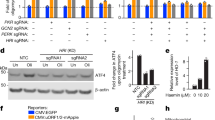
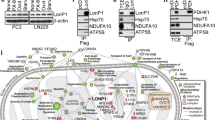
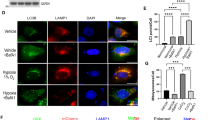
Abstract
Retrograde signaling is a mechanism by which mitochondrial dysfunction is communicated to the nucleus for inducing a metabolic shift essential for cell survival. Previously, we showed that partial mitochondrial DNA (mtDNA) depletion in different cell types induced mitochondrial retrograde signaling pathway (MtRS) involving Ca+2-sensitive Calcineurin (Cn) activation as an immediate upstream event of stress response. In multiple cell types, this stress signaling was shown to induce tumorigenic phenotypes in immortalized cells. In this study we show that MtRS also induces p53 expression, which was abrogated by Ca2+ chelators and short hairpin RNA-mediated knockdown of CnAβ mRNA. Mitochondrial dysfunction induced by mitochondrial ionophore, carbonyl cyanide m-chlorophenyl hydrazone and other respiratory inhibitors, which perturb the transmembrane potential, were equally efficient in inducing the expression of p53 and downregulation of MDM2. Stress-induced p53 physically interacted with hypoxia-inducible factor-1α (HIF-1α) and attenuated the latter’s binding to promoter DNA motifs. In addition, p53 promoted ubiquitination and degradation of HIF-1α in partial mtDNA-depleted cells. The mtDNA depleted cells, with inhibited HIF-1α, showed upregulation of glycolytic pathway genes, glucose transporter 1–4 (Glut1–4), phosphoglycerate kinase 1 and Glucokinase but not of prolyl hydroxylase isoforms. For the first time we show that p53 is induced as part of MtRS and it renders HIF-1α inactive by physical interaction. In this respect, our results show that MtRS induces tumor growth independent of the HIF-1α pathway.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Amundson SA, Myers TG, Fornace AJ Jr . Roles for p53 in growth arrest and apoptosis: putting on the brakes after genotoxic stress. Oncogene 1998; 17: 3287–3299.
Sun Y, Wicha M, Leopold WR . Regulation of metastasis-related gene expression by p53: a potential clinical implication. Mol Carcinog 1999; 24: 25–28.
Menendez D, Inga A, Snipe J, Krysiak O, Schonfelder G, Resnick MA . A single-nucleotide polymorphism in a half-binding site creates p53 and estrogen receptor control of vascular endothelial growth factor receptor 1. Mol Cell Biol 2007; 27: 2590–2600.
Roger L, Jullien L, Gire V, Roux P . Gain of oncogenic function of p53 mutants regulates E-cadherin expression uncoupled from cell invasion in colon cancer cells. J Cell Sci 2010; 123: 1295–1305.
Liu G, Chen X . Regulation of the p53 transcriptional activity. J Cell Biochem 2006; 97: 448–458.
Matoba S, Kang JG, Patino WD, Wragg A, Boehm M, Gavrilova O et al. p53 regulates mitochondrial respiration. Science 2006; 312: 1650–1653.
Prives C . Signaling to p53: breaking the MDM2-p53 circuit. Cell 1998; 95: 5–8.
Jiang P, Du W, Wang X, Mancuso A, Gao X, Wu M et al. p53 regulates biosynthesis through direct inactivation of glucose-6-phosphate dehydrogenase. Nat Cell Biol 2011; 13: 310–316.
Green DR, Kroemer G . Cytoplasmic functions of the tumour suppressor p53. Nature 2009; 458: 1127–1130.
Moll UM, Marchenko N, Zhang XK . p53 and Nur77/. Oncogene 2006; 25: 4725–4743.
Xu H, Tai J, Ye H, Kang CB, Yoon HS . The N-terminal domain of tumor suppressor p53 is involved in the molecular interaction with the anti-apoptotic protein Bcl-Xl. Biochem Biophys Res Commun 2006; 341: 938–944.
Wallace DC . Mitochondria and cancer: Warburg addressed. Cold Spring Harb Symp Quant Biol 2005; 70: 363–374.
Wallace DC . Mitochondria and cancer. Nat Rev Cancer 2012; 12: 685–698.
Chatterjee A, Mambo E, Sidransky D . Mitochondrial DNA mutations in human cancer. Oncogene 2006; 25: 4663–4674.
Boland ML, Chourasia AH, Macleod KF . Mitochondrial dysfunction in cancer. Front Oncol 2013; 3: 292.
Brar SS, Meyer JN, Bortner CD, Van HB, Martin WJ . Mitochondrial DNA-depleted A549 cells are resistant to bleomycin. Am J Physiol Lung Cell Mol Physiol 2012; 303: L413–L424.
Butow RA, Avadhani NG . Mitochondrial signaling: the retrograde response. Mol Cell 2004; 14: 1–15.
Haynes CM, Yang Y, Blais SP, Neubert TA, Ron D . The matrix peptide exporter HAF-1 signals a mitochondrial UPR by activating the transcription factor ZC376.7 in C. elegans. Mol Cell 2010; 37: 529–540.
Formentini L, Sanchez-Arago M, Sanchez-Cenizo L, Cuezva JM . The mitochondrial ATPase inhibitory factor 1 triggers a ROS-mediated retrograde prosurvival and proliferative response. Mol Cell 2012; 45: 731–742.
Amuthan G, Biswas G, Zhang SY, Klein-Szanto A, Vijayasarathy C, Avadhani NG . Mitochondria-to-nucleus stress signaling induces phenotypic changes, tumor progression and cell invasion. EMBO J 2001; 20: 1910–1920.
Biswas G, Adebanjo OA, Freedman BD, Anandatheerthavarada HK, Vijayasarathy C, Zaidi M et al. Retrograde Ca2+ signaling in C2C12 skeletal myocytes in response to mitochondrial genetic and metabolic stress: a novel mode of inter-organelle crosstalk. EMBO J 1999; 18: 522–533.
Biswas G, Anandatheerthavarada HK, Zaidi M, Avadhani NG . Mitochondria to nucleus stress signaling: a distinctive mechanism of NFkappaB/Rel activation through calcineurin-mediated inactivation of IkappaBbeta. J Cell Biol 2003; 161: 507–519.
Bell EL, Klimova TA, Eisenbart J, Moraes CT, Murphy MP, Budinger GR et al. The Qo site of the mitochondrial complex III is required for the transduction of hypoxic signaling via reactive oxygen species production. J Cell Biol 2007; 177: 1029–1036.
Chae S, Ahn BY, Byun K, Cho YM, Yu MH, Lee B et al. A systems approach for decoding mitochondrial retrograde signaling pathways. Sci Signal 2013; 6: rs4.
Jones RG, Plas DR, Kubek S, Buzzai M, Mu J, Xu Y et al. AMP-activated protein kinase induces a p53-dependent metabolic checkpoint. Mol Cell 2005; 18: 283–293.
Tang W, Chowdhury AR, Guha M, Huang L, Van WT, Rustgi AK et al. Silencing of IkBbeta mRNA causes disruption of mitochondrial retrograde signaling and suppression of tumor growth in vivo. Carcinogenesis 2012; 33: 1762–1768.
Guha M, Avadhani NG . Mitochondrial retrograde signaling at the crossroads of tumor bioenergetics, genetics and epigenetics. Mitochondrion 2013; 13: 577–591.
Biswas G, Anandatheerthavarada HK, Avadhani NG . Mechanism of mitochondrial stress-induced resistance to apoptosis in mitochondrial DNA-depleted C2C12 myocytes. Cell Death Differ 2005; 12: 266–278.
Hamanaka RB, Chandel NS . Mitochondrial reactive oxygen species regulate cellular signaling and dictate biological outcomes. Trends Biochem Sci 2010; 35: 505–513.
Iommarini L, Kurelac I, Capristo M, Calvaruso MA, Giorgio V, Bergamini C et al. Different mtDNA mutations modify tumor progression in dependence of the degree of respiratory complex I impairment. Hum Mol Genet 2014; 23: 1453–1466.
Obacz J, Pastorekova S, Vojtesek B, Hrstka R . Cross-talk between HIF and p53 as mediators of molecular responses to physiological and genotoxic stresses. Mol Cancer 2013; 12: 93.
Sermeus A, Michiels C . Reciprocal influence of the p53 and the hypoxic pathways. Cell Death Dis 2011; 2: e164.
Dhanasekaran A, Kotamraju S, Karunakaran C, Kalivendi SV, Thomas S, Joseph J et al. Mitochondria superoxide dismutase mimetic inhibits peroxide-induced oxidative damage and apoptosis: role of mitochondrial superoxide. Free Radic Biol Med 2005; 39: 567–583.
Guha M, Srinivasan S, Biswas G, Avadhani NG . Activation of a novel calcineurin-mediated insulin-like growth factor-1 receptor pathway, altered metabolism, and tumor cell invasion in cells subjected to mitochondrial respiratory stress. J Biol Chem 2007; 282: 14536–14546.
Berglind H, Pawitan Y, Kato S, Ishioka C, Soussi T . Analysis of p53 mutation status in human cancer cell lines: a paradigm for cell line cross-contamination. Cancer Biol Ther 2008; 7: 699–708.
Bunz F, Dutriaux A, Lengauer C, Waldman T, Zhou S, Brown JP et al. Requirement for p53 and p21 to sustain G2 arrest after DNA damage. Science 1998; 282: 1497–1501.
Wanka C, Brucker DP, Bahr O, Ronellenfitsch M, Weller M, Steinbach JP et al. Synthesis of cytochrome C oxidase 2: a p53-dependent metabolic regulator that promotes respiratory function and protects glioma and colon cancer cells from hypoxia-induced cell death. Oncogene 2012; 31: 3764–3776.
Minet E, Ernest I, Michel G, Roland I, Remacle J, Raes M et al. HIF1A gene transcription is dependent on a core promoter sequence encompassing activating and inhibiting sequences located upstream from the transcription initiation site and cis elements located within the 5'UTR. Biochem Biophys Res Commun 1999; 261: 534–540.
Guha M, Srinivasan S, Ruthel G, Kashina AK, Carstens RP, Mendoza A et al. Mitochondrial retrograde signaling induces epithelial-mesenchymal transition and generates breast cancer stem cells. Oncogene 2013; 33: 5238–5250.
Wang GL, Semenza GL . Molecular basis of hypoxia-induced erythropoietin expression. Curr Opin Hematol 1996; 3: 156–162.
Forsythe JA, Jiang BH, Iyer NV, Agani F, Leung SW, Koos RD et al. Activation of vascular endothelial growth factor gene transcription by hypoxia-inducible factor 1. Mol Cell Biol 1996; 16: 4604–4613.
Barliya T, Mandel M, Livnat T, Weinberger D, Lavie G . Degradation of HIF-1alpha under hypoxia combined with induction of Hsp90 polyubiquitination in cancer cells by hypericin: a unique cancer therapy. PLoS ONE 2011; 6: e22849.
Chen C, Pore N, Behrooz A, Ismail-Beigi F, Maity A . Regulation of glut1 mRNA by hypoxia-inducible factor-1. Interaction between H-ras and hypoxia. J Biol Chem 2001; 276: 9519–9525.
Jaakkola P, Mole DR, Tian YM, Wilson MI, Gielbert J, Gaskell SJ et al. Targeting of HIF-alpha to the von Hippel-Lindau ubiquitylation complex by O2-regulated prolyl hydroxylation. Science 2001; 292: 468–472.
Masson N, Willam C, Maxwell PH, Pugh CW, Ratcliffe PJ . Independent function of two destruction domains in hypoxia-inducible factor-alpha chains activated by prolyl hydroxylation. EMBO J 2001; 20: 5197–5206.
Cockman ME, Masson N, Mole DR, Jaakkola P, Chang GW, Clifford SC et al. Hypoxia inducible factor-alpha binding and ubiquitylation by the von Hippel-Lindau tumor suppressor protein. J Biol Chem 2000; 275: 25733–25741.
Hon WC, Wilson MI, Harlos K, Claridge TD, Schofield CJ, Pugh CW et al. Structural basis for the recognition of hydroxyproline in HIF-1 alpha by pVHL. Nature 2002; 417: 975–978.
Huang LE, Gu J, Schau M, Bunn HF . Regulation of hypoxia-inducible factor 1alpha is mediated by an O2-dependent degradation domain via the ubiquitin-proteasome pathway. Proc Natl Acad Sci USA 1998; 95: 7987–7992.
Berra E, Benizri E, Ginouves A, Volmat V, Roux D, Pouyssegur J . HIF prolyl-hydroxylase 2 is the key oxygen sensor setting low steady-state levels of HIF-1alpha in normoxia. EMBO J 2003; 22: 4082–4090.
Dang DT, Chen F, Gardner LB, Cummins JM, Rago C, Bunz F et al. Hypoxia-inducible factor-1alpha promotes nonhypoxia-mediated proliferation in colon cancer cells and xenografts. Cancer Res 2006; 66: 1684–1936.
Appelhoff RJ, Tian YM, Raval RR, Turley H, Harris AL, Pugh CW et al. Differential function of the prolyl hydroxylases PHD1, PHD2, and PHD3 in the regulation of hypoxia-inducible factor. J Biol Chem 2004; 279: 38458–38465.
Amuthan G, Biswas G, Ananadatheerthavarada HK, Vijayasarathy C, Shephard HM, Avadhani NG . Mitochondrial stress-induced calcium signaling, phenotypic changes and invasive behavior in human lung carcinoma A549 cells. Oncogene 2002; 21: 7839–7849.
Wallace DC . A mitochondrial bioenergetic etiology of disease. J Clin Invest 2013; 123: 1405–1412.
Lowe SW, Ruley HE, Jacks T, Housman DE . p53-dependent apoptosis modulates the cytotoxicity of anticancer agents. Cell 1993; 74: 957–967.
King MP, Attardi G . Isolation of human cell lines lacking mitochondrial DNA. Methods Enzymol 1996; 264: 304–313.
Bensaad K, Vousden KH . p53: new roles in metabolism. Trends Cell Biol 2007; 17: 286–291.
Puzio-Kuter AM . The role of p53 in metabolic regulation. Genes Cancer 2011; 2: 385–391.
Gu J, Milligan J, Huang LE . Molecular mechanism of hypoxia-inducible factor 1alpha -p300 interaction. A leucine-rich interface regulated by a single cysteine. J Biol Chem 2001; 276: 3550–3554.
Blagosklonny MV, An WG, Romanova LY, Trepel J, Fojo T, Neckers L . p53 inhibits hypoxia-inducible factor-stimulated transcription. J Biol Chem 1998; 273: 11995–11998.
Ravi R, Mookerjee B, Bhujwalla ZM, Sutter CH, Artemov D, Zeng Q et al. Regulation of tumor angiogenesis by p53-induced degradation of hypoxia-inducible factor 1alpha. Genes Dev 2000; 14: 34–44.
Kojima K, McQueen T, Chen Y, Jacamo R, Konopleva M, Shinojima N et al. p53 activation of mesenchymal stromal cells partially abrogates microenvironment-mediated resistance to FLT3 inhibition in AML through HIF-1alpha-mediated down-regulation of CXCL12. Blood 2011; 118: 4431–4439.
An WG, Kanekal M, Simon MC, Maltepe E, Blagosklonny MV, Neckers LM . Stabilization of wild-type p53 by hypoxia-inducible factor 1alpha. Nature 1998; 392: 405–408.
Cao Y, Li CY, Moeller BJ, Yu D, Zhao Y, Dreher MR et al. Observation of incipient tumor angiogenesis that is independent of hypoxia and hypoxia inducible factor-1 activation. Cancer Res 2005; 65: 5498–5505.
Camenisch G, Wenger RH, Gassmann M . DNA-binding activity of hypoxia-inducible factors (HIFs). Methods Mol Biol 2002; 196: 117–129.
Acknowledgements
We thank Drs Craig Thompson, Gregg Semenza and Bert Vogelstein for generously providing the promoter constructs and cell lines used in this study. This work was supported by NIH grant CA-22762, AR-067066 and an endowment from the Harriet Ellison Woodward trust (NGA) and NIH MSTP training grant AL. We also acknowledge the help of the Imaging Core facility at the School of Veterinary Medicine and the NIH/NIDDK Center for Molecular Studies in Digestive and Liver Diseases (P30DK050306) and its Cell collection core.
Author contributions
ARC, NGA, SF and AR conceived the experiments. ARC carried out experiments. AL generated mutant cDNA constructs. ARC and NGA wrote the paper.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on the Oncogene website
Rights and permissions
About this article
Cite this article
Chowdhury, A., Long, A., Fuchs, S. et al. Mitochondrial stress-induced p53 attenuates HIF-1α activity by physical association and enhanced ubiquitination. Oncogene 36, 397–409 (2017). https://doi.org/10.1038/onc.2016.211
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2016.211
This article is cited by
-
The role of ubiquitination and deubiquitination in cancer metabolism
Molecular Cancer (2020)