Abstract
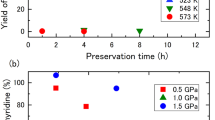
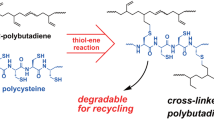
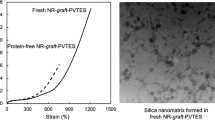
The kinetics of nitration of cis-1,4-polybutadiene rubber dissolved in dichloroethane in the range of concentrations up to 2.2% was studied in a stirred cell. It was found that diffusion of nitrogen dioxide is not important and the reaction is of the first order with respect to both reactants. The overall second-order reaction rate constant was found to be about 1.1×10−5l/(gmol sec) at 303K. The obtained value for the activation energy corresponds to kinetic regime. Based on these results a reaction mechanism is proposed.
Similar content being viewed by others
Article PDF
References
I. Mladenov, D. Sc. Thesis, Chemistry Technology Institute Sofia, 1975.
H. H. G. Jellinek and J. Toyoshima, J. Polym. Sci., Part A-1, 5, 3214 (1967).
H. H. G. Jellinek, F. Flaysman, and F. G. Kryman, J. Appl. Polym. Sci., 13, 107 (1969).
M. M. Sharma and P. V. Danckwerts, Chem. Eng. Sci., 19, 991 (1964).
B. Stojanove-Ivanova, Short Course of Organic Analysis, Nauka i Izkustvo, Sofia, 1967, p 191.
J. Royo, L. Gonzales, L. Ibarra, and M. Barbero, Makromol. Chem., 168, 41 (1973).
A. S. Jhaveri and M. M. Sharma, Chem. Eng. Sci., 22, 1 (1967).
G. Charlo, “Methods of Analytical Chemistry, Russian Translation,” Khimia, Moscow, 1965, p 551.
D. A. Frank-Kamenetzkii, “Diffusion and Heat Transfer in Chemical Kinetics,” Nauka, Moscow, 1951, p 124.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Mladenov, I., Kostov, G. & Rashev, V. Kinetics of Nitration of cis-1,4-Polybutadiene Rubber in Solution with Nitrogen Dioxide. Polym J 10, 451–455 (1978). https://doi.org/10.1295/polymj.10.451
Issue Date:
DOI: https://doi.org/10.1295/polymj.10.451