Abstract
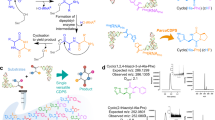
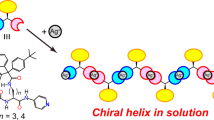
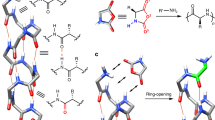
A cyclic hexapeptide cyclo(D-Leu-L-Glu-L-His)2 was synthesized and its conformational properties in (CD3)2SO and D2O were investigated. The major conformation in (CD3)2SO was asymmetric and possessed either a gramicidine-type β-turn structure or a random structure in one molecule. The major conformation in D2O was a C2-symmetric random structure. When Cu(ClO4)2 was added to an aqueous solution of cyclo(D-Leu-L-Glu-L-His)2 (pH 6.95, phosphate buffer), the major conformation of the latter changed from a random to a type-II β-turn structure. The cyclic hexapeptide/Cu2+ molar ratio of the complex was unity, and Cu2+ was coordinated with Glu-COOH. Induced by copper-ion coordination the backbone plane, the Leu-isobutyl group, and the His-imidazolylmethyl group of cyclo(D-Leu-L-Glu-L-His)2 had the form of a hydrophobic pocket. The highly efficient hydrolysis of leucine or valine p-nitrophenyl ester hydrochloride by the cycloD-Leu-L-Glu-L-His)2–Cu2+ complex may be closely related to the binding of a substrate by the hydrophobic pocket. The weak enantiomer-selectivity may also be related to a streochemical fit of a bound substrate with the hydrophobic pocket.
Similar content being viewed by others
Article PDF
References
M. Tanihara and Y. Imanishi, Polym. J., 15, 499 (1983).
Y. Chao and D. J. Cram, J. Am. Chem. Soc., 98, 1015 (1976).
Y. Chao, G. R. Weisman, G. D. Y. Sogah, and D. J. Cram, J. Am. Chem. Soc., 101, 4948 (1979).
J.-M. Lehn and C. Sirlin, J. Chem. Soc., Chem. Commun., 949 (1978).
K. Wüthrich, “NMR in Biological Research: Peptides and Proteins,” North Halland, Amsterdam, 1976, p 51.
G. M. Ramachandran, R. Chandrasekaran, and K. D. Kopple, Biopolymers, 10, 2113 (1971).
C. A. Venkatachalam, Biopolymers, 6, 1425 (1968).
E. R. Blout, “Fundamental Aspects and Recent Developments in Optical Rotatory Dispersion and Circular Dichroism,” F. Ciardelli and P. Salvadori, Ed., Heyden, London, 1973, pp 352—372.
C. A. Bush, S. K. Sarkar, and K. D. Kopple, Biopolymers, 17, 4951 (1978).
T. Sugihara, Y. Imanishi, T. Higashimura, and Y. Shimonishi, J. Org. Chem., 41, 2584 (1976).
T. A. Steitz, R. Henderson, and D. M. Blow, J. Mol Biol., 46, 337 (1969).
Y. Murakami, A. Nakano, K. Matsumoto, and K. Iwamoto, Bull. Chem. Soc. Jpn., 51, 2690 (1978).
M. Tanihara, Y. Kikuchi, and Y. Imanishi, Int. J. Biol. Macromol., 4, 297 (1982).
Y. Kikuchi, M. Tanihara, and Y. Imanishi, Int. J. Biol. Macromol., 4, 305 (1982).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Tanihara, M., Imanishi, Y. Solution Conformation of Cyclo(D-Leu-L-Glu-L-His)2 and Its Relation with Highly Efficient and Enantiomer-Selective Catalysis. Polym J 15, 509–518 (1983). https://doi.org/10.1295/polymj.15.509
Issue Date:
DOI: https://doi.org/10.1295/polymj.15.509