Abstract
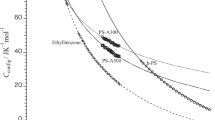
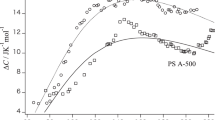
The osmotic compressibility up to high concentrations as well as the second virial coefficient were measured for low molecular weight polystyrenes dissolved in a poor solvent cyclohexane at 35, 25, and 15 °C, by sedimentation equilibrium. The results of the osmotic compressibility over wide concentration ranges were favorably compared with a recently developed thermodynamic perturbation theory based on the spherocylinder model bearing a square-well potential, and from the comparison, the hard-core diameter d and the depth ε of the attractive square-well potential including in the theory were determined for polystyrene in cyclohexane. Compared with the previous results of d and ε for the same polymer in 15 °C toluene (a good solvent), it turned out that ε increases and d decreases with reducing the solvent quality.
Similar content being viewed by others
Article PDF
References
H. Yamakawa, “Modern Theory of Polymer Solutions,” Harper & Row, New York, N.Y., 1971.
H. Fujita, “Polymer Solutions,” Vol 9, Elsevier, Amsterdam, 1990.
H. Yamakawa, “Helical Wormlike Chains in Polymer Solutions,” Springer-Verlag, Berlin and Heidelberg, 1997.
P.-G. de Gennes, “Scaling Concepts in Polymer Physics,” Cornell Univ. Press, Ithaca, N.Y., 1979.
Y. Oono, Adv. Chem. Phys., 61, 301 (1985).
K. F. Freed, “Renormalization Group Theory of Macromolecules,” John Wiley & Sons, New York, N.Y., 1987.
J. des Cloizeaux and G. Jannink, “Polymers in Solution. Their Modelling and Structure,” Clarendon Press, Oxford, U.K., 1990.
Y. Nakamura, T. Norisuye, and A. Teramoto, Macromolecules, 24, 4904 (1991).
I. Noda, Y. Higo, N. Ueno, and T. Fujimoto, Macromolecules, 17, 1055 (1984).
R. Koyama and T. Sato, Macromolecules, 35, 2235 (2002).
T. G. Scholte, Eur. Polym. J., 6, 1063 (1970).
T. G. Scholte, J. Polym. Sci., Part A-2: Polym. Phys., 8, 841 (1970).
T. G. Scholte, J. Polym. Sci., Part A-2: Polym. Phys., 9, 1553 (1971).
Z. Tong, S. Ohashi, Y. Einaga, and H. Fujita, Polym. J., 15, 835 (1983).
Y. Einaga, S. Ohashi, Z. Tong, and H. Fujita, Macromolecules, 17, 527 (1984).
Y. Einaga, F. Abe, and H. Yamakawa, Macromolecules, 26, 6243 (1993).
H. Yamakawa, F. Abe, and Y. Einaga, Macromolecules, 27, 5704 (1994).
H. Fujita, “Foundation of Ultracentrifugal Analysis,” Wiley-Interscience, New York, N.Y., 1975.
M. Kurata, “Thermodynamics of Polymer Solutions,” Harwood Academic Publishers, Chur, 1982.
There are errors in the previous paper. In eq 2.4, B = (π/2)Lc2dρ; eqs 2.6 and 2.8, the excess free energies should be divided by kBT; in eq 2.9, bme = 3Lcd2/2; in eq 2.11, the term with C should be multiplied by 2; in eq 2.14, δ3 should be replaced by δ3; in eqs 2.16 and 2.17, 3εme should be replaced by 6εme; in eq 2.19, 108 should be replaced by 216; in Table 3, εme/kBT = 2π(4δ+3δ′)/[36P(1)d + π(4δ + 3δ′)] = −0.17, δ′3 = 1.4 nm, and δ″3 = −1.9 nm.
J. A. Barker and D. Henderson, Rev. Mod. Phys., 48, 587 (1976).
H. Yamakawa, Macromolecules, 25, 1912 (1992).
H. Yamakawa and T. Yoshizaki, J. Chem. Phys., 119, 1257 (2003).
H. Yamakawa, F. Abe, and Y. Einaga, Macromolecules, 27, 3272 (1994).
Y. Nakamura, N. Inoue, T. Norisuye, and A. Teramoto, Macromolecules, 30, 631 (1997).
H. Yamakawa, J. Chem. Phys., 45, 2606 (1966).
T. Norisuye and Y. Nakamura, Macromolecules, 27, 2054 (1994).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Oribe, M., Sato, T. Repulsive and Attractive Interactions between Polystyrene Chains in a Poor Solvent. Polym J 36, 747–753 (2004). https://doi.org/10.1295/polymj.36.747
Published:
Issue Date:
DOI: https://doi.org/10.1295/polymj.36.747