed a self-complementary dimer via quadruple hydrogen-bonding (DDAA-AADD) with a remarkably strong self-association constant (Ka>107 l mol–1). Homoditopic bifunctional molecule 6 polymerized in chloroform and showed a high DP (Figure 5). The supramolecular polymers produced a highly viscous solution and behaved like a conventional polymer. However, the solution properties of the supramolecular polymers were dependent on the concentration and temperature because the polymers were constitutionally dynamic due to the reversible nature of hydrogen-bonding interactions.
Hydrogen-bonding-mediated supramolecular polymerization has been applied to the synthesis of white-light-emitting copolymers of π-conjugated fluorescent monomers.25 Three chromophores difunctionalized with UPy at both ends (a blue-emitting oligofluorenone, a green-emitting oligo(phenylene-vinylene) and a red-emitting perylene bisimide) were synthesized. The molecules assembled in solution and in the bulk to form hydrogen-bonded supramolecular random copolymers, which exhibited energy transfer upon excitation of the oligofluorene energy donor. A white-emissive supramolecular copolymer was created in solution and in the bulk when a specific chromophore ratio was used. The supramolecular copolymers were also demonstrated LED applications.
Another example of a quadruple hydrogen-bonded structure was based on a ureidotriazine (Figure 5).24 The self-complementary dimeric structure of the ureidotriazine moieties of 7 were stable even in an aqueous solution. The aromatic core of 7 stacked to form the polymeric structure while the hydrophilic chains are placed on the periphery, and the hydrogen-bonding pair enjoyed the hydrophobic environment. Interestingly, the peripheral chiral side chains directed the backbone helicity of the stacked polymeric structures.
Rebek Jr et al.26 showed a remarkable example of supramolecular polymers by multiple hydrogen bonding of a calix[4]arene bearing four urea groups on the upper rim (Figure 6). The tetra-urea-substituted calix[4]arene assembles to form a dimeric capsule that is capable of binding small molecules by inclusion. The self-assembly of bifunctional molecule 8 resulted in hydrogen-bonded polymeric calixarenes, which are called ‘polycaps’. A small guest complexation into the capsule greatly stabilized the dimeric form, which enhanced the polymerization of the assembly. Although the hydrogen bonds are weak enough to melt away on relevant timescales, the supramolecular polymeric chains maintained measurable and meaningful mechanical integrity even in solution. In fact, the ‘polycaps’ produced stable fibers that gelled and displayed an elastic component in rheological experiments.27
The hydrogen-bonding network formed between cyanuric acid and melamine is capable of generating infinite sheets in the solid state. However, crowding of the periphery directs the assembly into a discrete cyclic 3:3 rosette in solution.28, 29 Reinhoudt et al.30 and Whitesides et al.31 independently developed supramolecular polymers using the cyanuric acid–melamine pair (Figure 7). A 1:1 mixture of cyanuric acid 9 and melamine 10 formed a hexameric rosette structure via complementary hydrogen-bonding pairs. Subsequent supramolecular polymerization produced nanorod-like architectures. The spatial distance between the two cyanurate units was slightly different from the distance between the two melamine units. The mismatched distance disrupted the formation of the discrete 3:3 parallel rosette-like assembly and resulted in polymeric entities. Dicyanurate 9 and dimelamine 10 assembled in solution and produced a viscous solution, which implies that supramolecular polymers are formed. The rod-like nanostructures of the supramolecular polymers were directly observed by atomic force microscopy (AFM) and transmission electron microscopy (TEM) measurements.
Oligonucleotide base pairing has been applied to the synthesis of reversible supramolecular polymers. Craig's elegant approach successfully generates materials with reversible polymeric properties. Two oligonucleotide sequences are covalently connected with a synthetic linker. Duplex formation leads to a linear, supramolecular polymeric assembly through reversible base pairing.32, 33 For instance, (5′→3′)GGTATACC-X-GCTTAAGC polymerized through duplex formation. The specific viscosity was concentration dependent, and the scale exponent (3.3) was in good agreement with the theoretical expectation. The average molecular weights (approximately 200 kDa) were determined by static multiangle and dynamic light scattering (DLS) studies. In addition, the supramolecular polymers had semi-rigid rod structures with lengths of approximately 50 nm.
A strong dipolar molecule, for example, merocyanine, usually adopts an antiparallel orientation in supramolecular ensemble owing to strong dipole–dipole interaction.34, 35 Wüthner et al.36 demonstrated a unique supramolecular approach to drive the head-to-tail parallel orientation of an anisotropic dipolar molecule with an assistance of hydrogen bonding. A Hamilton receptor was installed on the merocyanine core (Figure 8). Merocyanine dye 11 assembled in a head-to-tail manner via hydrogen-bonding-driven host–guest interaction, which produced supramolecular polymers with a linear array of the molecular dipoles. The broad distribution of particle sizes ranging from 13 to 1606 nm, as determined by DLS measurements, indicated that sizable polymeric aggregates formed. In addition, the supramolecular polymer gelled in a chloroform-hexane solution. The gel phase showed a remarkably bluish-white fluorescence emission.
Main-chain supramolecular block copolymers
Main-chain supramolecular block copolymers have received significant attention for their regulable and processable material properties.37 The end-group modification of a telechelic poly(ethylene/butylene) copolymer with ureido-pyrimidone (UPy) resulted in a macroscopically visible change in material properties (Figure 9).38 The viscous liquid polymer became an elastic solid as a result of the quadruple hydrogen bonds that linked the individual polymer chains, increasing the virtual molecular weight. In this reversible polymer, breaking–recombination of the chain is an additional mode of stress relaxation; therefore, the temperature dependency of the viscoelastic properties was strongly enhanced. The solution viscosity of the polymer was pH responsive. The addition of a small amount of trifluoroacetic acid (TFA) reduced the solution viscosity because the hydrogen bonds were disrupted by TFA, which caused the main chain breaking that decreased the virtual molecular weight of the polymers.
Binder et al.39 demonstrated supramolecular block copolymerization using telechelic poly(isobutylene)s (PIBs) functionalized with thymine or diaminopyrimidine-substituted isophthalamide (Hamilton receptor) and poly(etherketone)s (PEKs) functionalized with triazine or barbituric acid at both chain ends (Figure 9). The complementary hydrogen-bonding interactions connected two incompatible polymers to generate multiblock copolymers. The thymine–triazine hydrogen bonding is weaker than that of the barbituric acid–Hamilton receptor. The microphase-separated state was stable up to the Tg of the PEK, but the stronger hydrogen-bonding pair (barbituric acid–Hamilton receptor) resulted in a higher phase-breaking transition temperature. Park and Zimmerman40 developed a complementary quadruple hydrogen-bonding pair composed of ureido-guanoshine (UG) and 2,7-diamino-1,8-naphthyridine (DAN) (Figure 9). DAN and UG were introduced as the termini of poly(butyl methacrylate) (PBMA) and poly(ethylene glycol) (PEG), respectively. The hydrogen-bonded pair of UG and DAN generated an association constant greater than 107 l mol–1. Solution viscometry of the polymers was informative for investigating the formation of supramolecular block copolymers. The specific viscosity of the mixture changed as a function of the ratio of components. For this case, the maximum viscosity was obtained at a ratio of 1:1, which indicated that the complementary hydrogen-bonding pair had a key role in producing supramolecular multiblock copolymers.
ABC-type block copolymers via supramolecular assembly has been synthesized by Weck et al.41, 42 The central heterotelechelic homopolymer contained two orthogonal recognition units, which promoted the stepwise self-assembly of complementary homopolymers. This methodology is particularly versatile for the synthesis of a variety of block copolymers.
Hydrogen-bonded supramolecular polymers are intriguing because of their unique properties and architectures. Currently, hydrogen-bonding interactions are recognized as a reliable tool for the synthesis of supramolecular polymers, as well as supramolecular functional materials.
Supramolecular polymers via molecular recognition
Host–guest supramolecular polymers
The great majority of supramolecular polymers developed thus far use hydrogen-bonding and coordination interactions. Host–guest interaction-driven supramolecular polymerizations have been less employed, although a vast number of host–guest complexes are known. Supramolecular polymerization requires host–guest interactions with very high affinities to obtain an appreciable DP. This requirement limits the number of possible variations of host–guest interactions that can be employed for supramolecular polymerizations. Crown ethers, cyclodextrins (CDs), cucurbit[n]urils, calix[n]arenes and others are well recognized as host molecules with very high affinities for guest molecules and provide useful supramolecular structures for polymerization.
A crown ether is the most famous host molecule, which captures cationic guests through cation–dipole interaction with large stabilization energy.43, 44 Cram et al.45 originally suggested the formation of polymeric species via host–guest complexation of a bis(crown ether) and a α,ω-diammonium salt in 1977. Shinkai et al.46 successfully introduced a photoresponsive feature into the supramolecular polymeric aggregates, which consisted of photo-responsive bis(crown ethers) and α,ω-diammonium salts. An azobenzene unit was incorporated between the two crown ethers. Trans-to-cis isomerization of the azobenzene unit led to large structural changes. The trans form of the bis(crown ether) strongly binds the α,ω-diammonium salt to produce polymeric aggregates with a DP of 20. The polymeric aggregates break up upon isomerization to the cis form.
Gibson et al.47, 48, 49 developed heteroditopic monomer 12, consisting of diphenylene-32-crown-10 and paraquat moieties, which assemble in a head-to-tail manner (Figure 10). The polymer formation was clearly evidenced by mass spectrometry, and a high viscosity of the polymer solutions in organic solvents is characteristic of linear polymeric aggregates. In addition, fibers of the supramolecular polymers were drawn from concentrated solutions. As a polymeric structure of high molecular weight is required for fiber formation, the supramolecular polymer of 12 has characteristics of a conventional polymer, even though the repeating units are connected by noncovalent bonds. This strategy has been extended to enable self-sorting organization of supramolecular alternating copolymers.50 Two heteroditopic host–guest pairs 13 and 14 were designed with each monomer composed of a guest moiety that is complementary to the host moiety of the other. The highly orthogonal host–guest interactions cause perfect self-sorting of the host–guest pairs. As a result, alternating copolymerization is established by the two heteroditopic monomers.
Cucurbit[n]urils (CB[n]) provide a hollow cavity51 in which π–π stacking and charge-transfer complexes of small aromatic donor–acceptor pairs are highly stabilized with high association constants in an aqueous environment.52 This host-stabilized charge transfer interaction is sufficient to generate supramolecular polymers (Figure 11).53 The synthesis of an amphiphilic block copolymer has been produced by the supramolecular complexation between polymers 15 and 16 with the assistance of CB[8].54 The charge transfer interaction between the respective ends of 15 and 16 was weak in water and the sizes were less than 5 nm, as determined using DLS. Even in the presence of CB[8], the host-assisted dimerization of 16 was not detected. Upon mixing 15, 16, and CB[8] in solution, the formation of a ternary complex was evidenced by the increased size of 244 nm. CB[8] surrounded the CT complex of the respective ends to facilitate the block copolymerization between 15 and 16.
The supramolecular polymerization of multifunctional monomer 17 via host-stabilized charge transfer interaction has been reported (Figure 11).55 The supramolecular polymerization of 17 occurred with the aid of CB[8] to form stable, deep-purple hydrogels. When the polymerization inhibitor, methylviologen, was added, the gel quickly collapsed, which suggested that supramolecular polymer networks were crucial for gelation. In fact, well-developed supramolecular gel networks were observed by scanning electron microscopy.
CDs are well-known host molecules that encapsulate a variety of guests into their cavity through hydrophobic interactions. This molecular recognition event is valuable for the synthesis of supramolecular polymers. Li and McGown56 demonstrated that β- and γ-CDs encapsulated diphenylhexatriene to form nanotube aggregates that comprised approximately 20 β-CDs or 20∼35 γ-CDs. Two phenyl groups of the diphenylhexatriene fit into the cavity of β-CD, whereas γ-CD encapsulated three phenyl groups. Each phenyl group was bound to a different CD cavity to connect two CDs. The iterative complexation resulted in supramolecular polymeric aggregates.
Harada et al.57 made a significant contribution to the field of the supramolecular chemistry of CDs. The adamantyl groups of homoditopic guests 19a–c were encapsulated in the hydrophobic cavity of homoditopic β-CD host 18 in water (Figure 12).58 Extended linker lengths between the two adamantyl groups had an unfavorable influence on the supramolecular polymer in an aqueous environment. The host–guest complexation between 18 and 19a resulted in supramolecular polymers with a molecular weight greater than 90 000 g mol–1, as obtained by vapor pressure osmometry analysis. The supramolecular assemblies formed between 18 and 19b,c had relatively smaller molecular weights (Mn=10 000∼15 000), which were independent of the concentration. No free adamantyl groups were detected in solution during a 1H NMR study, which indicated that 19b,c formed cyclic oligomers with 18 owing to the flexible linker units. The direct observation of the cyclic oligomers was accomplished using AFM.
An α-CD encapsulates small aromatic guests, including benzoic acid, nitrophenol and cinnamic acid. 6-Cinnamoyl-α-CD is a heteroditopic monomer that assembles to form supramolecular polymers.59, 60 Interestingly, the supramolecular polymer settled in helical superstructures, which was confirmed by circular dichroism spectroscopy.
The calix[n]arene family is probably the most recognized platform in the field of supramolecular chemistry. Gutsche61, 62, 63 published three key books on the topic in depth, which covered the synthesis, characterization and properties, as well as the conformation, reactions and host–guest chemistry of these unique molecules. For the small calix[n]arenes (n=4 or 5), the cyclic hydrogen bonds of the phenolic hydroxyl groups on the ‘lower rim’ produce a highly symmetric cone cavity in which a variety of complementary guest molecules can be encapsulated through weak noncovalent interaction such as cation/π, CH/π, aromatic stacking and van der Waals interactions.
The cation/π interaction-driven supramolecular polymerization of heteroditopic monomer 20 was demonstrated by Delcanale et al.64, 65 (Figure 13). The cavity of the phosphonate cavitand 20 encapsulated the positively charged pyridinium cation moiety with a very high association constant (Ka=107∼109 l mol–1). The supramolecular polymers of 20 formed in chloroform. The polymerization behavior was investigated using static light scattering. The weight-average molecular weight (Mw) of the polymer reached 26 300 g mol–1, which corresponded to an average DP of 18. Additionally, the supramolecular polymerization was turned ‘on’ and ‘off’ by acid/base treatment, respectively, in the presence of a competitive guest. The competitive guest complexation of N-butylmethyl ammonium salt into the cavity of 20 caused the complete disassembly of the supramolecular polymer, while the addition of a strong base, DBU, quenched the competitive ammonium salt to restore the supramolecular polymers.
A fully alkylated calix[5]arene encapsulated n-BuN+H3 with a large association constant (log Ka=4.7∼6.5).66 Parisi et al.67, 69 and Cohen et al.68 used the strong intermolecular interaction for a pH-responsive supramolecular polymerization (Figure 13). The heteroditopic calix[5]arene 21 bearing an amine group on the lower rim was synthesized. The self-assembly of calix[5]arene 21 into supramolecular oligomers through intermolecular iterative inclusion events was readily triggered by contact with acidic solutions and was reversed to the amino monomer precursor by treatment with a base. The supramolecular polymerizations were carefully studied using the DOSY technique. The number-averaged DP was dependent on the concentration of 21•H+ and the type of counter ion. Only the polymeric aggregates of 21•HPic formed. The tight contact ion-pair between the ammonium and the chloride probably reduced the intermolecular association, while the picrate anion probably had loose ion-pairing, which allowed the polymeric aggregates to gain a more attractive intermolecular interaction. DLS experiments supported the formation of the sizable supramolecular polymers in solution. Scanning electron microscopy provided clear evidence for the formation of the fibrillar networks of the supramolecular polymers.
Recently, Ogoshi et al.70 introduced a new class of the calix[5]arene family, which was named ‘pillarcalix[5]arenes’. Huang et al. reported that pillarcalix[5]arenes bearing a octyl tail assembled via CH/π interaction to form a supramolecular polymer in organic solutions (Figure 13).71 The crystal structure of the polymer indicated that close contact between the alkyl tail and the inner aromatic face was present. A sharp decrease in the diffusion coefficient of 22 demonstrated that supramolecular polymers formed as the concentration increased. The solution viscosity of the supramolecular polymer was also concentration dependent, which clearly indicates that physical contact between the supramolecular polymers caused entanglement of polymer main-chains. A fiber was drawn from a highly concentrated solution of 22, and a rod-like shape with a regular diameter of 9.5 μm was observed using scanning electron microscopy.
Supramolecular fullerene polymer
Fullerene has drawn significant attention due to its unique three-dimensional geometry and outstanding electrochemical and photophysical properties. In recent years, fullerene chemistry has been used in macromolecular science. This synergetic collaboration offers a great opportunity to generate a new fullerene-based polymer science with numerous applications.72 The synthesis of fullerene-containing polymeric materials was initially attempted via the direct polymerization of a [60]fullerene molecule through cycloaddition reactions.73, 74 However, the products contained serious problems regarding the structure and properties. The highly reactive thirty double bonds of [60]fullerene are chemically equivalent; therefore, the one-, two- and three-dimensionally extended polymeric architectures formed will lose stereo- and regio-regularity and were less processable than conventional polymers. The direct polymerization of [60]fullerene is not useful. Supramolecular methods of incorporating [60]fullerene onto a polymer main chain can overcome this limitation.
A fascinating [60]fullerene-appended supramolecular polymer has been built up by hydrogen-bonding interactions.75 A [60]fullerene derivative bearing two of Meijer's 2-ureido-4-pyrimidone components formed the self-complementary hydrogen-bonded polymer. The maximum molecular weight was theoretically estimated to be approximately 3.6 × 106. The supramolecular fullerene polymer was processable, and a macroscopically homogeneous film was produced.
Liu et al.76 synthesized a water-soluble bis(β-CD) derivative 23. Force field calculations indicated that 23 cannot form a 1:1 inclusion complex with [60]fullerene (Figure 14). However, 23 formed an end-to-end intermolecular inclusion complex with [60]fullerene at the wider end of the β-CD unit.76 The iterative host–guest complexation resulted in supramolecular polymers that incorporated [60]fullerene molecules into the polymer chain. The main-chain structure of the supramolecular polymer was studied using STM. A regular, linear arrangement of the [60]fullerene nano array was observed on an HOPG surface. TEM analysis of the supramolecular polymer showed the presence of a linear structure with a length in the range of 150–250 nm, which indicated that the polymer was composed of 60–80 units of fullerene. The polymer displayed DNA-cutting activity. In the dark, the polymer did not show any activity, but irradiation with visible light cleaved DNA to convert from Form I to Form II due to photoinduced electron transfer.
Haino et al.77, 78, 79, 80, 81, 82, 83 actively investigated the development of calixarene-based host molecules via ‘upper rim’ functionalization. The motivation of the research was to create a much larger cavity capable of encapsulating sizable guest molecules. A calix[5]arene was considered to be a potential candidate.84, 85, 86, 87 In fact, a large molecular guest, [60]fullerene, was encapsulated in the cone cavity.88, 89 In addition, the covalent connection of two calix[5]arene molecules generated a larger cavity capable of encapsulating [60]fullerene, as well as higher fullerenes, which resulted in a large increase in the association constants (Ka>104–5 l mol–1).90,91,92,93,94,95,96,97,98
The synthetic strategy of the supramolecular fullerene polymer has taken advantage of a ‘ball-and-socket’ interaction between [60]fullerene and a double-calix[5]arene structure (Figure 15). Homoditopic host 24 bearing two pockets—each composed of two calix[5]arenes—can capture two [60]fullerene molecules, one in each binding site. When the dumbbell-shaped fullerene 25 and 24 were mixed, the supramolecular polymerization started, via host–guest interactions, to generate supramolecular fullerene polymers.99 1H NMR and UV–vis absorption spectroscopic studies supported the supramolecular association between 24 and 25 in organic solvents. The resonance of the bridging methylene of a calix[5]arene appeared at approximately 4 p.p.m. and was broadened. The supramolecular complexation of 24 and 25 resulted in the observation of an AB-quartet-like split of the bridging methylene protons. The split indicated that the flipping process decreased in frequency, which provided evidence that the energetic barrier to the ring flip was increased due to the host–guest interaction of the [60]fullerene moiety and the cavity. The temperature-dependency of the absorption spectrum indicated that monomers 24 and 25 equilibrated with constitutionally dynamic aggregates. The ratio of the diffusion coefficients of 24 and a 1:1 mixture of 24 and 25 indicated that the assembly of 24 and 25 formed trimeric supramolecular oligomers even at very low concentrations.
In the solid state, the polymer-like morphologies were observed by scanning electron microscopy (Figure 16a). The thicker entwined fibers had length of more than 100 μm and widths of 250–500 nm. The fibrillar networks were produced by the supramolecular assembly of 24 and 25. The addition of [60]fullerene completely disrupted the network; [60]fullerene molecules competitively occupied the cavities of the hosts, which interfered the complexation of 25. As the result, the further growth of the supramolecular polymers was not permitted in the solid state. Figure 16b, an AFM image, shows the nano networks of the fibrous assemblies that originated from the aggregation of the supramolecular linear polymers. Judging from the calculated structure of the oligomers (Figure 16c), the alkyl chains should adopt a parallel arrangement on a mica surface, and the nano assemblies are probably composed of a bundle of 40–60 polymer chains generated by the interdigitation of the alkyl side chains through van der Waals interactions.
It was determined that a π-extended tetrathiafuluvalene (exTTF)-based host could encapsulate [60]fullerene with a very high association constant. Martín et al.100, 101 have recently reported that the heteroditopic molecule 26, which possesses the host and [60]fullerene moieties, assembles to form supramolecular fullerene polymers (Figure 17). DLS experiments on the polymer solution indicated that a broad particle size distribution was present (2–378 nm), which provided evidence for the formation of the large supramolecular fullerene polymers. AFM experiments showed that the self-assembly of 26 produced the fibrous morphology.
Supramolecular porphyrin polymer
A variety of porphyrin-based model compounds have been created to mimic natural light-harvesting complexes with the goal that the materials could be applied to artificial light-harvesting systems and molecular photonic devices. Nanometric multiporphyrin arrays are the most challenging target, wherein, their covalent synthesis offers a highly stabile and a precisely controlled arrangement of multiporphyrin arrays. The growth of the size and complexity of these systems requires tedious synthesis. As a way of overcoming this problem, supramolecular porphyrin polymers have recently attracted great attention owing to the creative application of the materials in photoactive devices. As certain metal–ligand pairs can produce an appreciably strong coordination bond, coordination-driven self-assembly is one of the most useful approaches for building large, elaborate porphyrin architectures.
Ogawa and Kobuke102 and Hunter and Michelsen103 independently reported the coordination-driven supramolecular polymerization of metalloporphyrins (Figure 18). Kobuke’s porphyrin 27 generated supramolecular polymers in a self-complementary manner. Size-exclusion chromatographic analysis of the supramolecular polymers indicated that approximately 400 monomers assembled to form the supramolecular polymers with a length that reached 550 nm. Hunter's cobalt porphyrin 28 also polymerized in a self-complementary manner. The size-exclusion chromatography trace was dependent on the concentration of the monomer, which indicated that the structures of the coordinated polymers are constitutionally dynamic in solution. The DP reached approximately 100 units in length and had a mean molecular weight of 136 kDa.
Hydrogen-bonding-mediated supramolecular porphyrin polymerization has been developed by Aida et al.104 (Figure 19). Carboxylic acid functionalities dimerized through complementary hydrogen-bonding. Porphyrin 29 had two carboxylic acid groups in opposite meso positions. Intermolecular hydrogen bonding induced the supramolecular polymerization, which generated large J aggregates via π–π stacking of the porphyrin cores. Strikingly, spin coating of the supramolecular polymer produced optically active films, where either of the two enantiomeric forms were selected by spinning direction.
Haino et al.105 have recently discovered that a bisporphyrin molecule connected by a pyridinedicarboxamide linker assembled to form a unique, complementary dimer in organic media. The competitive complexation of electron-deficient planar aromatic guests (for example, trinitrofluorenone, trinitrobenzene, tetracyanobenzene and pyromellitic dianhydride) into the bisporphyrin cleft led to a π donor–acceptor-type host–guest complex.106, 107
Based on these findings, a π donor–acceptor-type host–guest interaction was incorporated into a heteroditopic monomer 30 (Figure 20).108 The electron-deficient guest moiety, 4,5,7-trinitrofluorenone-2-carboxylate (TNF), binds within the bisporphyrin cleft via a charge transfer interaction. The iterative head-to-tail host–guest complexation produced a new supramolecular polymer.
The head-to-tail complexation between the bisporphyrin cleft and the TNF moiety resulted in large upfield shifts of the TNF protons, whereas the aromatic TNF protons appeared in unusually upfield regions. These observations indicated that the TNF moiety was located within the porphyrin cleft. The sizes of the aggregates in a variety of concentrations were investigated using DOSY (Figure 21a). The diffusion coefficients of 30 were highly dependent on the concentration, whereas those of its analog bearing the acetyl group instead of the TNF moiety were not influenced by the concentrations. At a low concentration, 30 existed in the monomeric form. At high concentrations, the diffusion coefficient significantly dropped to 10% of the initial value. 30 assembled to form the large polymeric aggregates in response to the concentration. The average size of the supramolecular polymer was calculated to have an approximate DP of 660 by simplistically assuming that all aggregates were hydrospherical.
(a) Diffusion coefficients D of 30 (open circle) and its acetyl analog (filled square) at 298 K in chloroform-d1. (b) Specific viscosity of 30 in chloroform (open circles) and toluene (filled circles) at 293 K. The values denote the slopes. AFM images of the drop-cast films: (c) a solution of 30 and (d) a solution of the acetyl analog.
A concentration-dependent viscosity change is indicative of a transition from a dilute to a semidilute concentration regime, where overlap between the chains contributes to viscous drag (Figure 21b). The dilute concentration regime indicates that the oligomeric assemblies are too short to be entangled. In the semidilute regime, the slope of 3.07 in chloroform indicated that sizable supramolecular polymers formed. The chains became entangled when the polymers came into contact, which is commonly observed in polymer solutions.
The dissociation and recombination of the supramolecular polymers occurs on an experimental time scale. The supramolecular relaxation regime should obey the mixed reptation-breakage regime.109 The noncovalent bond lifetime (τbreak) is longer than the reptation lifetime (τrep), whereas the relaxation time (τ) is approximately equal to τrep. The slope of 3.07 in chloroform demonstrates that the relaxation of the supramolecular polymers occurred via a reptation mechanism, even though the supramolecular polymer is constitutionally dynamic.
AFM visualized the well-developed supramolecular polymer networks of 30, whereas the acetyl analog randomly aggregated to form a particle-like morphology due to the lack of the supramolecular connections (Figures 21c and d). The head-to-tail host–guest complexation between the bisporphyrin cleft and the TNF moiety evidently drives the directional growth of the supramolecular polymer.
Another type of a stimuli-responsive supramolecular polymer was produced using the complementary affinity of a bisporphyrin (Figure 22).110 The bisporphyrin units were connected with 1,4-dibutyne units to produce homoditopic tetrakisporphyrin 31. The iterative self-assembly of 31 resulted in supramolecular nanometric polymeric assemblies. An electron-deficient aromatic guest can aggressively bind within the bisporphyrin cleft and dissociate the supramolecular polymeric assemblies.
The self-assembly of 31 was determined to have a remarkably large association constant of 1 500 000 l mol–1, which implies that sizable supramolecular polymeric aggregates formed at millimolar concentrations. The stimuli-responsive behavior of the supramolecular polymers was observed in solution (Figures 23a and b). The resonance peaks in the 1H NMR spectrum of the supramolecular polymers were highly broadened, which was probably owing to the numerous conformational and structural possibilities. Upon introduction of an electron-deficient aromatic guest (for example, 2,4,7-trinitrofluorenone) to the solution, the broad signals sharpened and the spectrum indicated that 2,4,7-TNF2 31 formed with the simultaneous dissociation of the supramolecular polymers.
In the solid state, AFM images of the long, winding and fibrous fragments demonstrated that 31 iteratively created complementary connections with each terminal functionality that resulted in the supramolecular polymers (Figures 23c and d). Coiled structures with a pitch of 64±6 nm were observed in the magnified images. The bundled supramolecular polymeric assemblies probably adopted coiled superstructures. Based on the value of the pitch, the coiled tape was composed of a bundle of approximately 30 chains of polymeric assemblies (Figure 22).
Host–guest interactions are useful for constructing supramolecular polymeric aggregates with high orthogonality. However, hydrogen and coordination bonds are popular because the units that provide the interactions are synthetically accessible. Unique hosts, including a calixarene, CD, crown ether and porphyrin, show large association constants with high guest selectivity that are caused by structural characteristics. Therefore, host–guest interactions provide numerous methods for supramolecular polymer synthesis, and further development of supramolecular polymer science for future materials is foreseen.
Supramolecular cross-linking of polymer chains
The cross-linking of polymeric chains is one of the most important topics in polymer science. The cross-linking of linear polymers creates three-dimensional networks and reduces structural flexibility, which lead to dramatic changes in the macroscopic morphologies and properties. However, cross-linking is irreversible because the three-dimensional networks are constructed with covalent bonds. The introduction of a reversible polymer cross-linkage is a challenging issue for generating stimuli-responsive polymer materials. Reversible cross-linkages can be realized by employing noncovalent bonds. In the event that supramolecular entities are grafted onto conventional polymeric scaffolds, then reversible cross-linking of polymer chains is established by a supramolecular method.
Hydrogen bonding has been used by Stadler et al.111, 112 as a secondary interaction that connects polymer main chains. 4-Urazoylbenzoic acid groups (U4A) were randomly attached to flexible 1,4-polybutadienes (Figure 24).113, 114, 115 Functionalized polymer 32 formed interchain associations through the intermolecular hydrogen bonding of the U4A functional groups. Further aggregation resulted in two-dimensional supramolecular structures, which were phase separated from the amorphous polymer matrix. The supramolecular structures highly stabilized the interchain interactions, which caused remarkable increases in the Tm and endothermic transition enthalpy. The changes are a function of the degree of U4A modification.
2-Ureido-4[1H]-pyrimidinone (UPy) was introduced to the main chain of polyolefins by Coates et al.116 (Figure 25). The pendant UPy dimerized to form self-complementary pairs within the copolymer 33 matrix and formed reversible hydrogen-bonding cross-linkages that generated polymer networks with regulable properties. The solution viscosity of the copolymer was intriguing. In the low-concentration regime, the copolymer behaved similar to the homopolymer without the UPy moiety. At higher concentrations, the UPy units dimerized to connect the polymer main chains, which dramatically increased the effective molecular weight. Polymer 33 became a gel in toluene at higher concentrations (> 20 g l–1). In bulk, the copolymer displayed elastomeric properties, verifying the formation of a network via noncovalent interactions. After initial deformation of the homopolymer, associated with uncoiling amorphous chains, the minimal stress was required for maintaining elongation, which implied that the lack of strong intermolecular forces permitted the main chains to reptate. During the initial deformation, the polymer main chains of 33 and the homopolymer required low stress to uncoil. However, the cross-linkage of the UP moieties inhibited the chains from passing each other; therefore, stress remarkably increases as deformation continues. The multipoint hydrogen bonds obviously resulted in the dramatic change of the bulk elastomeric properties, even though they are noncovalent bonds. At this point, the macroscopic properties of the copolymer are regulable and reversible.
Ritter et al.117 showed that a β-CD dimer cross-linked an adamantyl-containing poly(aclylamide) copolymer. The β-CD dimer selectively encapsulated the adamantyl moieties into the hydrophobic cavities, which led to a polymeric network. This cross-linkage produced supramolecular hydrogels. Auzély-Velty et al.118, 119, 120 reported that adamantyl-grafted chitosan chains were linked using a β-CD dimer via selective molecular recognition, which enhanced the viscosity of the copolymer solution.
The molecular recognition events of CDs have been successfully visualized into a macroscopic process (Figure 26).121 Polyacrylamide gels bearing either CD or a guest moiety were prepared as cubic gel objects: α-CD-gel, β-CD-gel, n-Bu-gel and t-Bu-gel. An α-CD host selectively encapsulates an n-Bu group, while a t-Bu group is the best guest for a β-CD host. This specific molecular-level recognition process even occurred at the interface between two gel cubes. A mixture of pieces of α-CD-gel, β-CD-gel, n-Bu-gel and t-Bu-gel exhibited excellent fidelity after being mixed in the same container: α-CD-gel and β-CD-gel selectively adhered to n-Bu-gel and t-Bu-gel, respectively. This was a beautiful demonstration of how molecular recognition can lead to macroscopic attraction.
Specific molecular recognition events (a, b, c) on a macroscopic scale. (Reproduced with permission from Harada et al.121. Copyright 2012 Nature Publishing Group.).
A novel approach to generate supramolecular cross-linking of polycrown ether 34 was demonstrated by Takata et al.122 using both supramolecular and dynamic covalent chemistry (Figure 27). The method involved two reversible interactions: one was the dynamic covalent bond of disulfide, and the other was the supramolecular interaction between the crown ether and the ammonium salt 35, that is, the formations of rotaxane structures in which each of the units are interlocked. Diammonium salt 35 threaded into the crown ether moieties of polymer 34 via a reversible cleavage of the disulfide bond. Upon recombination of the disulfide bond, the generation of the interchain topological linkage resulted in a polyrotaxane network that formed an elastic, transparent gel. In DMF or DMSO, the gel swelled appreciably. Breaking down the cross-linkages of the polymer networks permitted the quantitative recovery of the original polymer 34. This is a new concept in recyclable cross-linked polymers.
Rudkevich et al.123, 124, 125, 126 employed Rebek's ‘polycap’ supramolecular structure for capturing carbon dioxide gas in gel networks (Figure 28). The strategy was to build switchable polymer networks that consisted of ‘polycap’ and carbamate salt bridges. The dilysine-linked biscalixarene 36 formed a ‘polycap’. The polymeric chain had a high DP of approximately 300, which possessed basic amino groups on the periphery. Carbon dioxide molecules were captured by the amino groups of the polymer chains, and created the interchain carbamate salt bridges that cross-linked the main chains. The highly interconnected supramolecular polymer networks were disrupted in two ways: the addition of competitive solvents can break hydrogen bonds, and the thermal release of carbon dioxide can be accomplished with retention of the hydrogen-bonded polycap. Additionally, the three-dimensional networks produced fluorescent gels in benzene.126
Hydrogen-bonding functionalities of UG and DAN (Figure 9) have been incorporated into PBMA and PS polymer backbones by Park and Zimmerman.127, 128 UG and DAN units were useful supramolecular cross-linkers as a result of the high stability and fidelity of the heterodimer complex. The immiscible polymers, UG-PBMA and DAN-PS, were interlinked by the molecular recognition of the UG and DAN units. SEC, viscosity and DLS experiments indicated that the supramolecular cross-linkages increased the molecular weight and size of the polymer blends. This supramolecular connection allows the UG-PBMA and DAN-PS polymers to be blended together to produce transparent films without phase separation on either the nano- or macroscopic level. The supramolecular polymer composed of UG-PBMA and DAN-PS gelled in chloroform, whereas UG-PBMA and PBMA flowed freely due to a lack of specific interchain interactions.
Ditopic tetrakiscalix[5]arene host 24 selectively encapsulates [60]fullerene moieties, which are grafted on a large polymer backbone (Figure 29).129 This molecular recognition event creates a remarkably stable cross-linkage. The complexation of 24 to polymer 37 increased the molecular weight to more than twice the weight of free 37 (Figure 30a). Surprisingly, excess 24 does not break preformed cross-linkages in toluene, even though the host–guest complexation of a calix[5]arene and [60]fullerene is driven by noncovalent interactions. The cross-linking behavior is influenced by solvent properties. The more competitive solvation of chloroform and o-dichlorobenzene (relative to toluene) weakened the host–guest interaction, which caused host 24 to be unable to create stable cross-linkages with 37.
The macroscopic solid-state morphologies of 37 were highly influenced by supramolecular cross-linking. The agglomerated nanostructures of 37 consisted of particle-like aggregates that resulted from the cohesion of [60]fullerene moieties through π–π interactions and the immiscible nature of [60]fullerene (Figures 30b and d). The encapsulation of [60]fullerene moieties by the biscalix[5]arene moiety of 24 disrupts the interfullerene interactions, and then the polymer chain gets longer via the supramolecular cross-linking. This elongation led to a dramatic morphological change in which the particle-like nanostructures completely disappeared, and a widespread fibrous network formed (Figure 30c). The cross-linked fibrils aligned into a well-oriented 2D array on a HOPG surface (Figure 30e).
Self-healing is one of the most fascinating functions that appear in supramolecular polymer materials. The ability to mend damage becomes available by the dynamic properties of supramolecular polymers. Supramolecular cross-linkage provides a possibility to fabricate self-healing materials in practical use. Recently, a beautiful self-healing rubber was created by Leibler et al.130 Thermoreversible rubber elastic materials were prepared by the condensation of fatty di- and tri-acids with diethylene triamine, and were then reacted with urea. Each of the components was interconnected by hydrogen-bonding interactions to generate highly stable three-dimensional networks, which resulted in the rubber-like properties of the material. The stress–strain correlation of the material resembled those of soft rubbers. The strain-at-break was greater than 500%. After the material was ruptured, the broken hydrogen bonds assumed nonassociated forms at the interface. Upon interfacial contact, the nonassociated hydrogen bonds restored the hydrogen-bonded networks to mend the broken pieces.
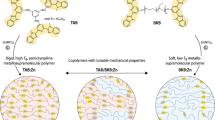
Spot-on healable polymer materials were developed by Rowan et al.131 A poly(ethylene-co-butylene) core with 2,6-bis(1′-methylbenzimidazolyl)pyridine (Mebip) ligands at the termini polymerized through the complexation of zinc or lanthanum ions. Zn2+ ions bound to the Mebip ligands in a 2:1 ratio, whereas the La3+ ions formed more dynamic 3:1 complexes with the ligands. The coordination-driven supramolecular polymers had microphase-separated lamellar morphologies in which the hydrophobic polymer domains were cross-linked by the metal–ligand complexes as a hard phase. The remarkable characteristic of the coordination polymers were the pinpoint healing properties driven by exposure to UV light. When the metal–ligand complexes were electronically excited, the energy turned into heat. The heat caused the disassembly of the metal–ligand complexes, which temporarily reduced the molecular weight and viscosity. The free metal ions were able to recombine with residual ligands. The damaged polymers were restored via photothermal healing, which sealed cuts in the film.
The supramolecular methods can be another choice to control polymer properties via cross-linking. Of course, hydrogen bonds, dipole–dipole, and van der Waals interactions direct the higher order of polymer organization. However, these interactions may unpredictably contribute to polymer organization. The rationally designed supramolecular structures provide high specificity in molecular recognition events, that is, the recognition events link polymer chains in a desired fashion. This method may provide easy access to a variety of fascinating functions that result from cross-linked polymeric materials.
Conclusion and outlook
In the last few decades, noncovalent interactions have been recognized to be an essential determinant of forthcoming structures and properties of polymer materials. Despite the fact that noncovalent interactions are relatively weak and some are only slightly directional, supramolecular chemistry allows weak noncovalent interactions to be manipulated in a desired manner. The rational design of a supramolecular structure distinguishes a complementary molecular pair from other competitive interactions.
As supramolecular chemistry has been integrated with polymer chemistry, supramolecular polymer chemistry is already considered to be a field of polymer science. However, the low stability of noncovalent bonds may restrict the flexible use of supramolecular polymers in real applications owing to poor robustness. In this review, many available options to compensate for this defect have been described. 1) Supramolecular complexes have been developed to produce remarkably large association constants. Employing interactions with large association constants in supramolecular polymerization induces a very high DP that results in tough supramolecular materials. 2) A polytopic monomer expands in two or three dimensions to form supramolecular polymer networks. Entanglement of the supramolecular networks leads to robust materials. 3) Block copolymerization and the cross-linking of polymer chains using supramolecular methods, that is, noncovalent bonds integrate mechanical integrity into supramolecular polymers. Because of the development of numerous synthetic strategies for supramolecular polymers, the focus has shifted to incorporate a variety of functionalities. The functionalities target practical uses of supramolecular polymers as advanced materials with adaptive properties that allow simple processing, facile recycling, self-healing and stimuli responsiveness. The structural and dynamic properties of supramolecular polymers have opened up a new avenue for the development of such materials. Supramolecular polymer-based materials present a bright future for the next generation of polymer chemistry. It is an exciting time for research in the field of supramolecular polymer chemistry.
References
Serpe, M. J. & Craig, S. L. Physical organic chemistry of supramolecular polymers. Langmuir 23, 1626–1634 (2007).
Lehn, J.-M. Supramolecular Polymer Chemistry – Scope and Perspectives, Supramolecular Polymer (ed. Ciferri, A) 2nd edn 3–27 (CRC Press LLC, 2005).
Ciferri, A. Growth of Supramolecular Structures, Supramolecular Polymer (ed. Ciferri, A) 2nd edn 29–75 (CRC Press LLC, 2005).
Brunsveld, L., Folmer, B. J. B., Meijer, E. W. & Sijbesma, R. P. Supramolecular polymers. Chem. Rev. 101, 4071–4097 (2001).
De Greef, T. F. A., Smulders, M. M. J., Wolffs, M., Schenning, A. P. H. J., Sijbesma, R. P. & Meijer, E. W. Supramolecular polymerization. Chem. Rev. 109, 5687–5754 (2009).
Craig, S. L. From ionic liquids to supramolecular polymers. Angew. Chem. Int. Ed. 48, 2645–2647 (2009).
Klosterman, J. K., Yamauchi, Y. & Fujita, M. Engineering discrete stacks of aromatic molecules. Chem. Soc. Rev. 38, 1714–1725 (2009).
Würthner, F., Kaiser, T. E. & Saha-Möller, C. R. J-aggregates: from serendipitous discovery to supramolecular engineering of functional dye materials. Angew. Chem. Int. Ed. 50, 3376–3410 (2011).
Krieg, E. & Rybtchinski, B. Noncovalent water-based materials: robust yet adaptive. Chem. Eur. J. 17, 9016–9026 (2011).
Hoeben, F. J. M., Jonkheijm, P., Meijer, E. W. & Schenning, A. P. H. J. About supramolecular assemblies of π-conjugated systems. Chem. Rev. 105, 1491–1546 (2005).
Bushey, M. L., Nguyen, T. Q., Zhang, W., Horoszewski, D. & Nuckolls, C. Using hydrogen bonds to direct the assembly of crowded aromatics. Angew. Chem. Int. Ed. 43, 5446–5453 (2004).
Vera, F., Serrano, J. L. & Sierra, T. Twists in mesomorphic columnar supramolecular assemblies. Chem. Soc. Rev. 38, 781–796 (2009).
Keizer, H. M. & Sijbesma, R. P. Hierarchical self-assembly of columnar aggregates. Chem. Soc. Rev. 34, 226–234 (2005).
Yagai, S. Supramolecular complexes of functional chromophores based on multiple hydrogen-bonding interactions. J. Photoch. Photobio. C 7, 164–182 (2006).
Fouquey, C., Lehn, J.-M. & Levelut, A.-M. Molecular recognition directed self-assembly of supramolecular liquid crystalline polymers from complementary chiral components. Adv. Mater. 2, 254–257 (1990).
Kotera, M., Lehn, J. M. & Vigneron, J. P. Self-Assembled Supramolecular Rigid Rods. J. Chem. Soc. Chem. Commun. 197–199 (1994).
Gulikkrzywicki, T., Fouquey, C. & Lehn, J. M. Electron-microscopic study of supramolecular liquid-crystalline polymers formed by molecular-recognition-directed self-assembly from complementary chiral components. Proc. Natl Acad. Sci. USA 90, 163–167 (1993).
Berl, V., Schmutz, M., Krische, M. J., Khoury, R. G. & Lehn, J. M. Supramolecular polymers generated from heterocomplementary monomers linked through multiple hydrogen-bonding arrays - Formation, characterization, and properties. Chem. Eur. J. 8, 1227–1244 (2002).
Ghadiri, M. R., Granja, J. R. & Buehler, L. K. Artificial transmembrane ion channels from self-assembling peptide nanotubes. Nature 369, 301–304 (1994).
Granja, J. R. & Ghadiri, M. R. Channel-mediated transport of glucose across lipid bilayers. J. Am. Chem. Soc. 116, 10785–10786 (1994).
Khazanovich, N., Granja, J. R., Mcree, D. E., Milligan, R. A. & Ghadiri, M. R. Nanoscale tubular ensembles with specified internal diameters - design of a self-assembled nanotube with a 13-angstrom pore. J. Am. Chem. Soc. 116, 6011–6012 (1994).
Sijbesma, R. P., Beijer, F. H., Brunsveld, L., Folmer, B. J. B., Hirschberg, J. H. K. K., Lange, R. F. M., Lowe, J. K. L. & Meijer, E. W. Reversible polymers formed from self-complementary monomers using quadruple hydrogen bonding. Science 278, 1601–1604 (1997).
Sontjens, S. H. M., Sijbesma, R. P., van Genderen, M. H. P. & Meijer, E. W. Selective formation of cyclic dimers in solutions of reversible supramolecular polymers. Macromolecules 34, 3815–3818 (2001).
Hirschberg, J. H. K. K., Brunsveld, L., Ramzi, A., Vekemans, J. A. J. M., Sijbesma, R. P. & Meijer, E. W. Helical self-assembled polymers from cooperative stacking of hydrogen-bonded pairs. Nature 407, 167–170 (2000).
Abbel, R., Grenier, C., Pouderoijen, M. J., Stouwdam, J. W., Leclere, P. E. L. G., Sijbesma, R. P., Meijer, E. W. & Schenning, A. P. H. J. White-light emitting hydrogen-bonded supramolecular copolymers based on π-conjugated oligomers. J. Am. Chem. Soc. 131, 833–843 (2009).
Castellano, R. K., Rudkevich, D. M. & Rebek, J. Polycaps: reversibly formed polymeric capsules. Proc. Natl Acad. Sci. USA 94, 7132–7137 (1997).
Castellano, R. K., Clark, R., Craig, S. L., Nuckolls, C. & Rebek, J. Emergent mechanical properties of self-assembled polymeric capsules. Proc. Natl Acad. Sci. USA 97, 12418–12421 (2000).
Whitesides, G. M., Simanek, E. E., Mathias, J. P., Seto, C. T., Chin, D. N., Mammen, M. & Gordon, D. M. Noncovalent synthesis - using physical-organic chemistry to make aggregates. Acc. Chem. Res. 28, 37–44 (1995).
Whitesides, G. M., Mathias, J. P. & Seto, C. T. Molecular self-assembly and nanochemistry - a chemical strategy for the synthesis of nanostructures. Science 254, 1312–1319 (1991).
Klok, H. A., Jolliffe, K. A., Schauer, C. L., Prins, L. J., Spatz, J. P., Moller, M., Timmerman, P. & Reinhoudt, D. N. Self-assembly of rodlike hydrogen-bonded nanostructures. J. Am. Chem. Soc. 121, 7154–7155 (1999).
Choi, I. S., Li, X. H., Simanek, E. E., Akaba, R. & Whitesides, G. M. Self-assembly of hydrogen-bonded polymeric rods based on the cyanuric acid center dot melamine lattice. Chem. Mater. 11, 684–690 (1999).
Fogleman, E. A., Yount, W. C., Xu, J. & Craig, S. L. Modular, well-behaved reversible polymers from DNA-based monomers. Angew. Chem. Int. Ed. 41, 4026–4028 (2002).
Xu, J., Fogleman, E. A. & Craig, S. L. Structure and properties of DNA-based reversible polymers. Macromolecules 37, 1863–1870 (2004).
Würthner, F., Yao, S. & Beginn, U. Highly ordered merocyanine dye assemblies by supramolecular polymerization and hierarchical self-organization. Angew. Chem. Int. Ed. 42, 3247–3250 (2003).
Yao, S., Beginn, U., Gress, T., Lysetska, M. & Würthner, F. Supramolecular polymerization and gel formation of Bis(merocyanine) dyes driven by dipolar aggregation. J. Am. Chem. Soc. 126, 8336–8348 (2004).
Schmidt, R., Stolte, M., Gruene, M. & Würthner, F. Hydrogen-bond-directed formation of supramolecular polymers incorporating head-to-tail oriented dipolar merocyanine dyes. Macromolecules 44, 3766–3776 (2011).
Yang, S. K., Ambade, A. V. & Weck, M. Main-chain supramolecular block copolymers. Chem. Soc. Rev. 40, 129–137 (2011).
Folmer, B. J. B., Sijbesma, R. P., Versteegen, R. M., van der Rijt, J. A. J. & Meijer, E. W. Supramolecular polymer materials: chain extension of telechelic polymers using a reactive hydrogen-bonding synthon. Adv. Mater. 12, 874–878 (2000).
Binder, W. H., Bernstorff, S., Kluger, C., Petraru, L. & Kunz, M. J. Tunable materials from hydrogen-bonded pseudo block copolymers. Adv. Mater. 17, 2824–2828 (2005).
Park, T. & Zimmerman, S. C. A supramolecular multi-block copolymer with a high propensity for alternation. J. Am. Chem. Soc. 128, 13986–13987 (2006).
Yang, S. K., Ambade, A. V. & Weck, M. Supramolecular ABC triblock copolymers via one-pot, orthogonal self-assembly. J. Am. Chem. Soc. 132, 1637–1645 (2010).
Ambade, A. V., Yang, S. K. & Weck, M. Supramolecular ABC triblock copolymers. Angew. Chem. Int. Ed. 48, 2894–2898 (2009).
Kyba, E. P., Helgeson, R. C., Madan, K., Gokel, G. W., Tarnowski, T. L., Moore, S. S. & Cram, D. J. Host-guest complexation.1. concept and illustration. J. Am. Chem. Soc. 99, 2564–2571 (1977).
Pedersen, C. J. The discovery of crown ethers (Noble Lecture). Angew. Chem. Int. Ed. Engl. 27, 1021–1027 (1988).
Helgeson, R. C., Tarnowski, T. L., Timko, J. M. & Cram, D. J. Host-guest complexation.6. [2.2]paracyclophanyl structural unit in host compounds. J. Am. Chem. Soc. 99, 6411–6418 (1977).
Shinkai, S., Yoshida, T., Manabe, O. & Fuchita, Y. Photoresponsive crown ethers.20. reversible photocontrol of association-dissociation equilibria between azobis(benzo-18-crown-6) and diammonium cations. J. Chem. Soc. Perkin Trans. 1, 1431–1437 (1988).
Yamaguchi, N., Nagvekar, D. S. & Gibson, H. W. Self-organization of a heteroditopic molecule to linear polymolecular arrays in solution. Angew. Chem. Int. Ed. 37, 2361–2364 (1998).
Gibson, H. W., Yamaguchi, N. & Jones, J. W. Supramolecular pseudorotaxane polymers from complementary pairs of homoditopic molecules. J. Am. Chem. Soc. 125, 3522–3533 (2003).
Niu, Z. B., Huang, F. H. & Gibson, H. W. Supramolecular AA-BB-type linear polymers with relatively high molecular weights via the self-assembly of bis(m-phenylene)-32-crown-10 cryptands and a bisparaquat derivative. J. Am. Chem. Soc. 133, 2836–2839 (2011).
Wang, F., Han, C. Y., He, C. L., Zhou, Q. Z., Zhang, J. Q., Wang, C., Li, N. & Huang, F. H. Self-sorting organization of two heteroditopic monomers to supramolecular alternating copolymers. J. Am. Chem. Soc. 130, 11254–11255 (2008).
Lagona, J., Mukhopadhyay, P., Chakrabarti, S. & Isaacs, L. The cucurbit[n]uril family. Angew. Chem. Int. Ed. 44, 4844–4870 (2005).
Kim, H. J., Heo, J., Jeon, W. S., Lee, E., Kim, J., Sakamoto, S., Yamaguchi, K. & Kim, K. Selective inclusion of a hetero-guest pair in a molecular host: formation of stable charge-transfer complexes in cucurbit[8]uril. Angew. Chem. Int. Ed. 40, 1526–1529 (2001).
Kim, K., Kim, D., Lee, J. W. & Ko, Y. H. Growth of poly(pseudorotaxane) on gold using host-stabilized charge-transfer interaction. Chem. Commun. 848–849 (2004).
Rauwald, U. & Scherman, O. A. Supramolecular block copolymers with cucurbit[8]uril in water. Angew. Chem. Int. Ed. 47, 3950–3953 (2008).
Liu, Y. L., Yu, Y., Gao, J. A., Wang, Z. Q. & Zhang, X. Water-soluble supramolecular polymerization driven by multiple host-stabilized charge-transfer interactions. Angew. Chem. Int. Ed. 49, 6576–6579 (2010).
Li, G. & McGown, L. B. Molecular nanotube aggregates of beta-cyclodextrins and gamma-cyclodextrins linked by diphenylhexatrienes. Science 264, 249–251 (1994).
Harada, A., Takashima, Y. & Yamaguchi, H. Cyclodextrin-based supramolecular polymers. Chem. Soc. Rev. 38, 875–882 (2009).
Ohga, K., Takashima, Y., Takahashi, H., Kawaguchi, Y., Yamaguchi, H. & Harada, A. Preparation of supramolecular polymers from a cyclodextrin dimer and ditopic guest molecules: control of structure by linker flexibility. Macromolecules 38, 5897–5904 (2005).
Miyauchi, M., Takashima, Y., Yamaguchi, H. & Harada, A. Chiral supramolecular polymers formed by host-guest interactions. J. Am. Chem. Soc. 127, 2984–2989 (2005).
Miyauchi, M. & Harada, A. A helical supramolecular polymer formed by host-guest interactions. Chem. Lett. 34, 104–105 (2005).
Gutsche, C. D. Calixarenes, Monographs in Supramolecular Chemistry (ed. Stoddart J. F., (Royal Society of Chemistry, Cambridge, 1989).
Gutsche, C. D. Calixarenes Revisited, Monographs in Supramolecular Chemistry (ed. Stoddart J. F., (Royal Society of Chemistry, Cambridge, 1998).
Gutsche, C. D. Calixarenes: An Introduction, 2nd Edition, Monographs in Supramolecular Chemistry (ed. Stoddart J. F., (Royal Society of Chemistry, Cambridge, 2008).
Dalcanale, E., Yebeutchou, R. M., Tancini, F., Demitri, N., Geremia, S. & Mendichi, R. Host-guest driven self-assembly of linear and star supramolecular polymers. Angew. Chem. Int. Ed. 47, 4504–4508 (2008).
Tancini, F., Yebeutchou, R. M., Pirondini, L., De Zorzi, R., Geremia, S., Scherman, O. A. & Dalcanale, E. Host-guest-driven copolymerization of tetraphosphonate cavitands. Chem. Eur. J. 16, 14313–14321 (2010).
Arnaud-Neu, F., Fuangswasdi, S., Notti, A., Pappalardo, S. & Parisi, M. F. Calix[5]arene-based molecular vessels for alkylammonium ions. Angew. Chem. Int. Ed. 37, 112–114 (1998).
Parisi, M. F., Garozzo, D., Gattuso, G., Kohnke, F. H., Notti, A., Pappalardo, S., Pisagatti, I., White, A. J. P. & Williams, D. J. Inclusion networks of a calix[5]arene-based exoditopic receptor and long-chain alkyldiammonium ions. Org. Lett. 5, 4025–4028 (2003).
Cohen, Y., Pappalardo, S., Villari, V., Slovak, S., Gattuso, G., Notti, A., Pappalardo, A., Pisagatti, I. & Parisi, M. R. Counterion-dependent proton-driven self-assembly of linear supramolecular oligomers based on amino-calix[5]arene building blocks. Chem. Eur. J. 13, 8164–8173 (2007).
Parisi, M. F., Gattuso, G., Notti, A., Pappalardo, A., Pisagatti, I., Pappalardo, S., Garozzo, D., Messina, A., Cohen, Y. & Slovak, S. Self-assembly dynamics of modular homoditopic bis-calix[5]arenes and long-chain alpha,omega-alkanediyldiammonium components. J. Org. Chem. 73, 7280–7289 (2008).
Ogoshi, T., Kanai, S., Fujinami, S., Yamagishi, T. A. & Nakamoto, Y. para-bridged symmetrical pillar[5]arenes: their Lewis acid catalyzed synthesis and host-guest property. J. Am. Chem. Soc. 130, 5022–5023 (2008).
Zhang, Z., Luo, Y., Chen, J., Dong, S., Yu, Y., Ma, Z. & Huang, F. Formation of linear supramolecular polymers that is driven by C-H•••π interactions in solution and in the solid state. Angew. Chem. Int. Ed. 50, 1397–1401 (2011).
Giacalone, F. & Martín, N. Fullerene polymers: Synthesis and properties. Chem. Rev. 106, 5136–5190 (2006).
Iwasa, Y., Arima, T., Fleming, R. M., Siegrist, T., Zhou, O., Haddon, R. C., Rothberg, L. J., Lyons, K. B., Carter, H. L., Hebard, A. F., Tycko, R., Dabbagh, G., Krajewski, J. J., Thomas, G. A. & Yagi, T. New phases of C-60 synthesized at high-pressure. Science 264, 1570–1572 (1994).
Zhou, P., Dong, Z.-H., Rao, A. M. & Eklund, P. C. Reaction-mechanism for the photopolymerization of solid fullerene C-60. Chem Phys Lett. 211, 337–340 (1993).
Sánchez, L., Rispens, M. T. & Hummelen, J. C. A supramolecular array of fullerenes by quadruple hydrogen bonding. Angew. Chem. Int. Ed. 41, 838–840 (2002).
Liu, Y., Wang, H., Liang, P. & Zhang, H.-Y. Water-soluble supramolecular fullerene assembly mediated by metallobridged β-cyclodextrins. Angew. Chem. Int. Ed. 43, 2690–2694 (2004).
Haino, T., Shio, H., Takano, R. & Fukazawa, Y. Asymmetric induction of supramolecular helicity in calix[4]arene-based triple-stranded helicate. Chem. Commun. 2481–2484 (2009).
Iwamoto, H., Niimi, K., Haino, T. & Fukazawa, Y. Energetics of guest binding to calix[4]arene molecular containers. Tetrahedron 65, 7259–7267 (2009).
Haino, T., Hirakata, M., Niimi, K., Kouchi, M., Iwamoto, H. & Fukazawa, Y. Synthesis of a molecular container having an open door that closes on guest binding. Chem. Lett. 37, 394–395 (2008).
Haino, T., Kobayashi, M. & Fukazawa, Y. Guest encapsulation and self-assembly of a cavitand-based coordination capsule. Chem. Eur. J. 12, 3310–3319 (2006).
Haino, T., Kobayashi, M., Chikaraishi, M. & Fukazawa, Y. A new self-assembling capsule via metal coordination. Chem. Commun. 2321–2323 (2005).
Haino, T., Nakamura, M., Kato, N., Hiraoka, M. & Fukazawa, Y. Calix[4]arene-based ditopic receptor for dicarboxylates. Tetrahedron Lett. 45, 2281–2284 (2004).
Haino, T., Katsutani, Y., Akii, H. & Fukazawa, Y. Allosteric receptor based on monodeoxycalix[4]arene crown ether. Tetrahedron Lett. 39, 8133–8136 (1998).
Haino, T., Fukuoka, H., Iwamoto, H. & Fukazawa, Y. Synthesis and enantioselective recognition of a calix[5]arene-based chiral receptor. Supramol. Chem. 20, 51–57 (2008).
Haino, T., Fukuoka, H., Katayama, M. & Fukazawa, Y. Enantioselective recognition of calix[5]arene-based artificial receptor bearing chiral macrocycle for chiral ethyltrimethylammonium salts. Chem. Lett. 36, 1054–1055 (2007).
Haino, T., Matsumura, K., Harano, T., Yamada, K., Saijyo, Y. & Fukazawa, Y. Synthesis and binding behavior of an artificial receptor based on ‘upper rim’ functionalized calix[5]arene. Tetrahedron 54, 12185–12196 (1998).
Haino, T., Harano, T., Matsumura, K. & Fukazawa, Y. Synthesis of a new receptor based on ‘upper rim’ functionalized calix[5]arene. Tetrahedron Lett. 36, 5793–5796 (1995).
Haino, T., Yanase, M. & Fukazawa, Y. New supramolecular complex of C60 based on calix[5]arene - its structure in the crystal and in solution. Angew. Chem. Int. Ed. Engl. 36, 259–260 (1997).
Haino, T., Yanase, M. & Fukazawa, Y. Crystalline supramolecular complexes of C60 with calix[5]arenes. Tetrahedron Lett. 38, 3739–3742 (1997).
Haino, T., Fukunaga, C. & Fukazawa, Y. A new calix[5]arene-based container: selective extraction of higher fullerenes. Org. Lett. 8, 3545–3548 (2006).
Haino, T., Yanase, M. & Fukazawa, Y. Fullerenes enclosed in bridged calix[5]arenes. Angew. Chem. Int. Ed. 37, 997–998 (1998).
Haino, T., Fukunaga, C. & Fukazawa, Y. Complexation of higher fullerenes by calix[5]arene-based host molecules. J. Nanosci. Nanotech. 7, 1386–1388 (2007).
Haino, T., Yanase, M., Fukunaga, C. & Fukazawa, Y. Fullerene encapsulation with calix[5]arenes. Tetrahedron 62, 2025–2035 (2006).
Haino, T., Seyama, J., Fukunaga, C., Murata, Y., Komatsu, K. & Fukazawa, Y. Calix[5]arene-based receptor for dumb-bell-shaped C120 . Bull. Chem. Soc. Jpn. 78, 768–770 (2005).
Haino, T., Yamanaka, Y., Araki, H. & Fukazawa, Y. Metal-induced regulation of fullerene complexation with double-calix[5]arene. Chem. Commun. 402–403 (2002).
Haino, T., Araki, H., Fujiwara, Y., Tanimoto, Y. & Fukazawa, Y. Fullerene sensors based on calix[5]arene. Chem. Commun. 2148–2149 (2002).
Haino, T., Araki, H., Yamanaka, Y. & Fukazawa, Y. Fullerene receptor based on calix[5]arene through metal-assisted self-assembly. Tetrahedron Lett. 42, 3203–3206 (2001).
Yanase, M., Haino, T. & Fukazawa, Y. A self-assembling molecular container for fullerenes. Tetrahedron Lett. 40, 2781–2784 (1999).
Haino, T., Matsumoto, Y. & Fukazawa, Y. Supramolecular nano networks formed by molecular-recognition-directed self-assembly of ditopic calix[5]arene and dumbbell [60]fullerene. J. Am. Chem. Soc 127, 8936–8937 (2005).
Fernández, G., Pérez, E. M., Sánchez, L. & Martín, N. Self-organization of electroactive materials: a head-to-tail donor-acceptor supramolecular polymer. Angew. Chem. Int. Ed. 47, 1094–1097 (2008).
Fernandez, G., Perez, E. M., Sanchez, L. & Martín, N. An electroactive dynamically polydisperse supramolecular dendrimer. J. Am. Chem. Soc. 130, 2410–2411 (2008).
Ogawa, K. & Kobuke, Y. Formation of a giant supramolecular porphyrin array by self-coordination. Angew. Chem. Int. Ed. 39, 4070–4073 (2000).
Michelsen, U. & Hunter, C. A. Self-assembled porphyrin polymers. Angew. Chem. Int. Ed. 39, 764–767 (2000).
Yamaguchi, T., Kimura, T., Matsuda, H. & Aida, T. Macroscopic spinning chirality memorized in spin-coated films of spatially designed dendritic zinc porphyrin J-aggregates. Angew. Chem. Int. Ed. 43, 6350–6355 (2004).
Haino, T., Fujii, T. & Fukazawa, Y. Self-complementary bis-porphyrins. Tetrahedron Lett. 46, 257–260 (2005).
Haino, T., Fujii, T. & Fukazawa, Y. Guest binding and new self-assembly of bisporphyrins. J. Org. Chem. 71, 2572–2580 (2006).
Ikeda, T., Watanabe, A., Oshita, T. & Haino, T. Electrochemically switchable supramolecular complex between bisporphyrin cleft and electron-deficient aromatic guest. Heteroatom Chem. 22, 590–593 (2011).
Haino, T., Watanabe, A., Hirao, T. & Ikeda, T. Supramolecular polymerization triggered by molecular recognition between bisporphyrin and trinitrofluorenone. Angew. Chem. Int. Ed. 1473–1476 (2012).
Cates, M. E. Reptation of living polymers - dynamics of entangled polymers in the presence of reversible chain-scission reactions. Macromolecules 20, 2289–2296 (1987).
Haino, T., Fujii, T., Watanabe, A. & Takayanagi, U. Supramolecular polymer formed by reversible self-assembly of tetrakisporphyrin. Proc. Natl Acad. Sci. USA 106, 10477–10481 (2009).
Stadler, R. & Freitas, L. D. Thermoplastic elastomers by hydrogen-bonding.1. Rheological properties of modified polybutadiene. Colloid. Polym. Sci. 264, 773–778 (1986).
Freitas, L. D., Burgert, J. & Stadler, R. Thermoplastic elastomers by hydrogen-bonding.5. Thermorheologically complex behavior by hydrogen-bond clustering. Polym. Bull. 17, 431–438 (1987).
Hilger, C., Stadler, R. & Freitas, L. L. D. Multiphase thermoplastic elastomers by combination of covalent and association chain structures.2. Small-strain dynamic mechanical-properties. Polymer 31, 818–823 (1990).
Hilger, C. & Stadler, R. Cooperative structure formation by directed noncovalent interactions in an unipolar polymer matrix.7. differential scanning calorimetry and small-angle X-ray-scattering. Macromolecules 25, 6670–6680 (1992).
Hilger, C., Drager, M. & Stadler, R. Cooperative structure formation by combination of covalent and association chain polymers.6. Molecular-origin of supramolecular self-assembling in statistical copolymers. Macromolecules 25, 2498–2501 (1992).
Rieth, L. R., Eaton, R. F. & Coates, G. W. Polymerization of ureidopyrimidinone-functionalized olefins by using late-transition metal Ziegler-Natta catalysts: Synthesis of thermoplastic elastomeric polyolefins. Angew. Chem. Int. Ed. 40, 2153–2156 (2001).
Kretschman, O., Choi, S. W., Miyauchi, M., Tomatsu, I., Harada, A. & Ritter, H. Switchable hydrogels obtained by supramolecular cross-linking of adamantyl-containing LCST copolymers with cyclodextrin dimers13. Angew. Chem. Int. Ed. 45, 4361–4365 (2006).
Lecourt, T., Sinay, P., Chassenieux, C., Rinaudo, M. & Auzély-Velty, R. Complexation between a hydrophobically modified chitosan and cyclodextrin homodimers singly or doubly connected through their primary sides: effects of their molecular architecture on the polymer properties in solution. Macromolecules 37, 4635–4642 (2004).
Lecourt, T., Blériot, Y., Auzély-Velty, R. & Sollogoub, M. Cyclodextrin tetraplexes: first syntheses and potential as cross-linking agent. Chem. Commun. 46, 2238–2240 (2010).
Bistri-Aslanoff, O., Blériot, Y., Auzély-Velty, R. & Sollogoub, M. Duplex of capped-cyclodextrins, synthesis and cross-linking behaviour with a biopolymer. Org. Biomol. Chem. 8, 3437–3443 (2010).
Harada, A., Kobayashi, R., Takashima, Y., Hashidzume, A. & Yamaguchi, H. Macroscopic self-assembly through molecular recognition. Nat. Chem. 3, 34–37 (2011).
Oku, T., Furusho, Y. & Takata, T. A concept for recyclable cross-linked polymers: topologically networked polyrotaxane capable of undergoing reversible assembly and disassembly. Angew. Chem. Int. Ed. 43, 966–969 (2004).
Xu, H., Hampe, E. M. & Rudkevich, D. M. Applying reversible chemistry of CO2 to supramolecular polymers. Chem. Commun. 2828–2829 (2003).
Xu, H. & Rudkevich, D. M. CO2 in supramolecular chemistry: preparation of switchable supramolecular polymers. Chem. Eur. J. 10, 5432–5442 (2004).
Rudkevich, D. M. & Xu, H. Carbon dioxide and supramolecular chemistry. Chem. Commun. 2651–2659 (2005).
Xu, H. & Rudkevich, D. M. Reversible chemistry of CO2 in the preparation of fluorescent supramolecular polymers. J. Org. Chem. 69, 8609–8617 (2004).
Park, T. & Zimmerman, S. C. Interplay of fidelity, binding strength, and structure in supramolecular polymers. J. Am. Chem. Soc. 128, 14236–14237 (2006).
Park, T. & Zimmerman, S. C. Formation of a miscible supramolecular polymer blend through self-assembly mediated by a quadruply hydrogen-bonded heterocomplex. J. Am. Chem. Soc. 128, 11582–11590 (2006).
Haino, T., Hirai, E., Fujiwara, Y. & Kashihara, K. Supramolecular cross-linking of [60]fullerene-tagged polyphenylacetylene by the host-guest interaction of calix[5]arene and [60]fullerene. Angew. Chem. Int. Ed. 49, 7899–7903 (2010).
Cordier, P., Tournilhac, F., Soulie-Ziakovic, C. & Leibler, L. Self-healing and thermoreversible rubber from supramolecular assembly. Nature 451, 977–980 (2008).
Burnworth, M., Tang, L., Kumpfer, J. R., Duncan, A. J., Beyer, F. L., Fiore, G. L., Rowan, S. J. & Weder, C. Optically healable supramolecular polymers. Nature 472, 334–337 (2011).
Acknowledgements
Financial support was provided by Grant-in-Aids for Scientific Research (B) (No. 21350066) and Challenging Exploratory Research (No. 23655105) of the Japan Society of the Promotion of Science. This work was partially supported by the Yamada Science Foundation, the Electric Technology Research Foundation of Chugoku and the Izumi Science and Technology Foundation.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Haino, T. Molecular-recognition-directed formation of supramolecular polymers. Polym J 45, 363–383 (2013). https://doi.org/10.1038/pj.2012.144
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/pj.2012.144
Keywords
This article is cited by
-
Macromolecular architectures constructed by biscalix[5]arene–[60]fullerene host–guest interactions
Polymer Journal (2023)
-
The macroscopic shape of assemblies formed from microparticles based on host–guest interaction dependent on the guest content
Scientific Reports (2021)
-
Biofunctional hydrogels based on host–guest interactions
Polymer Journal (2020)
-
Designer supramolecular polymers with specific molecular recognitions
Polymer Journal (2019)
-
Sequence-controlled supramolecular terpolymerization directed by specific molecular recognitions
Nature Communications (2017)