Abstract
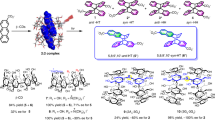
N′-tert-Butoxycarbonylmonoamino acid-binding β-CDs (1,2,3,4,5) and α-CD (6) were prepared by DCC coupling. NMR study suggests some of these novel modified CDs that act as host and guest to prefer “pseudo polymer” formation. The length of an arm between the N′-tert-butoxycarbonyl group and C6 position on the glucose ring was that of -NH-Cα-CO-NH-. Modified β-CDs having longer arm form intramolecular rather than intermolecular complexes.
Similar content being viewed by others
Article PDF
References
V. T. D’Souza and K. B. Lipkowitz, Ed., “Cyclodextrins”, Ckem. Rev., 98 (1998).
S. A. Nepogodiev and J. F. Stoddart, Chem. Rev., 98, 1956 (1998).
T. J. Hubin, A. G. Kolchinski, A. L. Vane, and D. H. Busch, ‘Template Control of Supramolecular Architecture’, in “Advances in Supramolecular Chemistry”, G. W. Gokel, Ed., JAI Press Inc., London, 1999, 5, p 237.
A. Harada, J. Li, and M. Kamachi, Macromolecules, 23, 2821 (1990).
A. Harada and M. Kamachi, J. Chem. Soc., Chem. Commun., 1322 (1990).
A. Harada, J. Li, and M. Kamachi, Nature (London), 356, 325 (1992).
A. Harada and M. Kamachi, Nature (London), 370, 126 (1994).
M. Born and H. Ritter, Adv. Mater., 8, 149 (1996).
I. Yamaguchi, K. Osakada, and T. Yamamoto, Macromolecules, 30, 4288 (1997).
M. Born, T. Koch, and H. Ritter, Macromol. Chem. Phys., 8, 1761, (1995).
M. Born and H. Ritter, Angew. Chem., Int. Ed. Engl., 35, 309 (1995).
M. Born, T. Koch, and H. Ritter, Acta Polym., 45, 68 (1994).
A. Lütringhaus, F. Cramer, H. Prinzbach, and F. M. Henglein, Leibigs Ann. Chem., 62, 527 (1958).
D. Armspach, P. R. Ashton, C. P. Moore, N. Spencer, J. F. Stoddart, T. J. Wear, and D. J. Williams, Angew. Chem., Int. Ed. Engl., 32, 854 (1993).
D. Armspach, P. R. Ashton, N. Spencer, J. F. Stoddart, and D. J. Williams, Pesticide Sci., 41, 232 (1994).
D. Armspach, P. R. Ashton, R. Ballardini, V. Balzani, A. Godi, C. P. Moore, L. Prodi, N. Spencer, J. F. Stoddart, M. S. Tolley, and D. J. Williams, Chem. Eur. J., 1, 33 (1995).
H. Murakami, A. Kawabuchi, K. Kotoo, M. Kunitake, and N. Nakajima, J. Am. Chem. Soc., 119, 7605 (1997).
H. Fujita, T. Ooya, and N. Yui, Macromolecules, 32, 2534 (1999).
S. Kamitori, K. Hirotsu, T. Higuchi, K. Fujita, H. Yamamura, T. Imoto, and I. Tabushi, J. Chem. Soc., Perkin Trans. 2, 1987, 7.
M. Dimitrius, A. Terzis, A. W. Coleman, and C. de Rango, Carbohydr. Res., 282, 125 (1996).
K. Takahashi, Y. Ohtsuka, and K. Hattori, Chem. Lett., 2227 (1990).
W. Saka, Y. Inoue, Y. Yamamoto, R. Chujo, K. Takahashi, and K. Hattori, Bull. Chem. Soc. Jpn., 63, 3175 (1990).
K. Takahashi, Y. Ohtsuka, S. Nakada, and K. Hattori, J. Incl. Phenomn. Mol. Recogn. Chem., 10, 63 (1991).
K. Takahashi, J. Chem. Soc. Chem. Commun., 1991, 929.
M. Akiyama, A. Kato, J. Kato, K. Takahashi, and K. Hattori, Chem. Lett., 1189 (1991).
K. Takahashi, Bull. Chem. Soc. Jpn., 66, 540 (1993).
K. Takahashi and K. Hattori, Supramol. Chem., 2, 305 (1993).
K. Takahashi and R. Furusho, Polym. J., 28, 458 (1996).
F. D-Pilard, N. A-Bellanger, M. Gonsnat, D. Vernet, and B. Perly, J. Chem. Soc., Perkin Trans. 2, 1995, 723.
T. Kuwabara and A. Ueno, Supramol. Chem., 7, 235 (1996).
C. S. Wilcox, ‘Design, Synthesis, and Evaluation of an Efficacious Functional Group Dyad. Methods and Limitations in the Use of NMR for Measuring Host-Guest Interactions’, in “Frontiers in Supramolecular Organic Chemistry and Photochemistry”, H-J. Schneider and H. Dürr, Ed., VCH, Weinheim, 1991, p 123.
A. Harada, H. Adachi, Y. Kawagachi, and M. Kamachi, Macromolecules, 30, 5181 (1997).
A. P. Demchenko, “Ultravioret Spectroscopy of Proteins”, Springer-Verlag, London, 1981.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Takahashi, K., Imotani, K. & Kitsuta, M. Formation of Superstructure Composed of Modified Cyclodextrins as Molecular “Blocks” in Aqueous Solution with Host-Guest Complexation. Correlation of Chemical Structure of Modified Group with Complexation. Polym J 33, 242–247 (2001). https://doi.org/10.1295/polymj.33.242
Issue Date:
DOI: https://doi.org/10.1295/polymj.33.242