Abstract
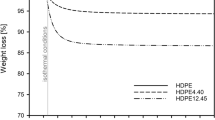
The thermal properties (viz., second-order transitions, melting points, dynamic and isothermal heat behaviour) of chlorinated polyethylene (CPE) samples are studied here. Thermomechanical analysis (TMA), differential thermal analysis (DTA), and thermogravimetric analysis (TGA) results are listed together to correlate the thermal properties and the microstructure of CPE samples, of various chlorine contents by weight, produced from thermal chlorinated or photochlorinated linear and branched polyethylene samples. The dehydrochlorination rate of a CPE sample is shown to be strongly dependent on its microtacticity up to a 56—61-% chlorine content by weight value. Beyond this 56—61-%Cl2 interval the microtacticity influence is screened by the steric hindrance introduced by the αI (—CH2—C<u>H</u>Cl—CCl2— or —CHCl—C<u>H</u>Cl—CCl2—), αII —CHCl—C<u>H</u>Cl—CHCl— or —CH2—C<u>H</u>Cl—CHCl—), and βII (—CHCl—C<u>H</u>2—CCl2—) structures. At the saturation level (73-% Cl2), the higher a CPE ratio, nb CHCl/nb CH2, is, the lower is its maximum dehydrochlorination rate. When its ratio, nb2 CHCl/nb CH2, is less than 4, a saturated (73%) CPE sample has a unique global dehydrochlorination activation energy (Ea); when this ratio stands between 4 and 6, there are two global dehydrochlorination Ea. The structures which could be regarded as responsible for these different global Ea and the second-order transitions are specified.
Similar content being viewed by others
Article PDF
References
H. J. Oswald and E. T. Kubu, SPE, Trans., 3, 168 (1963).
V. A. Erä and J. J. Lindberg, J. Polym. Sci., Part A-2, 10, 937 (1972).
I. A. Abu Isa, J. Polym. Sci., Part A-2, 10, 881 (1972).
N. Saglio, P. Berticat, and G. Vallet, J. Appl. Polym. Sci., 16, 2991 (1972).
B. M. Quenum, P. Berticat, and G. Vallet, Polym. J., 7, 287 (1975).
B. M. Quenum, P. Berticat, and G. Vallet, Polym. J., 7, 277 (1975).
B. M. Quenum, P. Berticat, and Q. T. Pham, Eur. Polym. J., 7, 1527 (1971).
B. M. Quenum, P. Berticat, and Q. T. Pham, Eur. Polym. J., 9, 777 (1973).
G. Humbert, B. M. Quenum, P. Berticat, Q. T. Pham, and G. Vallet, Makromol. Chem., 175, 1597 (1974).
G. Humbert, B. M. Quenum, P. Berticat, and G. Vallet, Makromol. Chem., 175, 1611 (1974).
T. F. Schatski, J. Polym. Sci., 57, 496 (1962).
R. F. Boyer, Rubber Chem. Tech., 36, 1303 (1963).
W. W. Wendlandt, “Thermal Methods of Analysis, Interscience, New York, N.Y., 1964.
A. Guyot and M. Bert, J. Appl. Polym. Sci., 17, 753 (1973).
J. D. Hoffman and B. M. Axilrod, J. Res. Nat. Bur. Stds, 54, 357, RP 2598 (1955).
J. Millan, M. Caransa, and J. Gusman, IUPAC Symposium on Macromolecules, Helsinki 1972, Preprint IV, 21, Vol. 5.
E. J. Arlman, J. Polym. Sci., 12, 543 (1959).
B. Baum and L. H. Wartman, J. Polym. Sci., 28, 537 (1968).
B. Baum and M. Thallmaier, Kunststoffe, 56, 80 (1966).
P. Berticat, J. J. Bejat, and G. Vallet, J. Chim. Phys., 67, 164 (1970).
P. Berticat, J. J. Bejat, J. Chim. Phys., 67, 170 (1970).
P. Berticat, J. J. Bejat, J. F. May, and G. Vallet, J. Chim. Phys., 67, 176 (1970).
N. Murayama and Y. Amagi, Polym. Lett., 4, 119 (1966).
A. V. Tobolsky, “Properties and Structure of Polymers, Wiley and Sons, New York, N.Y., 1960.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Quenum, BM., Berticat, P. & Vallet, G. Chlorinated Polyethylene. III. Relationships of Microstructure and Thermal Properties. Polym J 7, 300–311 (1975). https://doi.org/10.1295/polymj.7.300
Issue Date:
DOI: https://doi.org/10.1295/polymj.7.300
Keywords
This article is cited by
-
Thermal behavior and characterization of solid-state chlorinated polyethylenes
Journal of Thermal Analysis and Calorimetry (2006)