Abstract
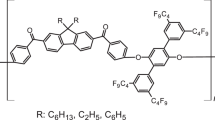
A series of poly(aryl ethers), derived from (3-methylphenyl)hydroquinone, (3-trifluoromethylphenyl)hydroquinone and (3,5-ditrifluoromethylphenyl)hydroquinone, were synthesized via a typical aromatic nucleophilic substitution reaction. The polycondensation quantitatively proceeded in the presence of anhydrous potassium carbonate to afford the polymers with an M̅n above 19000. The glass transition temperatures of the polymers ranged from 143 °C to 178 °C. The polymers showed high thermal decomposition temperatures, and the temperatures at which a 5% weight loss occurred were above 443 °C. Compared with the methylated polymers, the trifluoromethylated polymers exhibited a higher thermal stability. All the polymers showed good solubility due to the existence of pendant groups. Transparent and flexible thin polymer films could easily be obtained by solution casting. The dielectric constants of the polymers estimated from the refractive indices (εopt= 1.1 n2) were 2.45–2.96. Because of the strong hydrophobicity of the fluorine-containing groups, the water uptakes of the polymers decreased with an increase in the fluorine-containing content.
Similar content being viewed by others
Article PDF
References
R. J. Cotter, “Engineering Plastics: A Handbook of Polyarylethers”, Golden and Breach Publishers, Amsterdam, 1995.
A. A. Goodwin, F. W. Mercer, and M. T. McKenzie, Macromolecules, 30, 2767 (1997).
V. L. Rao, A. Saxena, and K. N. Ninan, J. Macromol. Sci., Polym. Rev., C42, 513 (2002).
P. E. Cassidy, T. M. Aminabhavi, and J. M. Farley, J. Macromol. Sci., Rev. Macromol. Chem. Phys., 29, 365 (1989).
M. L. Minges, “Electronic Materials Handbook”, ASM International, Materials Park, OH, 1989.
G. Maier, Prog. Polym. Sci., 26, 3 (2001).
S. H. Hsiao and C. T. Lee, J. Polym. Sci., Part A: Polym. Chem., 37, 1403 (1999).
H. Seino, A. Mochizuki, and M. Ueda, J. Polym. Sci., Part A: Polym. Chem., 37, 3584 (1999).
T. Takasaki, Y. Kuwana, T. Takahashi, and S. Hayashida, J. Polym. Sci., Part A: Polym. Chem., 38, 4832 (2000).
F. Mercer, T. Goodman, J. Wojtowicz, and D. J. Duff, J. Polym. Sci., Part A: Polym. Chem., 30, 1767 (1992).
H. Zhou, J. Liu, Z. Qian, S. Zhang, and S. Yang, J. Polym. Sci., Part A: Polym. Chem., 39, 2404 (2001).
H. Lee, E. Lee, M. Lee, M. Oh, J. Ahn, S. Han, and H. Kim, J. Polym. Sci., Part A: Polym. Chem., 36, 2881 (1998).
M. Kakimoto, S. Harada, Y. Oishi, and Y. Imai, J. Polym. Sci., Polym. Chem. Ed., 25, 2747 (1987).
B. Silverman, P. Sanda, P. Ho, and A. Rossi, J. Polym. Sci., Polym. Chem. Ed., 23, 2857 (1985).
L. Tan and F. Arnold, J. Polym. Sci., Polym. Chem. Ed., 25, 3159 (1987).
S. Ando, T. Matsuura, and S. Sasaki, Macromolecules, 25, 5858 (1992).
F. Liu, J. Ding, M. Li, M. Day, G. Robertson, and M. Zhou, Macromol. Rapid Commun., 23, 844 (2002).
S. Banerjee and G. Maier, Chem. Mater., 11, 2179 (1999).
B. J. Liu, G. B. Wang, W. Hu, Y. H. Jin, C. H. Chen, Z. H. Jiang, W. J. Zhang, Z. W. Wu, and Y. Wei, J. Polym. Sci., Part A: Polym. Chem., 40, 3392 (2002).
J. A. Irvin, C. J. Neef, K. M. Kane, P. E. Cassidy, G. Tullos, and A. K. St. Clair, J. Polym. Sci., Part A: Polym. Chem., 30, 1675 (1992).
G. Hougham, G. Tesoro, A. Viehbeck, and J. Chapple-Sokol, Macromolecules, 27, 5964 (1994).
T. Matsumoto, “Advances in Low-k Dielectrics And Thermally Stable Polymers for Microelectronics”, Society of Plastics Engineers, Mid Hudson Section, New York, N.Y., 2002, p 121.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Liu, B., Hu, W., Zhao, S. et al. Methylated and Trifluoromethylated Poly(aryl ethers). Polym J 35, 628–633 (2003). https://doi.org/10.1295/polymj.35.628
Issue Date:
DOI: https://doi.org/10.1295/polymj.35.628