Abstract
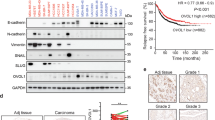
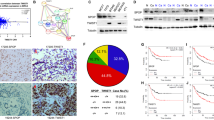
Coactosin-like protein (CLP, or Cotl1), is an F-actin-binding protein, whose role in cancer is largely unknown. Here we show that CLP/Cotl1 is highly expressed in a rat epithelial breast cancer cell line (FE1.3) compared with its mesenchymal counterpart (FE1.2). Knockdown of CLP/Cotl1 in FE1.3 cells increased cell proliferation, whereas its overexpression in FE1.2 cells inhibited proliferation in culture and reduced tumor growth in xenograft assays in mice. Mechanistically, we identified two major pathways through which CLP/Cotl1 exerts its suppressive effects. First, CLP/Cotl1 re-expression in FE1.2 and in human MCF7 breast cancer cells induced expression of the growth-suppressor gene interleukin-24 (IL-24), which independently of p53 upregulates the tumor-suppressor genes p53 apoptosis effector related to PMP-22 (PERP) and p21cip1. Second, overexpression of CLP/Cotl1 potentiated the growth-suppressive effect of transforming growth factor-β1 (TGFβ1), leading to downregulation of TGFβ-responsive genes vascular growth factor A/B (VEGFA/VEGFB), hypoxia inducing factor 1α (HIF-1α) and trombospondin 1 (TSP1), which mediate various hallmarks of cancer progression including angiogenesis, invasion and metastasis. CLP/Cotl1 inhibited TGFβ signaling via a non-canonical signaling involving IL-24-instigated inhibition of mitogen-activated protein kinase (MAPK)/extracellular signal-regulated kinase (ERK) phosphorylation and subsequent post-transcriptional downregulation of SMAD2 and SMAD4. We also showed that CLP/COTL1 expression sensitizes breast cancer cells to chemotherapeutic drugs, and this was further enhanced by addition of exogenous TGFβ1. CLP/Cotl1 expression is lost in many human malignancies including prostate, uterine and breast cancers. Thus, our results uncover a novel tumor-suppressor role for CLP/Cotl1 and identify the downstream effectors interleukin 24 (IL-24)/PERP and IL-24/MAPK/ERK/TGFβ as potential targets for precision therapy.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Provost P, Doucet J, Stock A, Gerisch G, Samuelsson B, Radmark O . Coactosin-like protein, a human F-actin-binding protein: critical role of lysine-75. Biochem J 2001; 359: 255–263.
Provost P, Doucet J, Hammarberg T, Gerisch G, Samuelsson B, Radmark O . 5-Lipoxygenase interacts with coactosin-like protein. J Biol Chem 2001; 276: 16520–16527.
Provost P, Samuelsson B, Radmark O . Interaction of 5-lipoxygenase with cellular proteins. Proc Natl Acad Sci USA 1999; 96: 1881–1885.
Rakonjac M, Fischer L, Provost P, Werz O, Steinhilber D, Samuelsson B et al. Coactosin-like protein supports 5-lipoxygenase enzyme activity and up-regulates leukotriene A4 production. Proc Natl Acad Sci USA 2006; 103: 13150–13155.
Wurthner JU, Frank DB, Felici A, Green HM, Cao Z, Schneider MD et al. Transforming growth factor-beta receptor-associated protein 1 is a Smad4 chaperone. J Biol Chem 2001; 276: 19495–19502.
Gevaert K, Goethals M, Martens L, Van Damme J, Staes A, Thomas GR et al. Exploring proteomes and analyzing protein processing by mass spectrometric identification of sorted N-terminal peptides. Nat Biotechnol 2003; 21: 566–569.
Li X, Liu X, Zhao Q, Liu Y, Duan X, Rao Z . Crystallization and preliminary crystallographic studies of human coactosin-like protein (CLP). Acta Crystallogr D Biol Crystallogr 2004; 60: 2387–2388.
Li X, Liu X, Lou Z, Duan X, Wu H, Liu Y et al. Crystal structure of human coactosin-like protein at 1.9 A resolution. Protein Sci 2004; 13: 2845–2851.
Liu L, Wang Y, Zhang P, Cheng Z, Wan M, Zhou Z et al. Expression, purification and preliminary crystallographic studies of human coactosin-like protein. Acta Crystallogr D Biol Crystallogr 2004; 60: 1651–1653.
Dai H, Wu J, Xu Y, Yajun T, Husheng D, Shi Y . 1H, 13C and 15N resonance assignments and the secondary structures of human coactosin like protein (hCLP) D123N. J Biomol NMR 2004; 29: 455–456.
Dai H, Huang W, Xu J, Yao B, Xiong S, Ding H et al. Binding model of human coactosin-like protein with filament actin revealed by mutagenesis. Biochim Biophys Acta 2006; 1764: 1688–1700.
Kim J, Shapiro MJ, Bamidele AO, Gurel P, Thapa P, Higgs HN et al. Coactosin-like 1 antagonizes cofilin to promote lamellipodial protrusion at the immune synapse. PLoS One 2014; 9: e85090.
Nakatsura T, Senju S, Ito M, Nishimura Y, Itoh K . Cellular and humoral immune responses to a human pancreatic cancer antigen, coactosin-like protein, originally defined by the SEREX method. Eur J Immunol 2002; 32: 826–836.
Guo S, Yang P, Jiang X, Li X, Wang Y, Zhang X et al. Genetic and epigenetic silencing of mircoRNA-506-3p enhances COTL1 oncogene expression to foster non-small lung cancer progression. Oncotarget 2017; 8: 644–657.
Xuan W, Li YJ, Liu G, Ben-David Y, Archer MC . Interleukin-24 induces expression of beta4 integrin but suppresses anchorage-independent growth of rat mammary tumor cells by a mechanism that is independent of beta4. Mol Cancer Res 2009; 7: 433–442.
Li YJ, Song R, Korkola JE, Archer MC, Ben-David Y . Cyclin D1 is necessary but not sufficient for anchorage-independent growth of rat mammary tumor cells and is associated with resistance of the Copenhagen rat to mammary carcinogenesis. Oncogene 2003; 22: 3452–3462.
Menezes ME, Shen XN, Das SK, Emdad L, Guo C, Yuan F et al. MDA-7/IL-24 functions as a tumor suppressor gene in vivo in transgenic mouse models of breast cancer. Oncotarget 2015; 6: 36928–36942.
Ihrie RA, Reczek E, Horner JS, Khachatrian L, Sage J, Jacks T et al. Perp is a mediator of p53-dependent apoptosis in diverse cell types. Curr Biol 2003; 13: 1985–1990.
Li YJ, Liu G, Xia L, Xiao X, Liu JC, Menezes ME et al. Suppression of Her2/Neu mammary tumor development in mda-7/IL-24 transgenic mice. Oncotarget 2015; 6: 36943–36954.
Sainz-Perez A, Gary-Gouy H, Gaudin F, Maarof G, Marfaing-Koka A, de Revel T et al. IL-24 induces apoptosis of chronic lymphocytic leukemia B cells engaged into the cell cycle through dephosphorylation of STAT3 and stabilization of p53 expression. J Immunol 2008; 181: 6051–6060.
Su ZZ, Lebedeva IV, Sarkar D, Gopalkrishnan RV, Sauane M, Sigmon C et al. Melanoma differentiation associated gene-7, mda-7/IL-24, selectively induces growth suppression, apoptosis and radiosensitization in malignant gliomas in a p53-independent manner. Oncogene 2003; 22: 1164–1180.
Cerami E, Gao J, Dogrusoz U, Gross BE, Sumer SO, Aksoy BA et al. The cBio cancer genomics portal: an open platform for exploring multidimensional cancer genomics data. Cancer Discov 2012; 2: 401–404.
Gao J, Aksoy BA, Dogrusoz U, Dresdner G, Gross B, Sumer SO et al. Integrative analysis of complex cancer genomics and clinical profiles using the cBioPortal. Sci Signal 2013; 6: l1.
Uhlen M, Fagerberg L, Hallstrom BM, Lindskog C, Oksvold P, Mardinoglu A et al. Proteomics. Tissue-based map of the human proteome. Science 2015; 347: 1260419.
Uhlen M, Oksvold P, Fagerberg L, Lundberg E, Jonasson K, Forsberg M et al. Towards a knowledge-based Human Protein Atlas. Nat Biotechnol 2010; 28: 1248–1250.
Zawel L, Dai JL, Buckhaults P, Zhou S, Kinzler KW, Vogelstein B et al. Human Smad3 and Smad4 are sequence-specific transcription activators. Mol Cell 1998; 1: 611–617.
de Caestecker MP, Piek E, Roberts AB . Role of transforming growth factor-beta signaling in cancer. J Natl Cancer Inst 2000; 92: 1388–1402.
Nakagawa T, Li JH, Garcia G, Mu W, Piek E, Bottinger EP et al. TGF-beta induces proangiogenic and antiangiogenic factors via parallel but distinct Smad pathways. Kidney Int 2004; 66: 605–613.
Gubin MM, Calaluce R, Davis JW, Magee JD, Strouse CS, Shaw DP et al. Overexpression of the RNA binding protein HuR impairs tumor growth in triple negative breast cancer associated with deficient angiogenesis. Cell Cycle 2014; 9: 3357–3366.
Tian M, Neil JR, Schiemann WP . Transforming growth factor-beta and the hallmarks of cancer. Cell Signal 2011; 23: 951–962.
Bhola NE, Balko JM, Dugger TC, Kuba MG, Sanchez V, Sanders M et al. TGF-beta inhibition enhances chemotherapy action against triple-negative breast cancer. J Clin Invest 2013; 123: 1348–1358.
Luo K . Signaling cross talk between TGF-beta/Smad and other signaling pathways. Cold Spring Harb Perspect Biol 2017; 9: pii: a022137.
Sun W, Guo C, Meng X, Yu Y, Jin Y, Tong D et al. Differential expression of PAI-RBP1, C1orf142, and COTL1 in non-small cell lung cancer cell lines with different tumor metastatic potential. J Investig Med 2012; 60: 689–694.
Wylie-Sears J, Levine RA, Bischoff J . Losartan inhibits endothelial-to-mesenchymal transformation in mitral valve endothelial cells by blocking transforming growth factor-beta-induced phosphorylation of ERK. Biochem Biophys Res Commun 2014; 446: 870–875.
Li CY, Suardet L, Little JB . Potential role of WAF1/Cip1/p21 as a mediator of TGF-beta cytoinhibitory effect. J Biol Chem 1995; 270: 4971–4974.
Menezes ME, Bhatia S, Bhoopathi P, Das SK, Emdad L, Dasgupta S et al. MDA-7/IL-24: multifunctional cancer killing cytokine. Adv Exp Med Biol 2014; 818: 127–153.
Wang CJ, Zhang H, Chen K, Zheng JW, Xiao CW, Ji WW et al. Ad.mda-7 (IL-24) selectively induces apoptosis in hepatocellular carcinoma cell lines, suppresses metastasis, and enhances the effect of doxorubicin on xenograft tumors. Oncol Res 2010; 18: 561–574.
Xia L, Plachynta M, Liu T, Xiao X, Song J, Li X et al. Pro-inflammatory effect of a traditional Chinese medicine formula with potent anti-cancer activity in vitro impedes tumor inhibitory potential in vivo. Mol Clin Oncol 2016; 5: 717–723.
Acknowledgements
This work was supported by research grants from the National Natural Science Foundation of China (81472609), the Science and Technology Department of Guizhou Province Innovation and Project Grant (2013-6012) and Thousand Talent Program of China (WQ20135200171) and to YBD.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on the Oncogene website
Supplementary information
Rights and permissions
About this article
Cite this article
Xia, L., Xiao, X., Liu, W. et al. Coactosin-like protein CLP/Cotl1 suppresses breast cancer growth through activation of IL-24/PERP and inhibition of non-canonical TGFβ signaling. Oncogene 37, 323–331 (2018). https://doi.org/10.1038/onc.2017.342
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2017.342
This article is cited by
-
The intrinsic defects of T cells impact the efficacy of CAR-T therapy in patients with diffuse large B-cell lymphoma
Blood Cancer Journal (2023)
-
A critical ETV4/Twist1/Vimentin axis in Ha-RAS-induced aggressive breast cancer
Cancer Gene Therapy (2022)
-
CCL19 suppresses angiogenesis through promoting miR-206 and inhibiting Met/ERK/Elk-1/HIF-1α/VEGF-A pathway in colorectal cancer
Cell Death & Disease (2018)
-
Further studies about Coactosin-like protein-1 affecting the migration of mouse neocortical neurons
Journal of Molecular Histology (2018)