Abstract
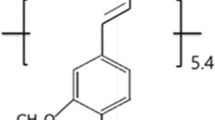
Organosolv lignin (OSL) alkyl esters (carbon number n, 2–5) were synthesized by homogeneous reaction with different acyl anhydrides/4-dimethylamino pyridine in pyridine solution. Miscibility of the esterified OSL with poly(ε-caprolactone) (PCL) was investigated through DSC, morphological observation, and spectroscopy. DSC revealed a composition-dependent shift in Tg for blends of OSL esters (n = 3–5) with PCL; thus these three systems can be regarded as miscible on a Tg-detection scale. AFM indicated that no remarkable phase separation occurred in these blends. In observations of crystallization behavior of the PCL component, the OSL derivatives of n = 4 and 5, the alkyl ester substituents of which make a higher similarity in chemical structure with a repeating unit of PCL, exhibited a higher level of miscibility with the aliphatic polyester, compared to the other OSL derivative of n = 3. Thermal-molded blend sheets of OSL esters of n = 3–5 with PCL attained ≥500% of elongation at rupture at 20 °C even when the blends contained 50 wt% of the respective esterified lignin components, reflecting the good miscibility of these OSL esters with PCL.
Similar content being viewed by others
Article PDF
References
A. E. Farrell, R. J. Plevin, B. T. Turner, A. D. Jones, M. O'Hare, and D. M. Kammen, Science, 311, 506 (2006).
N. Mosier, C. Wyman, B. Dale, R. Elander, Y. Y. Lee, M. Holtzapple, and M. Ladisch, Bioresour. Technol., 96, 673 (2005).
Y. Teramoto, S. H. Lee, and T. Endo, Bioresour. Technol., 99, 8856 (2008).
Y. Teramoto, N. Tanaka, S. H. Lee, and T. Endo, Biotechnol. Bioeng., 99, 75 (2008).
“Lignin: Properties and Materials,” W. G. Glasser and S. Sarkanen, Ed., American Chemical Society, Washington, DC, 1989.
S. L. Ciemniecki and W. G. Glasser, Polymer, 29, 1021 (1988).
S. L. Ciemniecki and W. G. Glasser, Polymer, 29, 1030 (1988).
D. Feldman, D. Banu, M. Lacasse, J. Wang, and C. Luchian, J. Macromol. Sci., Pure Appl. Chem., A32, 1613 (1995).
D. Hasegawa, Y. Teramoto, and Y. Nishio, J. Wood Sci., 54, 143 (2008).
J. F. Kadla and S. Kubo, Macromolecules, 36, 7803 (2003).
S. Kubo and J. F. Kadla, Biomacromolecules, 4, 561 (2003).
S. Kubo and J. F. Kadla, Macromolecules, 37, 6904 (2004).
S. Kubo and J. F. Kadla, J. Appl. Polym. Sci., 98, 1437 (2005).
S. Kubo and J. F. Kadla, Holzforschung, 60, 245 (2006).
J. C. Li, Y. He, and Y. Inoue, Polym. J., 33, 336 (2001).
Y. Li, J. Mlynar, and S. Sarkanen, J. Polym. Sci., Part B: Polym. Phys., 35, 1899 (1997).
Y. Li and S. Sarkanen, Macromolecules, 35, 9707 (2002).
Y. Li and S. Sarkanen, Macromolecules, 38, 2296 (2005).
W. Thielemans and R. P. Wool, Biomacromolecules, 6, 1895 (2005).
I. Ghosh, R. K. Jain, and W. G. Glasser, ACS Symp. Ser., 742, 331 (2000).
C. L. Chen, “Determination of methoxyl groups,” in “Methods in lignin chemistry,” S. Y. Lin and C. W. Dence, Ed., Springer-Verlag, Berlin/Heidelberg/New York, 1992, p 465.
M. Y. Balakshin, E. A. Capanema, C. L. Chen, and H. S. Gracz, J. Agric. Food Chem., 51, 6116 (2003).
P. M. Froass, A. J. Ragauskas, and J. E. Jiang, Holzforschung, 52, 385 (1998).
R. H. Marchessault, S. Coulombe, H. Morikawa, and D. Robert, Can. J. Chem.-Rev. Can. Chim., 60, 2372 (1982).
E. Tiainen, T. Drakenberg, T. Tamminen, K. Kataja, and A. Hase, Holzforschung, 53, 529 (1999).
D. S. Kaplan, J. Appl. Polym. Sci., 20, 2615 (1976).
L. A. Utracki, “Polymer Alloys and Blends,” Hanser, Munich, 1990.
Y. Nishio, “Cellulose Polymers, Blends, and Composites,” R. D. Gilbert, E., Hanser, Munich/New York, 1994, chap. 5.
Y. Miyashita, Y. Yamada, N. Kimura, H. Suzuki, M. Iwata, and Y. Nishio, Polymer, 38, 6181 (1997).
M. Avella and E. Martuscelli, Polymer, 29, 1731 (1988).
H. D. Keith, F. J. Padden, and T. P. Russell, Macromolecules, 22, 666 (1989).
T. Nishi and T. T. Wang, Macromolecules, 8, 909 (1975).
K. Schulze, J. Kressler, and H. W. Kammer, Polymer, 34, 3704 (1993).
S. Nojima, D. J. Wang, and T. Ashida, Polym. J., 23, 1473 (1991).
J. Xu, B. H. Guo, G. Q. Chen, and Z. M. Zhang, Polym. J., 35, 460 (2003).
Y. Teramoto and Y. Nishio, Biomacromolecules, 5, 397 (2004).
R. Kusumi, Y. Inoue, M. Shirakawa, Y. Miyashita, and Y. Nishio, Cellulose, 15, 1 (2008).
Y. Nishio, Adv. Polym. Sci., 205, 97 (2006).
Y. Nishio, K. Matsuda, Y. Miyashita, N. Kimura, and H. Suzuki, Cellulose, 4, 131 (1997).
L. E. Nielsen, “Mechanical Properties of Polymers and Composites,” Marcel Dekker, New York, 1974, p303.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Teramoto, Y., Lee, SH. & Endo, T. Phase Structure and Mechanical Property of Blends of Organosolv Lignin Alkyl Esters with Poly(ε-caprolactone). Polym J 41, 219–227 (2009). https://doi.org/10.1295/polymj.PJ2008301
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1295/polymj.PJ2008301
Keywords
This article is cited by
-
Catalytic esterification reactions of model lignin phenols towards hydrophobicity
Biomass Conversion and Biorefinery (2022)
-
Injection-Molded Bioblends from Lignin and Biodegradable Polymers: Processing and Performance Evaluation
Journal of Polymers and the Environment (2018)
-
Biodegradable blends of stereocomplex polylactide and lignin by supercritical carbon dioxide-solvent system
Macromolecular Research (2014)
-
Poly(Lactic Acid) Blended with Cellulolytic Enzyme Lignin: Mechanical and Thermal Properties and Morphology Evaluation
Journal of Polymers and the Environment (2012)
-
Conversion of sulfuric acid lignin generated during bioethanol production from lignocellulosic materials into polyesters with ɛ-caprolactone
Journal of Wood Science (2011)