Abstract
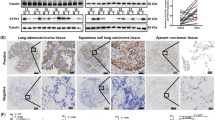
SIRT4 is well-known for its deacetylase activity in energy metabolism, but little is known about its roles in carcinogenesis. We demonstrated that SIRT4 was decreased in 70 out of 133 non-small cell lung cancer (NSCLC) cases by immunohistochemical staining and localized in the mitochondria using confocal microscopy. Low levels of SIRT4 expression was correlated with tumor node metastasis (TNM) stage, histological type of tumor (adenocarcinoma), lymph nodal status, Ki-67 (proliferation index) and poor overall survival. We also studied the biological role of SIRT4 in lung cancer cell lines transfected with SIRT4 plasmid or SIRT4-siRNA. SIRT4 inhibited lung cancer cell proliferation, blocked the cell cycle and repressed cell invasion and migration. Mitochondrial dynamics has been implicated in malignant properties of cells, particularly metastasis that is the major cause of death in patients diagnosed with cancer including lung cancer. This is the first study to identify an association between SIRT4 expression and decreased mitochondrial fission, which was driven by Drp1. SIRT4 inhibited Drp1 phosphorylation and weakened Drp1 recruitment to the mitochondrial membrane via an interaction with Fis-1. SIRT4 expression was lower in nodal metastatic tumor samples than their corresponding primary tumors, and cases with low expression of SIRT4 tended to have high p-Drp1 labeling. Also, MEK/ERK activity appeared to be hampered by SIRT4 expression, which may have implications for cells’ invasive capacities. In conclusion, our findings suggest that SIRT4 functions as an important antitumor protein in NSCLC, and should be investigated further with respect to future anticancer strategies.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
DeSantis CE, Lin CC, Mariotto AB, Siegel RL, Stein KD, Kramer JL et al. Cancer treatment and survivorship statistics, 2014. CA Cancer J Clin 2014; 64: 252–271.
Siegel RL, Miller KD, Jemal A . Cancer statistics, 2015. CA Cancer J Clin 2015; 65: 5–29.
Torre LA, Bray F, Siegel RL, Ferlay J, Lortet-Tieulent J, Jemal A . Global cancer statistics, 2012. CA Cancer J Clin 2015; 65: 87–108.
Chambers AF, Groom AC, MacDonald IC . Dissemination and growth of cancer cells in metastatic sites. Nat Rev Cancer 2002; 2: 563–572.
Haigis MC, Mostoslavsky R, Haigis KM, Fahie K, Christodoulou DC, Murphy AJ et al. SIRT4 inhibits glutamate dehydrogenase and opposes the effects of calorie restriction in pancreatic beta cells. Cell 2006; 126: 941–954.
Saunders LR, Verdin E . Sirtuins: critical regulators at the crossroads between cancer and aging. Oncogene 2007; 26: 5489–5504.
Ahuja N, Schwer B, Carobbio S, Waltregny D, North BJ, Castronovo V et al. Regulation of insulin secretion by SIRT4, a mitochondrial ADP-ribosyltransferase. J Biol Chem 2007; 282: 33583–33592.
Argmann C, Auwerx J . Insulin secretion: SIRT4 gets in on the act. Cell 2006; 126: 837–839.
Jeong SM, Xiao C, Finley LW, Lahusen T, Souza AL, Pierce K et al. SIRT4 has tumor-suppressive activity and regulates the cellular metabolic response to DNA damage by inhibiting mitochondrial glutamine metabolism. Cancer Cell 2013; 23: 450–463.
Nasrin N, Wu X, Fortier E, Feng Y, Bare OC, Chen S et al. SIRT4 regulates fatty acid oxidation and mitochondrial gene expression in liver and muscle cells. J Biol Chem 2010; 285: 31995–32002.
Csibi A, Fendt SM, Li C, Poulogiannis G, Choo AY, Chapski DJ et al. The mTORC1 pathway stimulates glutamine metabolism and cell proliferation by repressing SIRT4. Cell 2013; 153: 840–854.
Jeong SM, Lee A, Lee J, Haigis MC . SIRT4 protein suppresses tumor formation in genetic models of Myc-induced B cell lymphoma. J Biol Chem 2014; 289: 4135–4144.
Huang G, Cui F, Yu F, Lu H, Zhang M, Tang H et al. Sirtuin-4 (SIRT4) is downregulated and associated with some clinicopathological features in gastric adenocarcinoma. Biomed Pharmacother 2015; 72: 135–139.
Miyo M, Yamamoto H, Konno M, Colvin H, Nishida N, Koseki J et al. Tumour-suppressive function of SIRT4 in human colorectal cancer. Br J Cancer 2015; 113: 492–499.
Antico Arciuch VG, Elguero ME, Poderoso JJ, Carreras MC . Mitochondrial regulation of cell cycle and proliferation. Antioxid Redox Signal 2012; 16: 1150–1180.
Chan DC . Mitochondrial fusion and fission in mammals. Annu Rev Cell Dev Biol 2006; 22: 79–99.
Zhao J, Zhang J, Yu M, Xie Y, Huang Y, Wolff DW et al. Mitochondrial dynamics regulates migration and invasion of breast cancer cells. Oncogene 2013; 32: 4814–4824.
Boland ML, Chourasia AH, Macleod KF . Mitochondrial dysfunction in cancer. Front Oncol 2013; 3: 292.
van der Bliek AM, Shen Q, Kawajiri S . Mechanisms of mitochondrial fission and fusion. Cold Spring Harbor Perspect BIol 2013; 5: a011072.
Westermann B . Mitochondrial fusion and fission in cell life and death. Nat Rev Mol Cell Biol 2010; 11: 872–884.
Archer SL . Mitochondrial dynamics—mitochondrial fission and fusion in human diseases. N Engl J Med 2013; 369: 2236–2251.
Grandemange S, Herzig S, Martinou JC . Mitochondrial dynamics and cancer. Semin Cancer Biol 2009; 19: 50–56.
Roberts ER, Thomas KJ . The role of mitochondria in the development and progression of lung cancer. Comput Struct Biotechnol J 2013; 6: e201303019.
Lennon FE, Salgia R . Mitochondrial dynamics: biology and therapy in lung cancer. Expert Opin Invest Drugs 2014; 23: 675–692.
Rehman J, Zhang HJ, Toth PT, Zhang Y, Marsboom G, Hong Z et al. Inhibition of mitochondrial fission prevents cell cycle progression in lung cancer. FASEB J 2012; 26: 2175–2186.
Thomas KJ, Jacobson MR . Defects in mitochondrial fission protein dynamin-related protein 1 are linked to apoptotic resistance and autophagy in a lung cancer model. PloS One 2012; 7: e45319.
Wang Y, Dong QZ, Fu L, Stoecker M, Wang E, Wang EH . Overexpression of CRKL correlates with poor prognosis and cell proliferation in non-small cell lung cancer. Mol Carcinogenes 2013; 52: 890–899.
Laurent G, German NJ, Saha AK, de Boer VC, Davies M, Koves TR et al. SIRT4 coordinates the balance between lipid synthesis and catabolism by repressing malonyl CoA decarboxylase. Mol Cell 2013; 50: 686–698.
Carafa V, Nebbioso A, Altucci L . Sirtuins and disease: the road ahead. Front Pharmacol 2012; 3: 4.
Pal AD, Basak NP, Banerjee AS, Banerjee S . Epstein-Barr virus latent membrane protein-2 A alters mitochondrial dynamics promoting cellular migration mediated by Notch signaling pathway. Carcinogenesis 2014; 35: 1592–1601.
Wan YY, Zhang JF, Yang ZJ, Jiang LP, Wei YF, Lai QN et al. Involvement of Drp1 in hypoxia-induced migration of human glioblastoma U251 cells. Oncol Rep 2014; 32: 619–626.
Taguchi N, Ishihara N, Jofuku A, Oka T, Mihara K . Mitotic phosphorylation of dynamin-related GTPase Drp1 participates in mitochondrial fission. J Biol Chem 2007; 282: 11521–11529.
Cassidy-Stone A, Chipuk JE, Ingerman E, Song C, Yoo C, Kuwana T et al. Chemical inhibition of the mitochondrial division dynamin reveals its role in Bax/Bak-dependent mitochondrial outer membrane permeabilization. Dev Cell 2008; 14: 193–204.
Lin F, Chengyao X, Qingchang L, Qianze D, Enhua W, Yan W . CRKL promotes lung cancer cell invasion through ERK-MMP9 pathway. Mol Carcinog 2015; 54: E35–E44.
Sebolt-Leopold JS, Herrera R . Targeting the mitogen-activated protein kinase cascade to treat cancer. Nat Rev Cancer 2004; 4: 937–947.
Webb CP, Van Aelst L, Wigler MH, Vande Woude GF . Signaling pathways in Ras-mediated tumorigenicity and metastasis. Proc Natl Acad Sci USA 1998; 95: 8773–8778.
Kashatus JA, Nascimento A, Myers LJ, Sher A, Byrne FL, Hoehn KL et al. Erk2 phosphorylation of Drp1 promotes mitochondrial fission and MAPK-driven tumor growth. Mol Cell 2015; 57: 537–551.
Serasinghe MN, Wieder SY, Renault TT, Elkholi R, Asciolla JJ, Yao JL et al. Mitochondrial division is requisite to RAS-induced transformation and targeted by oncogenic MAPK pathway inhibitors. Mol Cell 2015; 57: 521–536.
Acknowledgements
The study was supported by the National Natural Science Foundation of China (No. 81672964 to Qingchang Li and No. 81302022 to Qianze Dong).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on the Oncogene website
Rights and permissions
About this article
Cite this article
Fu, L., Dong, Q., He, J. et al. SIRT4 inhibits malignancy progression of NSCLCs, through mitochondrial dynamics mediated by the ERK-Drp1 pathway. Oncogene 36, 2724–2736 (2017). https://doi.org/10.1038/onc.2016.425
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2016.425
This article is cited by
-
Mitochondrial homeostasis: shaping health and disease
Current Medicine (2024)
-
Sirtuin 4 activates autophagy and inhibits tumorigenesis by upregulating the p53 signaling pathway
Cell Death & Differentiation (2023)
-
Mitochondrial dynamics in health and disease: mechanisms and potential targets
Signal Transduction and Targeted Therapy (2023)
-
Rab32 promotes glioblastoma migration and invasion via regulation of ERK/Drp1-mediated mitochondrial fission
Cell Death & Disease (2023)
-
SIRT4 in ageing
Biogerontology (2023)