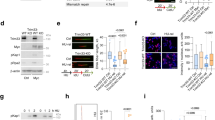
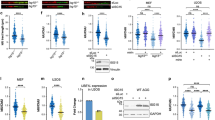
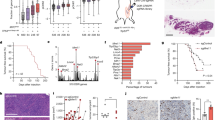
Abstract
Inhibiting the bromodomain and extra-terminal (BET) domain family of epigenetic reader proteins has been shown to have potent anti-tumoral activity, which is commonly attributed to suppression of transcription. In this study, we show that two structurally distinct BET inhibitors (BETi) interfere with replication and cell cycle progression of murine Myc-induced lymphoma cells at sub-lethal concentrations when the transcriptome remains largely unaltered. This inhibition of replication coincides with a DNA-damage response and enhanced sensitivity to inhibitors of the upstream replication stress sensor ATR in vitro and in mouse models of B-cell lymphoma. Mechanistically, ATR and BETi combination therapy cause robust transcriptional changes of genes involved in cell death, senescence-associated secretory pathway, NFkB signaling and ER stress. Our data reveal that BETi can potentiate the cell stress and death caused by ATR inhibitors. This suggests that ATRi can be used in combination therapies of lymphomas without the use of genotoxic drugs.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Belkina AC, Denis GV . BET domain co-regulators in obesity, inflammation and cancer. Nat Rev Cancer 2012; 12: 465–477.
Sinha A, Faller DV, Denis GV . Bromodomain analysis of Brd2-dependent transcriptional activation of cyclin A. Biochem J 2005; 387: 257–269.
Denis GV, Vaziri C, Guo N, Faller DV . RING3 kinase transactivates promoters of cell cycle regulatory genes through E2F. Cell Growth Differ 2000; 11: 417–424.
Jang MK, Mochizuki K, Zhou M, Jeong HS, Brady JN, Ozato K . The bromodomain protein Brd4 is a positive regulatory component of P-TEFb and stimulates RNA polymerase II-dependent transcription. Mol Cell 2005; 19: 523–534.
Yang Z, Yik JH, Chen R, He N, Jang MK, Ozato K et al. Recruitment of P-TEFb for stimulation of transcriptional elongation by the bromodomain protein Brd4. Mol Cell 2005; 19: 535–545.
Marshall NF, Peng J, Xie Z, Price DH . Control of RNA polymerase II elongation potential by a novel carboxyl-terminal domain kinase. J Biol Chem 1996; 271: 27176–27183.
Loven J, Hoke HA, Lin CY, Lau A, Orlando DA, Vakoc CR et al. Selective inhibition of tumor oncogenes by disruption of super-enhancers. Cell 2013; 153: 320–334.
Venkataraman S, Alimova I, Balakrishnan I, Harris P, Birks DK, Griesinger A et al. Inhibition of BRD4 attenuates tumor cell self-renewal and suppresses stem cell signaling in MYC driven medulloblastoma. Oncotarget 2014; 5: 2355–2371.
Pastori C, Daniel M, Penas C, Volmar CH, Johnstone AL, Brothers SP et al. BET bromodomain proteins are required for glioblastoma cell proliferation. Epigenetics 2014; 9: 611–620.
Loosveld M, Castellano R, Gon S, Goubard A, Crouzet T, Pouyet L et al. Therapeutic targeting of c-Myc in T-cell acute lymphoblastic leukemia, T-ALL. Oncotarget 2014; 5: 3168–3172.
Bandopadhayay P, Bergthold G, Nguyen B, Schubert S, Gholamin S, Tang Y et al. BET bromodomain inhibition of MYC-amplified medulloblastoma. Clin Cancer Res 2014; 20: 912–925.
Asangani IA, Dommeti VL, Wang X, Malik R, Cieslik M, Yang R et al. Therapeutic targeting of BET bromodomain proteins in castration-resistant prostate cancer. Nature 2014; 510: 278–282.
Tolani B, Gopalakrishnan R, Punj V, Matta H, Chaudhary PM . Targeting Myc in KSHV-associated primary effusion lymphoma with BET bromodomain inhibitors. Oncogene 2013; 33: 2928–2937.
Puissant A, Frumm SM, Alexe G, Bassil CF, Qi J, Chanthery YH et al. Targeting MYCN in neuroblastoma by BET bromodomain inhibition. Cancer Discov 2013; 3: 308–323.
Gao L, Schwartzman J, Gibbs A, Lisac R, Kleinschmidt R, Wilmot B et al. Androgen receptor promotes ligand-independent prostate cancer progression through c-Myc upregulation. PLoS One 2013; 8: e63563.
Cheng Z, Gong Y, Ma Y, Lu K, Lu X, Pierce LA et al. Inhibition of BET bromodomain targets genetically diverse glioblastoma. Clin Cancer Res 2013; 19: 1748–1759.
Mertz JA, Conery AR, Bryant BM, Sandy P, Balasubramanian S, Mele DA et al. Targeting MYC dependence in cancer by inhibiting BET bromodomains. Proc Natl Acad Sci USA 2011; 108: 16669–16674.
Delmore JE, Issa GC, Lemieux ME, Rahl PB, Shi J, Jacobs HM et al. BET bromodomain inhibition as a therapeutic strategy to target c-Myc. Cell 2011; 146: 904–917.
Keene RG, Mueller A, Landick R, London L . Transcriptional pause, arrest and termination sites for RNA polymerase II in mammalian N- and c-myc genes. Nucleic Acids Res 1999; 27: 3173–3182.
Kerppola TK, Kane CM . Intrinsic sites of transcription termination and pausing in the c-myc gene. Mol Cell Biol 1988; 8: 4389–4394.
Bhadury J, Nilsson LM, Muralidharan SV, Green LC, Li Z, Gesner EM et al. BET and HDAC inhibitors induce similar genes and biological effects and synergize to kill in Myc-induced murine lymphoma. Proc Natl Acad Sci USA 2014; 111: E2721–E2730.
Fiskus W, Sharma S, Qi J, Valenta JA, Schaub LJ, Shah B et al. Highly active combination of BRD4 antagonist and histone deacetylase inhibitor against human acute myelogenous leukemia cells. Mol Cancer Ther 2014; 13: 1142–1154.
Pajic A, Spitkovsky D, Christoph B, Kempkes B, Schuhmacher M, Staege MS et al. Cell cycle activation by c-myc in a burkitt lymphoma model cell line. Int J Cancer 2000; 87: 787–793.
Bhadury J, Lopez MD, Muralidharan SV, Nilsson LM, Nilsson JA . Identification of tumorigenic and therapeutically actionable mutations in transplantable mouse tumor cells by exome sequencing. Oncogenesis 2013; 2: e44.
Ember SW, Zhu JY, Olesen SH, Martin MP, Becker A, Berndt N et al. Acetyl-lysine binding site of bromodomain-containing protein 4 (BRD4) interacts with diverse kinase inhibitors. ACS Chem Biol 2014; 9: 1160–1171.
Ciceri P, Muller S, O'Mahony A, Fedorov O, Filippakopoulos P, Hunt JP et al. Dual kinase-bromodomain inhibitors for rationally designed polypharmacology. Nat Chem Biol 2014; 10: 305–312.
Dey A, Chitsaz F, Abbasi A, Misteli T, Ozato K . The double bromodomain protein Brd4 binds to acetylated chromatin during interphase and mitosis. Proc Natl Acad Sci USA 2003; 100: 8758–8763.
Boi M, Gaudio E, Bonetti P, Kwee I, Bernasconi E, Tarantelli C et al. The BET bromodomain inhibitor OTX015 affects pathogenetic pathways in preclinical B-cell tumor models and synergizes with targeted drugs. Clin Cancer Res 2015; 21: 1628–1638.
Stratikopoulos EE, Dendy M, Szabolcs M, Khaykin AJ, Lefebvre C, Zhou MM et al. Kinase and BET inhibitors together clamp inhibition of PI3K signaling and overcome resistance to therapy. Cancer Cell 2015; 27: 837–851.
Lecona E, Fernandez-Capetillo O . Replication stress and cancer: it takes two to tango. Exp Cell Res 2014; 329: 26–34.
Ferrao PT, Bukczynska EP, Johnstone RW, McArthur GA . Efficacy of CHK inhibitors as single agents in MYC-driven lymphoma cells. Oncogene 2012; 31: 1661–1672.
Murga M, Campaner S, Lopez-Contreras AJ, Toledo LI, Soria R, Montana MF et al. Exploiting oncogene-induced replicative stress for the selective killing of Myc-driven tumors. Nat Struct Mol Biol 2011; 18: 1331–1335.
Höglund A, Strömvall K, Li Y, Forshell LP, Nilsson JA . Chk2 deficiency in Myc overexpressing lymphoma cells elicits a synergistic lethal response in combination with PARP inhibition. Cell Cycle 2011; 10: 3598–3607.
Höglund A, Nilsson L, Muralidharan SV, Hasvold LA, Merta P, Rudelius M et al. Therapeutic implications for the induced levels of Chk1 in Myc-expressing cancer cells. Clin Cancer Res 2011; 17: 7067–7079.
Shortt J, Martin BP, Newbold A, Hannan KM, Devlin JR, Baker AJ et al. Combined inhibition of PI3K-related DNA damage response kinases and mTORC1 induces apoptosis in MYC-driven B-cell lymphomas. Blood 2013; 121: 2964–2974.
Toledo LI, Murga M, Zur R, Soria R, Rodriguez A, Martinez S et al. A cell-based screen identifies ATR inhibitors with synthetic lethal properties for cancer-associated mutations. Nat Struct Mol Biol 2011; 18: 721–727.
Reaper PM, Griffiths MR, Long JM, Charrier JD, Maccormick S, Charlton PA et al. Selective killing of ATM- or p53-deficient cancer cells through inhibition of ATR. Nat Chem Biol 2011; 7: 428–430.
Floyd SR, Pacold ME, Huang Q, Clarke SM, Lam FC, Cannell IG et al. The bromodomain protein Brd4 insulates chromatin from DNA damage signalling. Nature 2013; 498: 246–250.
Josse R, Martin SE, Guha R, Ormanoglu P, Pfister TD, Reaper PM et al. ATR inhibitors VE-821 and VX-970 sensitize cancer cells to topoisomerase i inhibitors by disabling DNA replication initiation and fork elongation responses. Cancer Res 2014; 74: 6968–6979.
Foote KM, Blades K, Cronin A, Fillery S, Guichard SS, Hassall L et al. Discovery of 4-{4-[(3R)-3-Methylmorpholin-4-yl]-6-[1-(methylsulfonyl)cyclopropyl]pyrimidin-2-y l}-1H-indole (AZ20): a potent and selective inhibitor of ATR protein kinase with monotherapy in vivo antitumor activity. J Med Chem 2013; 56: 2125–2138.
Keller U, Huber J, Nilsson JA, Fallahi M, Hall MA, Peschel C et al. Myc suppression of Nfkb2 accelerates lymphomagenesis. BMC Cancer 2010; 10: 348.
Klapproth K, Sander S, Marinkovic D, Baumann B, Wirth T . The IKK2/NF-{kappa}B pathway suppresses MYC-induced lymphomagenesis. Blood 2009; 114: 2448–2458.
Keller U, Nilsson JA, Maclean KH, Old JB, Cleveland JL . Nfkb 1 is dispensable for Myc-induced lymphomagenesis. Oncogene 2005; 24: 6231–6240.
Dorr JR, Yu Y, Milanovic M, Beuster G, Zasada C, Dabritz JH et al. Synthetic lethal metabolic targeting of cellular senescence in cancer therapy. Nature 2013; 501: 421–425.
Jing H, Kase J, Dorr JR, Milanovic M, Lenze D, Grau M et al. Opposing roles of NF-kappaB in anti-cancer treatment outcome unveiled by cross-species investigations. Genes Dev 2011; 25: 2137–2146.
Chien Y, Scuoppo C, Wang X, Fang X, Balgley B, Bolden JE et al. Control of the senescence-associated secretory phenotype by NF-kappaB promotes senescence and enhances chemosensitivity. Genes Dev 2011; 25: 2125–2136.
Moscat J, Diaz-Meco MT . p62 at the crossroads of autophagy, apoptosis, and cancer. Cell 2009; 137: 1001–1004.
Fang J, Barker B, Bolanos L, Liu X, Jerez A, Makishima H et al. Myeloid malignancies with chromosome 5q deletions acquire a dependency on an intrachromosomal NF-kappaB gene network. Cell Rep 2014; 8: 1328–1338.
Sanz L, Diaz-Meco MT, Nakano H, Moscat J . The atypical PKC-interacting protein p62 channels NF-kappaB activation by the IL-1-TRAF6 pathway. EMBO J 2000; 19: 1576–1586.
Stanlie A, Yousif AS, Akiyama H, Honjo T, Begum NA . Chromatin reader Brd4 functions in Ig class switching as a repair complex adaptor of nonhomologous end-joining. Mol Cell 2014; 55: 97–110.
Bolden JE, Tasdemir N, Dow LE, van Es JH, Wilkinson JE, Zhao Z et al. Inducible in vivo silencing of Brd4 identifies potential toxicities of sustained BET protein inhibition. Cell Rep 2014; 8: 1919–1929.
Tang C, Wang X, Soh H, Seyedin S, Cortez MA, Krishnan S et al. Combining radiation and immunotherapy: a new systemic therapy for solid tumors? Cancer Immunol Res 2014; 2: 831–838.
Denis GV, McComb ME, Faller DV, Sinha A, Romesser PB, Costello CE . Identification of transcription complexes that contain the double bromodomain protein Brd2 and chromatin remodeling machines. J Proteome Res 2006; 5: 502–511.
Maruyama T, Farina A, Dey A, Cheong J, Bermudez VP, Tamura T et al. A mammalian bromodomain protein, brd4, interacts with replication factor C and inhibits progression to S phase. Mol Cell Biol 2002; 22: 6509–6520.
Herold S, Wanzel M, Beuger V, Frohme C, Beul D, Hillukkala T et al. Negative regulation of the mammalian UV response by Myc through association with Miz-1. Mol Cell 2002; 10: 509–521.
Zou Z, Huang B, Wu X, Zhang H, Qi J, Bradner J et al. Brd4 maintains constitutively active NF-kappaB in cancer cells by binding to acetylated RelA. Oncogene 2013; 33: 2395–2404.
Huang B, Yang XD, Zhou MM, Ozato K, Chen LF . Brd4 coactivates transcriptional activation of NF-kappaB via specific binding to acetylated RelA. Mol Cell Biol 2009; 29: 1375–1387.
Acknowledgements
We thank Sofia Nordstrand for animal care, and Eric Campeau and Zenith Epigenetics for RVX2135 and helpful discussions. This work was supported by grants from the Swedish Cancer Society, the Swedish Research Council, the Region Västra Götaland (Sahlgrenska University Hospital, Gothenburg), the Knut and Alice Wallenberg Foundation, the Sahlgrenska Academy and BioCARE—a National Strategic Cancer Research Program at University of Gothenburg (to JAN), and from the Assar Gabrielsson Foundation and the W&M Lundgren Foundation (to SVM, JB and LCG).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
KGM was an employee of Zenith Epigenetics Corp at the begining of this project. The remaining authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on the Oncogene website
Supplementary information
Rights and permissions
About this article
Cite this article
Muralidharan, S., Bhadury, J., Nilsson, L. et al. BET bromodomain inhibitors synergize with ATR inhibitors to induce DNA damage, apoptosis, senescence-associated secretory pathway and ER stress in Myc-induced lymphoma cells. Oncogene 35, 4689–4697 (2016). https://doi.org/10.1038/onc.2015.521
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2015.521
This article is cited by
-
Bromodomain and extraterminal (BET) proteins: biological functions, diseases, and targeted therapy
Signal Transduction and Targeted Therapy (2023)
-
BET inhibitor trotabresib in heavily pretreated patients with solid tumors and diffuse large B-cell lymphomas
Nature Communications (2023)
-
Targeting replication stress in cancer therapy
Nature Reviews Drug Discovery (2023)
-
Bromodomain-containing protein BRPF1 is a therapeutic target for liver cancer
Communications Biology (2021)
-
Novel therapeutic strategies targeting telomere maintenance mechanisms in high-risk neuroblastoma
Journal of Experimental & Clinical Cancer Research (2020)