Abstract
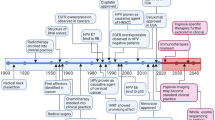
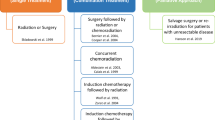
In light of a detailed characterization of genetic aberrations in cancer, nucleic acid targeting represents an attractive therapeutic approach with significant translational potential. Head and neck squamous cell carcinoma (HNSCC) is a leading cause of cancer deaths worldwide with stagnant 5-year survival rates. Advances in conventional treatment have done little to improve survival and combined chemoradiation is associated with significant adverse effects. Recent reports have characterized the genetic alterations in HNSCC and demonstrated that mutations confer resistance to conventional and molecular targeted therapies. The ability to use specific nucleic acid sequences to inhibit cancer-associated genes including non-druggable targets facilitates personalized medicine approaches with less adverse effects. Additionally, advances in drug delivery mechanisms have increased the transfection efficiency aiding in greater therapeutic responses. Given these advances, the stage has been set to translate the information garnered from genomic studies into personalized treatment strategies. Genes involved in the tumor protein 53 and epidermal growth factor receptor pathways have been extensively investigated and many promising preclinical studies have shown tumor inhibition through genetic modulation. We, and others, have demonstrated that targeting oncogene expression with gene therapy approaches is feasible in patients. Other methods such as RNA interference have proven to be effective and are potential candidates for clinical studies. This review summarizes the major advances in sequence-specific gene modulation in the preclinical setting and in clinical trials in head and neck cancer patients.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Döbróssy L . Epidemiology of head and neck cancer: magnitude of the problem. Cancer Metastasis Rev 2005; 24: 9–17.
Accardi R, Adebamowo C, Anderson B, Autier P, Baan R, Barbacid M et alHead and neck cancers. In: Boyle P, Levin B (eds). World Cancer Report 2008. International Agency for Research on Cancer: Lyon, 2008, pp 30–337.
Cohen EEW, Lingen MW, Vokes EE . The expanding role of systemic therapy in head and neck cancer. J Clin Oncol 2004; 22: 1743–1752.
The Cancer Genome Atlas Network. Comprehensive genomic characterization of head and neck squamous cell carcinomas. Nature 2015; 517: 576–582.
Skinner HD, Sandulache VC, Ow TJ, Meyn RE, Yordy JS, Beadle BM et al. TP53 disruptive mutations lead to head and neck cancer treatment failure through inhibition of radiation-induced senescence. Clin Cancer Res 2012; 18: 290–300.
van den Broek GB, Wreesmann VB, van den Brekel MWM, Rasch CRN, Balm AJM, Rao PH . Genetic abnormalities associated with chemoradiation resistance of head and neck squamous cell carcinoma. Clin Cancer Res 2007; 13: 4386–4391.
Lui VW, Hedberg ML, Li H, Vangara BS, Pendleton K, Zeng Y et al. Frequent mutation of the PI3K pathway in head and neck cancer defines predictive biomarkers. Cancer Discov 2013; 3: 761–769.
Vermorken JB, Trigo J, Hitt R, Koralewski P, Diaz-Rubio E, Rolland F et al. Open-label, uncontrolled, multicenter phase II study to evaluate the efficacy and toxicity of cetuximab as a single agent in patients with recurrent and/or metastatic squamous cell carcinoma of the head and neck who failed to respond to platinum-based therapy. J Clin Oncol 2007; 25: 2171–2177.
Saba NF, Haigentz M Jr, Vermorken JB, Strojan P, Bossi P, Rinaldo A et al. Prevention of head and neck squamous cell carcinoma: removing the “chemo” from “chemoprevention”. Oral Oncol 2015; 51: 112–118.
Sok JC, Coppelli FM, Thomas SM, Lango MN, Xi S, Hunt JL et al. Mutant epidermal growth factor receptor (EGFRvIII) contributes to head and neck cancer growth and resistance to EGFR targeting. Clin Cancer Res 2006; 12: 5064–5073.
Cancer Genome Atlas N. Comprehensive genomic characterization of head and neck squamous cell carcinomas. Nature 2015; 517: 576–582.
Lui VWY, Hedberg ML, Li H, Vangara BS, Pendleton K, Zeng Y et al. Frequent mutation of the PI3K pathway in head and neck cancer defines predictive biomarkers. Cancer Discov 2013; 3: 761–769.
Stransky N, Egloff AM, Tward AD, Kostic AD, Cibulskis K, Sivachenko A et al. The mutational landscape of head and neck squamous cell carcinoma. Science 2011; 333: 1157–1160.
Agrawal N, Frederick MJ, Pickering CR, Bettegowda C, Chang K, Li RJ et al. Exome sequencing of head and neck squamous cell carcinoma reveals inactivating mutations in NOTCH1. Science 2011; 333: 1154–1157.
Lechardeur D, Sohn KJ, Haardt M, Joshi PB, Monck M, Graham RW et al. Metabolic instability of plasmid DNA in the cytosol: a potential barrier to gene transfer. Gene Ther 1999; 6: 482–497.
Konopka K, Overlid N, Nagaraj A, Düzgüneş N . Serum decreases the size of Metafectene-and Genejammer-DNA complexes but does not affect significantly their transfection activity in SCCVII murine squamous cell carcinoma cells. Cell Mol Biol Lett 2006; 11: 171–190.
Grandis JR, Drenning SD, Zeng Q, Watkins SC, Melhem MF, Endo S et al. Constitutive activation of Stat3 signaling abrogates apoptosis in squamous cell carcinogenesis in vivo. Proc Natl Acad Sci USA 2000; 97: 4227–4232.
Kedmi R, Ben-Arie N, Peer D . The systemic toxicity of positively charged lipid nanoparticles and the role of Toll-like receptor 4 in immune activation. Biomaterials 2010; 31: 6867–6875.
Tousignant JD, Gates AL, Ingram LA, Johnson CL, Nietupski JB, Cheng SH et al. Comprehensive analysis of the acute toxicities induced by systemic administration of cationic lipid: plasmid DNA complexes in mice. Hum Gene Ther 2000; 11: 2493–2513.
Xu L, Pirollo KF, Tang W-H, Rait A, Chang EH . Transferrin-liposome-mediated systemic p53 gene therapy in combination with radiation results in regression of human head and neck cancer xenografts. Hum Gene Ther 1999; 10: 2941–2952.
Xu L, Huang C-C, Huang W, Tang W-H, Rait A, Yin YZ et al. Systemic tumor-targeted gene delivery by anti-transferrin Rrceptor scFv-immunoliposomes. Mol Cancer Ther 2002; 1: 337–346.
Boussif O, Lezoualc'h F, Zanta MA, Mergny MD, Scherman D, Demeneix B et al. A versatile vector for gene and oligonucleotide transfer into cells in culture and in vivo: polyethylenimine. Proc Natl Acad Sci USA 1995; 92: 7297–7301.
Dolivet G, Merlin J-L, Barberi-Heyob M, Ramacci C, Erbacher P, Parache RM et al. In vivo growth inhibitory effect of iterative wild-type p53 gene transfer in human head and neck carcinoma xenografts using glucosylated polyethylenimine nonviral vector. Cancer Gene Ther 2002; 9: 708–714.
Ndoye A . Sustained gene transfer and enhanced cell death following glucosylated-PEI-mediated p53 gene transfer with photochemical internalisation in p53-mutated head and neck carcinoma cells. Int J Oncol 2004; 25: 1575–1581.
Park I-K, Cook S-E, Kim Y-K, Kim H-W, Cho M-H, Jeong H-J et al. Glucosylated polyethylenimine as a tumor-targeting gene carrier. Arch Pharm Res 2005; 28: 1302–1310.
Ren H, Zhou L, Liu M, Lu W, Gao C . Peptide GE11–polyethylene glycol–polyethylenimine for targeted gene delivery in laryngeal cancer. Med Oncol 2015; 32: 1–8.
Sugano M, Negishi Y, Endo-Takahashi Y, Suzuki R, Maruyama K, Yamamoto M et al. Gene delivery system involving Bubble liposomes and ultrasound for the efficient in vivo delivery of genes into mouse tongue tissue. Int J Pharm 2012; 422: 332–337.
Carson AR, McTiernan CF, Lavery L, Grata M, Leng X, Wang J et al. Ultrasound-targeted microbubble destruction to deliver siRNA cancer therapy. Cancer Res 2012; 72: 6191–6199.
Suzuki R, Takizawa T, Negishi Y, Hagisawa K, Tanaka K, Sawamura K et al. Gene delivery by combination of novel liposomal bubbles with perfluoropropane and ultrasound. J Control Release 2007; 117: 130–136.
Smith NB, Lee S, Maione E, Roy RB, McElligott S, Shung KK . Ultrasound-mediated transdermal transport of insulin in vitro through human skin using novel transducer designs. Ultrasound Med Biol 2003; 29: 311–317.
Sorace AG, Korb M, Warram JM, Umphrey H, Zinn KR, Rosenthal E et al. Ultrasound-stimulated drug delivery for treatment of residual disease following incomplete resection of head and neck cancer. Ultrasound Med Biol 2014; 40: 755–764.
Okunaga S, Takasu A, Meshii N, Imai T, Hamada M, Iwai S et al. Ultrasound as a method to enhance antitumor ability of oncolytic herpes simplex virus for head and neck cancer. Cancer Gene Ther 2015; 22: 163–168.
Blackwell JL, Miller C, Douglas JT, Li H, Reynolds PN, Carroll WR et al. Retargeting to EGFR enhances adenovirus infection efficiency of squamous cell carcinoma. Arch Otolaryngol Head Neck Surg 1999; 125: 856–863.
Lalonde ES, Beyer G, Friedlander PL, Kolls JK . Efficacy of transfection rates on head and neck squamous cell cancer by a novel adenovirus: an in vivo and in vitro study. Head Neck 2002; 24: 1038–1046.
He Y, Zeng Q, Drenning SD, Melhem MF, Tweardy DJ, Huang L et al. EGFR antisense RNA transcribed from the U6 promotor inhibits human squamous cell carcinoma growth in vivo. National Cancer Institute 1998; 90: 1080–1087.
Li M, Ye C, Feng C, Riedel F, Liu X, Zeng Q et al. Enhanced antiangiogenic therapy of squamous cell carcinoma by combined endostatin and epidermal growth factor receptor-antisense therapy. Clin Cancer Res 2002; 8: 3570–3578.
Zeng Q, Kanter PM, Dhir R, Gooding WE, Huang L, Grandis JR . Lack of toxicity of EGFR antisense gene therapy. J Exp Ther Oncol 2002; 2: 174–186.
Thomas SM, Zeng Q, Dyer KF, Suscovich TJ, Kanter PM, Whalen JD et al. Tissue distribution of liposome-mediated epidermal growth factor receptor antisense gene therapy. Cancer Gene Ther 2003; 10: 518–528.
Li D, Jiang W, Bishop JS, Ralston R, O’Malley BW . Combination surgery and nonviral interleukin 2 gene therapy for head and neck cancer. Clin Cancer Res 1999; 5: 1551–1556.
Matsui M, Kishida T, Nakano H, Yoshimoto K, Shin-Ya M, Shimada T et al. Interleukin-27 activates natural killer cells and suppresses NK-resistant head and neck squamous cell carcinoma through inducing antibody-dependent cellular cytotoxicity. Cancer Res 2009; 69: 2523–2530.
Kawakami K, Kawakami M, Joshi BH, Puri RK . Interleukin-13 receptor-targeted cancer therapy in an immunodeficient animal model of human head and neck cancer. Cancer Res 2001; 61: 6194–6200.
Lai SY, Koppikar P, Thomas SM, Childs EE, Egloff AM, Seethala RR et al. Intratumoral epidermal growth factor receptor antisense DNA therapy in head and neck cancer: first human application and potential antitumor mechanisms. J Clin Oncol 2009; 27: 1235–1242.
Michaluart P, Abdallah KA, Lima FD, Smith R, Moyses RA, Coelho V et al. Phase I trial of DNA-hsp65 immunotherapy for advanced squamous cell carcinoma of the head and neck. Cancer Gene Ther 2008; 15: 676–684.
Villaret D, Glisson B, Kenady D, Hanna E, Carey M, Gleich L et al. A multicenter phase II study of tgDCC‐E1A for the intratumoral treatment of patients with recurrent head and neck squamous cell carcinoma. Head Neck 2002; 24: 661–669.
Gleich LL, Gluckman JL, Armstrong S, Biddinger PW, Miller MA, Balakrishnan K et al. Alloantigen gene therapy for squamous cell carcinoma of the head and neck: results of a phase 1 trial. Arch Otolaryngol Head Neck Surg 1998; 124: 1097–1104.
Gleich LL, Gluckman JL, Nemunaitis J, Suen JY, Hanna E, Wolf GT et al. Clinical experience with HLA-B7 plasmid DNA/lipid complex in advanced squamous cell carcinoma of the head and neck. Arch Otolaryngol Head Neck Surg 2001; 127: 775–779.
Grandis JR, Chakraborty A, Melhem MF, Zeng Q, Tweardy DJ . Inhibition of epidermal growth factor receptor gene expression and function decreases proliferation of head and neck squamous carcinoma but not normal mucosal epithelial cells. Oncogene 1997; 15: 409–416.
Yuen AR, Halsey J, Fisher GA, Holmlund JT, Geary RS, Kwoh TJ et al. Phase I study of an antisense oligonucleotide to protein kinase C-α (ISIS 3521/CGP 64128A) in patients with cancer. Clin Cancer Res 1999; 5: 3357–3363.
Thomas SM, Sahu B, Rapireddy S, Bahal R, Wheeler SE, Procopio EM et al. Antitumor effects of EGFR antisense guanidine-based peptide nucleic acids in cancer models. ACS Chem Biol 2012; 8: 345–352.
Thomas SM, Ogagan MJ, Freilino ML, Strychor S, Walsh DR, Gooding WE et al. Antitumor mechanisms of systemically administered epidermal growth factor receptor antisense oligonucleotides in combination with docetaxel in squamous cell carcinoma of the head and neck. Mol Pharmacol 2008; 73: 627–638.
Demidov VV, Potaman VN, Frank-Kamenetskil MD, Egholm M, Buchard O, Sönnichsen SH et al. Stability of peptide nucleic acids in human serum and cellular extracts. Biochem Pharmacol 1994; 48: 1310–1313.
Sahu B, Chenna V, Lathrop KL, Thomas SM, Zon G, Livak KJ et al. Synthesis of conformationally preorganized and cell-permeable guanidine-based gamma-peptide nucleic acids (gammaGPNAs). J Org Chem 2009; 74: 1509–1516.
Leong PL, Andrews GA, Johnson DE, Dyer KF, Xi S, Mai JC et al. Targeted inhibition of Stat3 with a decoy oligonucleotide abrogates head and neck cancer cell growth. Proc Natl Acad Sci USA 2003; 100: 4138–4143.
Boehm AL, Sen M, Seethala R, Gooding WE, Freilino M, Wong SMY et al. Combined targeting of epidermal growth factor receptor, signal transducer and activator of transcription-3, and Bcl-XL enhances antitumor effects in squamous cell carcinoma of the head and neck. Mol Pharmacol 2008; 73: 1632–1642.
Klein JD, Sano D, Sen M, Myers JN, Grandis JR, Kim S . STAT3 oligonucleotide inhibits tumor angiogenesis in preclinical models of squamous cell carcinoma. PloS one 2014; 9: e81819.
Jing N, Zhu Q, Yuan P, Li Y, Mao L, Tweardy DJ . Targeting signal transducer and activator of transcription 3 with G-quartet oligonucleotides: a potential novel therapy for head and neck cancer. Mol Cancer Ther 2006; 5: 279–286.
Guttmann DM, Hart L, Du K, Seletsky A, Koumenis C . Inhibition of Hsp27 radiosensitizes head-and-neck cancer by modulating deoxyribonucleic acid repair. Int J Radiat Oncol Biol Phys 2013; 87: 168–175.
Graff JR, Konicek BW, Vincent TM, Lynch RL, Monteith D, Weir SN et al. Therapeutic suppression of translation initiation factor eIF4E expression reduces tumor growth without toxicity. J Clin Invest 2007; 117: 2638–2648.
Hong DS, Kurzrock R, Oh Y, Wheler J, Naing A, Brail L et al. A phase 1 dose escalation, pharmacokinetic, and pharmacodynamic evaluation of eIF-4E antisense oligonucleotide LY2275796 in patients with advanced cancer. Clin Cancer Res 2011; 17: 6582–6591.
Sen M, Thomas SM, Kim S, Yeh JI, Ferris RL, Johnson JT et al. First-in-human trial of a STAT3 decoy oligonucleotide in head and neck tumors: implications for cancer therapy. Cancer Discov 2012; 2: 694–705.
Mansoori B, Shotorbani SS, Baradaran B . RNA interference and its role in cancer therapy. Adv Pharm Bull 2014; 4: 313–321.
Fabian MR, Sonenberg N . The mechanics of miRNA-mediated gene silencing: a look under the hood of miRISC. Nat Struct Mol Biol 2012; 19: 586–593.
Jackson AL, Burchard J, Schelter J, Chau BN, Cleary M, Lim L et al. Widespread siRNA "off-target" transcript silencing mediated by seed region sequence complementarity. RNA 2006; 12: 1179–1187.
Persengiev SP, Zhu X, Green MR . Nonspecific, concentration-dependent stimulation and repression of mammalian gene expression by small interfering RNAs (siRNAs). RNA 2004; 10: 12–18.
Doench JG, Petersen CP, Sharp PA . siRNAs can function as miRNAs. Genes Dev 2003; 17: 438–442.
Pan Q, Bao LW, Teknos TN, Merajver SD . Targeted disruption of protein kinase Cɛ reduces cell invasion and motility through inactivation of RhoA and RhoC GTPases in head and neck squamous cell carcinoma. Cancer Res 2006; 66: 9379–9384.
Zhang Q, Zhang ZF, Rao JY, Sato JD, Brown J, Messadi DV et al. Treatment with siRNA and antisense oligonucleotides targeted to HIF‐1α induced apoptosis in human tongue squamous cell carcinomas. Int J Cancer 2004; 111: 849–857.
Mazumdar A, Henderson YC, El‐Naggar AK, Sen S, Clayman GL . Aurora kinase A inhibition and paclitaxel as targeted combination therapy for head and neck squamous cell carcinoma. Head Neck 2009; 31: 625–634.
Ferris RL, Grandis JR . NF-κB gene signatures and p53 mutations in head and neck squamous cell carcinoma. Clin Cancer Res 2007; 13: 5663–5664.
Loercher A, Lee TL, Ricker JL, Howard A, Geoghegen J, Chen Z et al. Nuclear factor-κB is an important modulator of the altered gene expression profile and malignant phenotype in squamous cell carcinoma. Cancer Res 2004; 64: 6511–6523.
Duan J, Friedman J, Nottingham L, Chen Z, Ara G, Van Waes C . Nuclear factor-κB p65 small interfering RNA or proteasome inhibitor bortezomib sensitizes head and neck squamous cell carcinomas to classic histone deacetylase inhibitors and novel histone deacetylase inhibitor PXD101. Mol Cancer Ther 2007; 6: 37–50.
Lun M, Zhang PL, Pellitteri PK, Law A, Kennedy TL, Brown RE . Nuclear factor-κB pathway as a therapeutic target in head and neck squamous cell carcinoma: pharmaceutical and molecular validation in human cell lines using Velcade and siRNA/NF-κB. Ann Clin Lab Sci 2005; 35: 251–258.
Nozawa H, Tadakuma T, Ono T, Sato M, Hiroi S, Masumoto K et al. Small interfering RNA targeting epidermal growth factor receptor enhances chemosensitivity to cisplatin, 5‐fluorouracil and docetaxel in head and neck squamous cell carcinoma. Cancer Sci 2006; 97: 1115–1124.
Rahman MA, Amin ARM, Wang X, Zuckerman JE, Choi CHJ, Zhou B et al. Systemic delivery of siRNA nanoparticles targeting RRM2 suppresses head and neck tumor growth. J Control Release 2012; 159: 384–392.
Chen WH, Lecaros RL, Tseng YC, Huang L, Hsu YC . Nanoparticle delivery of HIF1alpha siRNA combined with photodynamic therapy as a potential treatment strategy for head-and-neck cancer. Cancer Lett 2015; 359: 65–74.
Rao DD, Vorhies JS, Senzer N, Nemunaitis J . siRNA vs. shRNA: similarities and differences. Adv Drug Deliv Rev 2009; 61: 746–759.
Deng Y, Wang CC, Choy KW, Du Q, Chen J, Wang Q et al. Therapeutic potentials of gene silencing by RNA interference: Principles, challenges, and new strategies. Gene 2014; 538: 217–227.
Kindt N, Lechien J, Nonclercq D, Laurent G, Saussez S . Involvement of CD74 in head and neck squamous cell carcinomas. J Cancer Res Clin Oncol 2014; 140: 937–947.
Sobral LM, Sousa LO, Coletta RD, Cabral H, Greene LJ, Tajara EH et al. Stable SET knockdown in head and neck squamous cell carcinoma promotes cell invasion and the mesenchymal-like phenotype in vitro, as well as necrosis, cisplatin sensitivity and lymph node metastasis in xenograft tumor models. Mol Cancer 2014; 13: 32–32.
Liu D, Tao Z-Z, Xiao B-K, Chen S, Chi H . Inhibitory effect of silencing hTERT gene by short hairpin RNA on growth of human laryngeal squamous cell carcinoma xenograft in nude mice. Ai Zheng 2006; 25: 11–16.
Hsin C-H, Chen M-K, Tang C-H, Lin H-P, Chou M-Y, Lin C-W et al. High level of plasma matrix metalloproteinase-11 is associated with clinicopathological characteristics in patients with oral squamous cell carcinoma. PLoS One 2014; 9: e113129.
Chen S-M, Wang Y, Xiao B-K, Tao Z-Z . Effect of blocking VEGF, hTERT and Bcl-xl by multiple shRNA expression vectors on the human laryngeal squamous carcinoma xenograft in nude mice. Cancer Biol Ther 2008; 7: 734–739.
Hazar-Rethinam M, Merida de Long L, Gannon O, Topkas E, Boros S, Vargas AC et al. A novel E2F/Sphingosine kinase 1 axis regulates anthracycline response in squamous cell carcinoma. Clin Cancer Res 2014; 21: 417–427.
Wang Y-D, Li S-J, Liao J-X . Inhibition of glucose transporter 1 (GLUT1) chemosensitized head and neck cancer cells to cisplatin. Technol Cancer Res Treat 2013; 12: 525–535.
Kothari V, Mulherkar R . Inhibition of cyclin D1 by shRNA is associated with enhanced sensitivity to conventional therapies for head and neck squamous cell carcinoma. Anticancer Res 2012; 32: 121–128.
Tassone P, Old M, Teknos TN, Pan Q . p53-based therapeutics for head and neck squamous cell carcinoma. Oral Oncol 2013; 49: 733–737.
Liu T-J, El-Naggar AK, McDonnell TJ, Steck KD, Wang M, Taylor DL et al. Apoptosis induction mediated by wild-type p53 adenoviral gene transfer in squamous cell carcinoma of the head and neck. Cancer Res 1995; 55: 3117–3122.
Xu L, Tang W-H, Huang C-C, Alexander W, Xiang L-M, Pirollo KF et al. Systemic p53 gene therapy of cancer with immunolipoplexes targeted by anti-transferrin receptor scFv. Mol Med 2001; 7: 723.
Chang EH, Jang Y-J, Hao Z, Murphy G, Rait A, Fee WE Jr et al. Restoration of the G1 checkpoint and the apoptotic pathway mediated by wild-type p53 sensitizes squamous cell carcinoma of the head and neck to radiotherapy. Arch Otolaryngol Head Neck Surg 1997; 123: 507.
Clayman GL, Frank DK, Bruso PA, Goepfert H . Adenovirus-mediated wild-type p53 gene transfer as a surgical adjuvant in advanced head and neck cancers. Clin Cancer Res 1999; 5: 1715–1722.
O'Malley BW, Chen S-H, Schwartz MR, Woo SLC . Adenovirus-mediated gene therapy for human head and neck squamous cell cancer in a nude mouse model. Cancer Res 1995; 55: 1080–1085.
Morris JC, Wildner O . Therapy of head and neck squamous cell carcinoma with an oncolytic adenovirus expressing HSV-tk. Mol Ther 2000; 1: 56–62.
Kanazawa T, Mizukami H, Okada T, Hanazono Y, Kume A, Nishino H et al. Suicide gene therapy using AAV-HSVtk/ganciclovir in combination with irradiation results in regression of human head and neck cancer xenografts in nude mice. Gene Ther 2003; 10: 51–58.
Masuelli L, Fantini M, Benvenuto M, Sacchetti P, Giganti MG, Tresoldi I et al. Intratumoral delivery of recombinant vaccinia virus encoding for ErbB2/Neu inhibits the growth of salivary gland carcinoma cells. J Transl Med 2014; 12: 122.
Yoo GH, Moon J, LeBlanc M, Lonardo F, Urba S, Kim H et al. A phase 2 trial of surgery with perioperative INGN 201 (Ad5CMV-p53) gene therapy followed by chemoradiotherapy for advanced, resectable squamous cell carcinoma of the oral cavity, oropharynx, hypopharynx, and larynx: report of the Southwest Oncology Group. Arch Otolaryngol Head Neck Surg 2009; 135: 869–874.
Li Y, Li L-J, Wang L-J, Zhang Z, Gao N, Liang C-Y et al. Selective intra-arterial infusion of rAd-p53 with chemotherapy for advanced oral cancer: a randomized clinical trial. BMC Med 2014; 12: 16–16.
Nemunaitis J, Clayman G, Agarwala SS, Hrushesky W, Wells JR, Moore C et al. Biomarkers predict p53 gene therapy efficacy in recurrent squamous cell carcinoma of the head and neck. Clin Cancer Res 2009; 15: 7719–7725.
Mace ATM, Harrow SJ, Ganly I, Brown SM . Cytotoxic effects of the oncolytic herpes simplex virus HSV1716 alone and in combination with cisplatin in head and neck squamous cell carcinoma. Acta Otolaryngol (Stockh) 2007; 127: 880–887.
Chahlavi A, Todo T, Martuza RL, Rabkin SD . Replication-competent herpes simplex virus vector G207 and cisplatin combination therapy for head and neck squamous cell carcinoma. Neoplasia 1999; 1: 162–169.
Fujimoto Y, Mizuno T, Sugiura S, Goshima F, Kohno S-I, Nakashima T et al. Intratumoral injection of herpes simplex virus HF10 in recurrent head and neck squamous cell carcinoma. Acta Otolaryngol (Stockh) 2006; 126: 1115–1117.
Bischoff JR, Kirn DH, Williams A, Heise C, Horn S, Muna M et al. An adenovirus mutant that replicates selectively in p53-deficient human tumor cells. Science 1996; 274: 373–376.
Heise CC, Williams AM, Xue S, Propst M, Kirn DH . Intravenous administration of ONYX-015, a selectively replicating adenovirus, induces antitumoral efficacy. Cancer Res 1999; 59: 2623–2628.
Vannucci L, Lai M, Chiuppesi F, Ceccherini-Nelli L, Pistello M . Viral vectors: a look back and ahead on gene transfer technology. New Microbiol 2013; 36: 1–22.
Shin EJ, Wanna GB, Choi B, Aguila D, Ebert O, Genden EM et al. Interleukin‐12 expression enhances vesicular stomatitis virus oncolytic therapy in murine squamous cell carcinoma. Laryngoscope 2007; 117: 210–214.
Zaoui K, Bossow S, Grossardt C, Leber MF, Springfeld C, Plinkert PK et al. Chemovirotherapy for head and neck squamous cell carcinoma with EGFR-targeted and CD/UPRT-armed oncolytic measles virus. Cancer Gene Ther 2011; 19: 181–191.
Mace A, Ganly I, Soutar DS, Brown SM . Potential for efficacy of the oncolytic Herpes simplex virus 1716 in patients with oral squamous cell carcinoma. Head Neck 2008; 30: 1045–1051.
Ganly I, Kirn D, Eckhardt SG, Rodriguez GI, Soutar DS, Otto R et al. A phase I study of Onyx-015, an E1B attenuated adenovirus, administered intratumorally to patients with recurrent head and neck cancer. Clin Cancer Res 2000; 6: 798–806.
Nemunaitis J, Khuri F, Ganly I, Arseneau J, Posner M, Vokes E et al. Phase II trial of intratumoral administration of ONYX-015, a replication-selective adenovirus, in patients with refractory head and neck cancer. J Clin Oncol 2001; 19: 289–298.
Lamont JP, Nemunaitis J, Kuhn JA, Landers SA, McCarty TM . A prospective phase II trial of ONYX-015 adenovirus and chemotherapy in recurrent squamous cell carcinoma of the head and neck (the Baylor experience). Ann Surg Oncol 2000; 7: 588–592.
Garber K . China approves world's first oncolytic virus therapy for cancer treatment. J Natl Cancer Inst 2006; 98: 298–300.
Hu JC, Coffin RS, Davis CJ, Graham NJ, Groves N, Guest PJ et al. A phase I study of OncoVEXGM-CSF, a second-generation oncolytic herpes simplex virus expressing granulocyte macrophage colony-stimulating factor. Clin Cancer Res 2006; 12: 6737–6747.
Senzer NN, Kaufman HL, Amatruda T, Nemunaitis M, Reid T, Daniels G et al. Phase II clinical trial of a granulocyte-macrophage colony-stimulating factor-encoding, second-generation oncolytic herpesvirus in patients with unresectable metastatic melanoma. J Clin Oncol 2009; 27: 5763–5771.
Kaufman HL, Kim DW, DeRaffele G, Mitcham J, Coffin RS, Kim-Schulze S . Local and distant immunity induced by intralesional vaccination with an oncolytic herpes virus encoding GM-CSF in patients with stage IIIc and IV melanoma. Ann Surg Oncol 2010; 17: 718–730.
Hu JCC, Coffin RS, Davis CJ, Graham NJ, Groves N, Guest PJ et al. A phase I study of OncoVEXGM-CSF, a second-generation oncolytic herpes simplex virus expressing granulocyte macrophage colony-stimulating factor. Clin Cancer Res 2006; 12: 6737–6747.
Hallahan DE, Mauceri HJ, Seung LP, Dunphy EJ, Wayne JD, Hanna NN et al. Spatial and temporal control of gene therapy using ionizing radiation. Nat Med 1995; 1: 786–791.
Seiwert TY, Darga T, Haraf D, Blair EA, Stenson K, Cohen EEW et al. A phase I dose escalation study of Ad GV.EGR.TNF.11D (TNFerade™ Biologic) with concurrent chemoradiotherapy in patients with recurrent head and neck cancer undergoing reirradiation. Ann Oncol 2013; 24: 769–776.
Shimizu S, Tsukada J, Sugimoto T, Kikkawa N, Sasaki K, Chazono H et al. Identification of a novel therapeutic target for head and neck squamous cell carcinomas: a role for the neurotensin‐neurotensin receptor 1 oncogenic signaling pathway. Int J Cancer 2008; 123: 1816–1823.
Wang Y, Tao Z-Z, Chen S-M, Xiao B-K, Zhou X-H, Liu J-P . Application of combination of short hairpin RNA segments for silencing VEGF, TERT, and Bcl-xl expression in laryngeal squamous carcinoma. Cancer Biol Ther 2008; 7: 896–901.
Clayman GL, El-Naggar AK, Lippman SM, Henderson YC, Frederick M, Merritt JA et al. Adenovirus-mediated p53 gene transfer in patients with advanced recurrent head and neck squamous cell carcinoma. J Clin Oncol 1998; 16: 2221–2232.
Zhang SW, Xiao SW, Liu CQ, Sun Y, Su X, Li DM et al. [Treatment of head and neck squamous cell carcinoma by recombinant adenovirus-p53 combined with radiotherapy: a phase II clinical trial of 42 cases]. Zhonghua Yi Xue Za Zhi 2003; 83: 2023–2028.
Acknowledgements
We thank Dr Hinrich Staecker, Department of Otolaryngology, University of Kansas Medical Center, Kansas City, KS and Dr Stefan Rothenberg, K-State University, Manhattan, KS for critical review of the manuscript. We also thank Mr Phil Shafer for the artwork. We apologize to authors whose work was not cited due to space constraints. Department of Otolaryngology, University of Kansas Medical Center and University of Kansas Cancer Center’s CCSG (1-P30-CA168524-02), and the Kansas Intellectual and Developmental Disabilities Center (NICHD HD00258) were the funding sources.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Parsel, S., Grandis, J. & Thomas, S. Nucleic acid targeting: towards personalized therapy for head and neck cancer. Oncogene 35, 3217–3226 (2016). https://doi.org/10.1038/onc.2015.424
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2015.424