Abstract
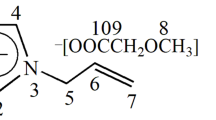
N-Methyl morpholine N-oxide (NMMO) hydrates, used in the preparation of cellulose spinning dope for lyocell manufacturing, have various crystal structures according to the hydration number (n). A single melting peak for solid-solid transition from NMMO monohydrate to anhydrous NMMO at 94°C was previously reported. However, we observed the melting peak at 90°C ascribed to the solid-solid transition from NMMO monohydrate to anhydrous NMMO and another melting peak observed in the range from 98 to 105°C when n<1. This discrepancy resulted from the experimental errors in differential scanning calorimetry (DSC) measurement. The pressure capsule enabled us to obtain precise and reproducible DSC data of NMMO hydrates. The three different crystal structures of NMMO hydrates with n<1 were identified by crystallization experiments. The crystal, which had melted in the range from 90 to 105°C during heating (shown by double peaks), was recrystallized at 80°C during cooling, which has never been reported. The NMMO hydrates with n<1 required heating above 130°C to produce a homogeneous melt, and the required temperature was increased up to 180°C for anhydrous NMMO. If the heating temperature was not high enough to fully melt the NMMO hydrate crystal, the unmelted NMMO hydrate crystals affected the thermal responses of the hydrate and its phase behavior of cellulose solutions in the NMMO hydrate.
Similar content being viewed by others
Article PDF
References
G. Graenacher and R. Sallmann, U. S. Patent, 2 179 181 (Nov. 7, 1939).
D. L. Johnson, U. S. Patent, 3 447 939 (Jun. 3, 1969).
D. L. Johnson, U. S. Patent, 3 508 941 (Apr. 28, 1970).
C. C. McCorsley and J. K. Varga, U. S. Patent, 4 142 913 (Mar. 6, 1979).
N. E. Franks and S. K. Varga, U. S. Patent, 4 145 532 (Mar. 20, 1979).
N. E. Franks and S. K. Varga, U. S. Patent, 4 196 282 (Apr. 1, 1980).
C. C. McCorsley, U. S. Patent, 4 246 221 (Jan. 20, 1981).
H. Chanzy, M. Dube, and R. H. Marchessault, J. Polym. Sci., Polym. Lett. Ed., 17, 219 (1978).
H. Chanzy, A. Peguy, S. Chaunis, and P. Monzie, J. Polym. Sci., Polym. Phys. Ed., 18, 1137 (1980).
H. Chanzy, S. Nawrot, A. Peguy, and P. Smith, J. Polym. Sci., Polym. Phys. Ed., 20, 1909 (1982).
E. Maia, A. Peguy, and S. Perez, Acta Crystallogr., B: Struct. Sci., 37, 1858 (1981).
E. Maia and S. Perez, Acta Crystallogr., B: Struct. Sci., 38, 849 (1982).
D. Gagnaire, D. Mancier, and M. Vincendon, J. Polym. Sci., Polym. Chem. Ed., 18, 1137 (1980).
D. L. Patel and R. D. Gilbert, J. Polym. Sci., Polym. Phys. Ed., 19, 1231 (1981).
P. Navard and J. M. Haudin, Br. Polym. J., 22, 174 (1980).
B. C. Kim, W. S. Lee, S. M. Jo, C. S. Park, D. B. Kim, and Y. M. Lee, Polym. Mater. Sci. and Eng., 77, 475 (1997).
D. B. Kim, W. S. Lee, and H. J. Kang, Polymer (Korea), 22, 770 (1998).
D. B. Kim, W. S. Lee, S. M. Jo, Y. M. Lee, and B. C. Kim, Polym. J., 33, 18 (2001).
D. Loubinoux and S. Chauis, Text. Res. J., 57, 61 (1987).
P. Navard and J. M. Haudin, Polym. Pro. Eng., 3, 291 (1985).
H. Chanzy, M. Paillet, and R. Hagege, Polymer, 31, 400 (1990).
S. W. Chun, S. M. Jo, W. S. Lee, and J. D. Kim, J. Kor. Fiber. Soc., 29, 44 (1992).
D. B. Kim, W. S. Lee, B. C. Kim, S. M. Jo, J. S. Park, and Y. M. Lee, Polymer (Korea), 22, 231 (1998).
K. Fischer, Angew. Chem., 48, 394 (1935).
H. Chanzy, Carbohydrate Polymers, 2, 229 (1982).
E. A. Turi, “Thermal Characterization of Polymeric Materials”, Academic Press, Inc., New York, N.Y., 1997, vol. 1, p 415.
B. Wunderlich, “Macromolecular Physics”, Academic Press, Inc., New York, N.Y., 1976, vol. 2, p 348.
S. H. Park, Y. H. Jeong, W. S. Lee, and H. J. Kang, Polymer (Korea), 22, 779 (1998).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kim, D., Lee, W., Jo, S. et al. Effect of Thermal History on the Phase Behavior of N-Methyl Morpholine N-Oxide Hydrates and Their Solutions of Cellulose. Polym J 33, 139–146 (2001). https://doi.org/10.1295/polymj.33.139
Issue Date:
DOI: https://doi.org/10.1295/polymj.33.139