Abstract
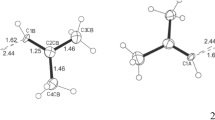
The electronic structure and the energy of CH3Li and its dimer have been calculated using a CNDO/2-method. The geometry of a planar rhombus was adopted for (LiCH3)2. Variation of the rhombus dimensions led to equilibrium distances of Li—Li=1.65 Å and C—C=3.85 Å. The dimerization energy was equal to 430 kcal/ mol. Dimerization decreased the energy of all molecular orbitals including the frontier ones, i.e., it increased the acidity. The calculations for the complexes of LiCH3 with formaldehyde and acrylonitrile were carried out for various relative positions of the components. Sometimes the energy minimum was found when LiCH3 was located out of the plane of the Lewis base near to C atom which is adjacent to the heteroatom. The complexation energies of LiCH3 with acrolein, vinyl formate and methyl acrylate were also calculated and found to be 120, 80 and 140 kcal/mol respectively. It corroborates the assumption that conjugated bases should form with organometallics complexes of higher stability than the unconjugated ones (the complexation energy with formaldehyde was equal to 100 kcal/mol). The complexation energy with acrylonitrile was found to be 180 kcal/mol. This correlates with the higher activity of this monomer when compared to methyl acrylate in anionic systems and correlates with the lower activity of acrylonitrile compared to methyl acrylate if they are in the same system but in the presence of a small amount of strong Lewis base competing for the complexing center.
Similar content being viewed by others
Article PDF
References
Yu. Ye. Eizner, S. S. Skorokhodov, and T. P. Zubova, Eur. Polym. J., 7, 869 (1971).
B. L. Erussalimsky, IUPAC Intern. Symp. on Macrornol. Chem. Plenary and Main Lect., Akad. Kiadó, Budapest, 1971, p. 281.
B. L. Erussalimsky, V. N. Krasulina, and Yu. Ye. Eizner, Vysokomol. Soedin., Ser. B, 12, 327 (1970).
B. L. Erussalimsky, Ved N. Krasulina, and I. L. Artamonova, V Intern. Conference on Organomet. Chem., Moscow, 1971, 1, 147.
L. V. Zamoiskaya, S. I. Vinogradova, and E. B. Milovskaya, Vysokomol. Soedin., Ser. A, 13, 1484 (1971).
K. Starovieyski, S. Pasynkiewicz, and E. Jankowska, J. Organometal. Chem., 22, 283 (1970).
H. Lewis and T. L. Brown, J. Am. Chem. Soc., 92, 4664 (1970).
M. Morton, L. J. Fetters, and R. A. Pett, Macromolecules, 3, 327 (1970).
Ch. B. Tsvetanov, V. N. Zgonnik, B. L. Erussalimsky, and I. M. Panaiotov, Vysokomol. Soedin., Ser. B, 14, 404 (1972).
Ch. B. Tsvetanov, V. N. Zgonnik, B. L. Erussalimsky, and I. M. Panaiotov, Lieb. Ann., 763, 53 (1972).
J. A. Pople, D. P. Santry, and G. A. Segal, J. Chem. Phys., 43, S 130, (1965).
J. A. Pople and G. A. Segal, J. Chem. Phys., 43, S 136 (1965).
J. A. Pople and G. A. Segal, J. Chem. Phys., 44, 3289 (1966).
J. A. Pople and D. L. Beveridge, “Approximate Molecular Orbital Theory,” McGraw-Hill, New York, N. Y., 1970.
R. B. Davidson, W. L. Jorgenson, and L. C. Allen, J. Am. Chem. Soc., 92, 749 (1970).
H. Betsuyaki, J. Chem. Phys., 50, 3118 (1969).
W. H. Hehre and J. A. Pople, J. Am. Chem. Soc., 92, 2191 (1970).
H. Imamura, K. Morio, M. Oki, and T. L. Kunii, Tetrahedran Lett., 4575 (1970).
L. E. Sutton, “Interatomic Distances,” Spec. Publ. No. 11, Chem. Soc., London, 1958.
L. Andrews, J. Chem. Phys., 47, 4834 (1967).
H. Lewis and T. L. Brown, J. Am. Chem. Soc., 92, 4664 (1970).
W. B. Pearson, Canad. J. Phys., 32, 708 (1954).
E. A. Owen and G. I. Williams, Proc. Phys. Soc., 67, 895 (1954).
J. Krug, H. Witte, and E. Wölfel, Z. Phys. Chem. (N.F.), 4, 36 (1955).
L. Pauling, “The Nature of the Chemical Bond,” Cornell Univ. Press Ithaca, N. Y. 1960.
G. R. Peyton and W. H. Glaze, Theor. Chim. Acta (Berl.), 13, 259 (1969).
A. H. Cowley and W. D. White, J. Am. Chem. Soc., 91, 34, (1969).
G. A. Segal, J. Chem. Phys., 47, 1899 (1967).
H. Dietrich, Acta Crystallogr., 16, 681 (1963).
P. A. Kolmar, J. F. Liebman, and L. C. Allen, J. Am. Chem. Soc., 92, 1142 (1970).
W. N. Lipscomb, “Boron Hydrides,” New York, N.Y. 1963.
L. M. Seitz and T. L. Brown, J. Am. Chem. Soc., 88, 2174 (1966).
L. D. McKeever, R. Waack, M. A. Doran, and E. B. Baker, J. Am. Chem. Soc., 90, 3244 (1968).
L. D. McKeever, R. Waack, M. A. Doran, and E. B. Baker, J. Am. Chem. Soc., 91, 1057 (1969).
T. L. Brown, L. M. Seitz, and B. Y. Kimura, J. Am. Chem. Soc., 90, 3245 (1968).
T. A. Koopmans, Physica, 1, 104 (1934).
B. J. Nicholson, Adv. Chem. Phys., 18, 249 (1970).
K. Takaji and T. Oka, J. Phys. Soc. Jpn., 18, 1174 (1963).
N. P. Borisova, G. B. Erussalimsky, and Yu. Ye. Eizner, Zh. Strukt. Khim., 14, 855 (1973).
C. A. Coulson, “Valence,” Oxford Univ. Press, 1961.
R. Wagner, J. Fine, J. W. Simmons, and J. H. Goldstein, J. Chem. Phys., 26, 634 (1957).
T. Ukaji, Bull. Chem. Soc. Jpn., 32, 1275 (1959).
J. A. Pople and M. Gordon, J. Am. Chem. Soc., 89, 4253 (1967)
N. M. Geller, B. L. Erussalimsky, T. P. Zubova, V. N. Krasulina, V. A. Kropachev, S. S. Skorokhodov, and Yu. Ye. Eizner, Thesis of the XVI Conference of the Institute of High Molecular Weight Compounds of the Academy of Science, USSR, Leningrad, 1970, p. 10.
B. L. Erussalimsky, “Ionic Polymerization of Polar Monomers” Nauka, Leningrad, 1970.
C. C. Costain and B. P. Stoicheff, J. Chem. Phys., 30, 777 (1959).
N. M. Geller, G. B. Erussalimsky, V. A. Kropachev, and Yu. Ye. Eizner, to be published.
S. I. Vinogradova, V. M. Denisov, and A. I. Koltzov, Zh. Org. Khim., 42, 1031 (1972).
E. B. Milovskaya, L. V. Zamoiskaya, and S. I. Vinogradova, Eur. Polym. J., 6, 1589 (1970).
E. B. Milovskaya, E. L. Kopp, O. S. Mikhailycheva, V. M. Denisov, and A. I. Koltzov, Polymer, 13, 288 (1972).
E. N. Guryanova, Usp. Khim., 37, 1981 (1968).
J. M. Godenow and M. Tamres, J. Chem. Phys., 43, 3393 (1965).
M. J. S. Dewar, Angew. Chem., 82, 960 (1970).
H. Fischer and H. Kollmar, Theor. Chim. Acta (Berl.), 13, 213 (1969).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Eizner, Y., Erussalimsky, B. & Milovskaya, E. Electronic Structure and Energy of the Complexes: Methyllithium—Unsaturated Polar Monomer. Polym J 5, 1–10 (1973). https://doi.org/10.1295/polymj.5.1
Issue Date:
DOI: https://doi.org/10.1295/polymj.5.1
Keywords
This article is cited by
-
Choice of semiempirical parameters for CNDO calculation of the electronic structure of organolithium compounds
Journal of Structural Chemistry (1977)