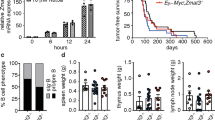
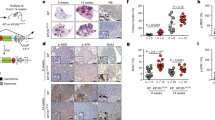
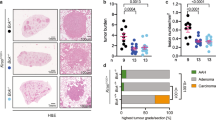
Abstract
Activating mutations in KRAS and EGFR, the two most frequent oncogenes in human lung adenocarcinoma, are mutually exclusive, a phenotype attributed to functional redundancy implying lack of positive selection. Employing a mouse model expressing EGFRL858R in advanced KrasG12V-driven tumors we show that their mutual exclusivity can be explained by detrimental effects of their co-expression in lung adenocarcinoma. In vivo, expression of EGFRL858R in KrasG12V-driven tumors triggers replicative stress and apoptosis, while the surviving cells enter a transient cytostatic state incompatible with tumor development that is fully reversible upon discontinued EGFRL858R expression. Eventually, sustained expression of both mutants induces attenuation of oncogenic signaling to levels compatible with proliferation and tumor growth resulting in high sensitivity to Mek inhibition. Our results indicate that the mutual exclusivity of KRAS and EGFR mutations occurs as a combination of cellular toxicity and signal adjustment resulting in lack of selective advantage for cells expressing both oncogenes.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Kosaka T, Yatabe Y, Endoh H, Kuwano H, Takahashi T, Mitsudomi T . Mutations of the epidermal growth factor receptor gene in lung cancer: biological and clinical implications. Cancer Res 2004; 64: 8919–8923.
Pao W, Wang TY, Riely GJ, Miller VA, Pan Q, Ladanyi M et al. KRAS mutations and primary resistance of lung adenocarcinomas to gefitinib or erlotinib. PLoS Med 2005; 2: 0057–0061.
Shigematsu H, Lin L, Takahashi T, Nomura M, Suzuki M, Wistuba II et al. Clinical and biological features associated with epidermal growth factor receptor gene mutations in lung cancers. J Natl Cancer Inst 2005; 97: 339–346.
Tam IYS, Chung LP, Suen WS, Wang E, Wong MCM, Ho KK et al. Distinct epidermal growth factor receptor and KRAS mutation patterns in non-small cell lung cancer patients with different tobacco exposure and clinicopathologic features. Clin Cancer Res 2006; 12: 1647–1653.
Gazdar AF, Shigematsu H, Herz J, Minna JD . Mutations and addiction to EGFR: the Achilles ‘heal’ of lung cancers? Trends Mol Med 2004; 10: 481–486.
Unni AM, Lockwood WW, Zejnullahu K, Lee-Lin S-Q, Varmus HE . Evidence that synthetic lethality underlies the mutual exclusivity of oncogenic KRAS and EGFR mutations in lung adenocarcinoma. Elife 2015; 4: e06907.
Blasco RB, Francoz S, Santamaría D, Cañamero M, Dubus P, Charron J et al. C-Raf, but not B-Raf, is essential for development of K-Ras oncogene-driven non-small cell lung carcinoma. Cancer Cell 2011; 19: 652–663.
Gu Z, Steinmetz LM, Gu X, Scharfe C, Davis RW, Li WH . Role of duplicate genes in genetic robustness against null mutations. Nature 2003; 421: 63–66.
Xu AM, Huang PH . Receptor tyrosine kinase coactivation networks in cancer. Cancer Res 2010; 70: 3857–3860.
Sun C, Bernards R . Feedback and redundancy in receptor tyrosine kinase signaling: relevance to cancer therapies. Trends Biochem Sci 2014; 10: 465–474.
Avraham R, Yarden Y . Feedback regulation of EGFR signalling: decision making by early and delayed loops. Nat Rev Mol Cell Biol 2011; 12: 104–117.
Guerra C, Mijimolle N, Dhawahir A, Dubus P, Barradas M, Serrano M et al. Tumor induction by an endogenous K-ras oncogene is highly dependent on cellular context. Cancer Cell 2003; 4: 111–120.
Politi K, Zakowski MF, Fan PD, Schonfeld EA, Pao W, Varmus HE . Lung adenocarcinomas induced in mice by mutant EGF receptors found in human lung cancers respond to a tyrosine kinase inhibitor or to down-regulation of the receptors. Genes Dev 2006; 20: 1496–1510.
Belteki G, Haigh J, Kabacs N, Haigh K, Sison K, Costantini F et al. Conditional and inducible transgene expression in mice through the combinatorial use of Cre-mediated recombination and tetracycline induction. Nucleic Acids Res 2005; 33: e51.
Ambrogio C, Carmona FJ, Vidal A, Falcone M, Nieto P, Romero OA et al. Modeling lung cancer evolution and pre-clinical response by orthotopic mouse allografts. Cancer Res 2014; 74: 5978–5988.
Anastasi S, Lamberti D, Alemá S, Segatto O . Regulation of the ErbB network by the MIG6 feedback loop in physiology, tumor suppression and responses to oncogene-targeted therapeutics. Semin Cell Dev Biol 50: 115–124.
Segatto O, Anastasi S, Alemà S . Regulation of epidermal growth factor receptor signalling by inducible feedback inhibitors. J Cell Sci 2011; 124: 1785–1793.
Maity TK, Venugopalan A, Linnoila I, Cultraro CM, Giannakou A, Nemati R et al. Loss of MIG6 accelerates initiation and progression of mutant epidermal growth factor receptor-driven lung adenocarcinoma. Cancer Discov 2015; 5: 534–549.
Jänne PA, Shaw AT, Pereira JR, Jeannin G, Vansteenkiste J, Barrios C et al. Selumetinib plus docetaxel for KRAS-mutant advanced non-small-cell lung cancer: a randomised, multicentre, placebo-controlled, phase 2 study. Lancet Oncol 2013; 14: 38–47.
Pikor LA, Ramnarine VR, Lam S, Lam WL . Genetic alterations defining NSCLC subtypes and their therapeutic implications. Lung Cancer 2013; 82: 179–189.
Sunaga N, Shames DS, Girard L, Peyton M, Larsen JE, Imai H et al. Knockdown of oncogenic KRAS in non-small cell lung cancers suppresses tumor growth and sensitizes tumor cells to targeted therapy. Mol Cancer Ther 2011; 10: 336–346.
Guha U, Chaerkady R, Marimuthu A, Patterson AS, Kashyap MK, Harsha HC et al. Comparisons of tyrosine phosphorylated proteins in cells expressing lung cancer-specific alleles of EGFR and KRAS. Proc Natl Acad Sci USA 2008; 105: 14112–14117.
Li S, Li L, Zhu Y, Huang C, Qin Y, Liu H et al. Coexistence of EGFR with KRAS, or BRAF, or PIK3CA somatic mutations in lung cancer: a comprehensive mutation profiling from 5125 Chinese cohorts. Br J Cancer 2014; 110: 2812–2820.
Sharifnia T, Rusu V, Piccioni F, Bagul M, Imielinski M, Cherniack AD et al. Genetic modifiers of EGFR dependence in non-small cell lung cancer. Proc Natl Acad Sci USA 2014; 111: 18661–18666.
Choughule A, Sharma R, Trivedi V, Thavamani A, Noronha V, Joshi A et al. Coexistence of KRAS mutation with mutant but not wild-type EGFR predicts response to tyrosine-kinase inhibitors in human lung cancer. Br J Cancer 2014; 111: 2203–2204.
Cisowski J, Sayin VI, Liu M, Karlsson C, Bergo MO . Oncogene-induced senescence underlies the mutual exclusive nature of oncogenic KRAS and BRAF. Oncogene 2015; 35: 1–6.
Puyol M, Martín A, Dubus P, Mulero F, Pizcueta P, Khan G et al. A synthetic lethal interaction between K-Ras oncogenes and Cdk4 unveils a therapeutic strategy for non-small cell lung carcinoma. Cancer Cell 2010; 18: 63–73.
Ambrogio C, Cámara J, Nieto P, Santamaría D, Mulero F . Analysis of murine lung tumors by micro PET-CT imaging. BioProtocol 2015; 5: e1692.
Acknowledgements
We thank J. Haigh for providing Rosa26-LSLrtTA mice. This work was supported by the European Research Council (ERC-AG/ 250297-RAS AEAD), EU-Framework Programme (HEALTH-F2-2010-259770/LUNGTARGET and HEALTH-2010-260791/EUROCANPLATFORM), Spanish Ministry of Economy and Competitiveness (SAF2011-30173) and Autonomous Community of Madrid (S2011/BDM-2470/ONCOCYCLE) to MB. CA is the recipient of a postdoctoral fellowship from the Spanish Association Against Cancer (AECC). We thank A. Ventura, O. Segatto and T. Cash for critical reading of the manuscript and S. Antón for constructive discussion.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on the Oncogene website
Supplementary information
Rights and permissions
About this article
Cite this article
Ambrogio, C., Barbacid, M. & Santamaría, D. In vivo oncogenic conflict triggered by co-existing KRAS and EGFR activating mutations in lung adenocarcinoma. Oncogene 36, 2309–2318 (2017). https://doi.org/10.1038/onc.2016.385
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2016.385
This article is cited by
-
Cooperative function of oncogenic MAPK signaling and the loss of Pten for melanoma migration through the formation of lamellipodia
Scientific Reports (2024)
-
ERK pathway agonism for cancer therapy: evidence, insights, and a target discovery framework
npj Precision Oncology (2024)
-
Oncogenic KRAS promotes growth of lung cancer cells expressing SLC3A2-NRG1 fusion via ADAM17-mediated shedding of NRG1
Oncogene (2022)
-
RAS oncogenic activity predicts response to chemotherapy and outcome in lung adenocarcinoma
Nature Communications (2022)
-
The origins and genetic interactions of KRAS mutations are allele- and tissue-specific
Nature Communications (2021)