Abstract
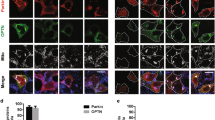
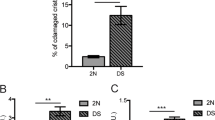
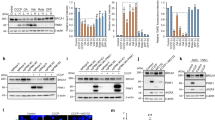
PINK1 (phosphatase and tensin homolog deleted on chromosome 10 (PTEN)-induced kinase 1), a Parkinson’s disease-associated gene, was identified originally because of its induction by the tumor-suppressor PTEN. PINK1 promotes cell survival and potentially metastatic functions and protects against cell stressors including chemotherapeutic agents. However, the mechanisms underlying PINK1 function in cancer cell biology are unclear. Here, using several model systems, we show that PINK1 deletion significantly reduced cancer-associated phenotypes including cell proliferation, colony formation and invasiveness, which were restored by human PINK1 overexpression. Results show that PINK1 deletion causes major defects in cell cycle progression in immortalized mouse embryonic fibroblasts (MEFs) from PINK1−/− mice, and in BE(2)-M17 cells stably transduced with short hairpin RNA against PINK1. Detailed cell cycle analyses of MEF cell lines from several PINK1−/− mice demonstrate an increased proportion of cells in G2/M and decreased number of cells in G1 following release from nocodazole block. This was concomitant with increased double and multi-nucleated cells, a reduced ability to undergo cytokinesis and to re-enter G1, and significant alterations in cell cycle markers, including failure to increase cyclin D1, all indicative of mitotic arrest. PINK1−/− cells also demonstrated ineffective cell cycle exit following serum deprivation. Cell cycle defects associated with PINK1 deficiency occur at points critical for cell division, growth and stress resistance in cancer cells were rescued by ectopic expression of human PINK1 and demonstrated PINK1 kinase dependence. The importance of PINK1 for cell cycle control is further supported by results showing that cell cycle deficits induced by PINK1 deletion were linked mechanistically to aberrant mitochondrial fission and its regulation by dynamin-related protein-1 (Drp1), known to be critical for progression of mitosis. Our data indicate that PINK1 has tumor-promoting properties and demonstrates a new function for PINK1 as a regulator of the cell cycle.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Unoki M, Nakamura Y . Growth-suppressive effects of BPOZ and EGR2, two genes involved in the PTEN signaling pathway. Oncogene 2001; 20: 4457–4465.
Valente EM, Abou-Sleiman PM, Caputo V, Muqit MMK, Harvey K, Gispert S et al. Hereditary early-onset Parkinson’s disease caused by mutations in PINK1. Science 2004; 304: 1158–1160.
Dodson MW, Guo M . Pink1, Parkin, DJ-1 and mitochondrial dysfunction in Parkinson’s disease. Curr Opin Neurobiol 2007; 17: 331–337.
Gispert S, Ricciardi F, Kurz A, Azizov M, Hoepken H-H, Becker D et al. Parkinson phenotype in aged PINK1-deficient mice is accompanied by progressive mitochondrial dysfunction in absence of neurodegeneration. PloS One 2009; 4: e5777.
Haque ME, Thomas KJ, D’Souza C, Callaghan S, Kitada T, Slack RS et al. Cytoplasmic Pink1 activity protects neurons from dopaminergic neurotoxin MPTP. Proc Natl Acad Sci USA 2008; 105: 1716–1721.
Petit A, Kawarai T, Paitel E, Sanjo N, Maj M, Scheid M et al. Wild-type PINK1 prevents basal and induced neuronal apoptosis, a protective effect abrogated by Parkinson disease-related mutations. J Biol Chem 2005; 280: 34025–34032.
Kitada T, Pisani A, Porter DR, Yamaguchi H, Tscherter A, Martella G et al. Impaired dopamine release and synaptic plasticity in the striatum of PINK1-deficient mice. Proc Natl Acad Sci USA 2007; 104: 11441–11446.
Wang H-L, Chou A-H, Yeh T-H, Li AH, Chen Y-L, Kuo Y-L et al. PINK1 mutants associated with recessive Parkinson’s disease are defective in inhibiting mitochondrial release of cytochrome c. Neurobiol Dis 2007; 28: 216–226.
Silvestri L, Caputo V, Bellacchio E, Atorino L, Dallapiccola B, Valente EM et al. Mitochondrial import and enzymatic activity of PINK1 mutants associated to recessive parkinsonism. Human Mol Genet 2005; 14: 3477–3492.
Cardona F, Sánchez-Mut JV, Dopazo H, Pérez-Tur J . Phylogenetic and in silico structural analysis of the Parkinson disease-related kinase PINK1. Hum Mutat 2011; 32: 369–378.
Gandhi S, Muqit MMK, Stanyer L, Healy DG, Abou-Sleiman PM, Hargreaves I et al. PINK1 protein in normal human brain and Parkinson’s disease. Brain 2006; 129: 1720–1731.
Liu W, Acín-Peréz R, Geghman KD, Manfredi G, Lu B, Li C . Pink1 regulates the oxidative phosphorylation machinery via mitochondrial fission. Proc Natl Acad Sci USA 2011; 108: 12920–12924.
Sandebring A, Thomas KJ, Beilina A, Van der Brug M, Cleland MM, Ahmad R et al. Mitochondrial alterations in PINK1 deficient cells are influenced by calcineurin-dependent dephosphorylation of dynamin-related protein 1. PLoS One 2009; 4: e5701.
Morais VA, Verstreken P, Roethig A, Smet J, Snellinx A, Vanbrabant M et al. Parkinson’s disease mutations in PINK1 result in decreased Complex I activity and deficient synaptic function. EMBO Mol Med 2009; 1: 99–111.
Pridgeon JW, Olzmann Ja, Chin L-S, Li L . PINK1 protects against oxidative stress by phosphorylating mitochondrial chaperone TRAP1. PLoS Biol 2007; 5: e172.
Arena G, Gelmetti V, Torosantucci L, Vignone D, Lamorte G, De Rosa P et al. PINK1 protects against cell death induced by mitochondrial depolarization, by phosphorylating Bcl-xL and impairing its pro-apoptotic cleavage. Cell Death Differ 2013; 20: 920–930.
Murata H, Sakaguchi M, Jin Y, Sakaguchi Y, Futami J, Yamada H et al. A new cytosolic pathway from a Parkinson disease-associated kinase, BRPK/PINK1: activation of AKT via mTORC2. J Biol Chem 2011; 286: 7182–7189.
Akundi RS, Zhi L, Büeler H . PINK1 enhances insulin-like growth factor-1-dependent Akt signaling and protection against apoptosis. Neurobiol Dis 2012; 45: 469–478.
Rakovic A, Grünewald A, Kottwitz J, Brüggemann N, Pramstaller PP, Lohmann K et al. Mutations in PINK1 and Parkin impair ubiquitination of mitofusins in human fibroblasts. PLoS One 2011; 6: e16746.
Klinkenberg M, Thurow N, Gispert S, Ricciardi F, Eich F, Prehn JHM et al. Enhanced vulnerability of PARK6 patient skin fibroblasts to apoptosis induced by proteasomal stress. Neuroscience 2010; 166: 422–434.
Muqit MMK, Abou-Sleiman PM, Saurin AT, Harvey K, Gandhi S, Deas E et al. Altered cleavage and localization of PINK1 to aggresomes in the presence of proteasomal stress. J Neurochem 2006; 98: 156–169.
Michiorri S, Gelmetti V, Giarda E, Lombardi F, Romano F, Marongiu R et al. The Parkinson-associated protein PINK1 interacts with Beclin1 and promotes autophagy. Cell Death Differ 2010; 17: 962–974.
Kawajiri S, Saiki S, Sato S, Sato F, Hatano T, Eguchi H et al. PINK1 is recruited to mitochondria with parkin and associates with LC3 in mitophagy. FEBS Lett 2010; 584: 1073–1079.
Berthier A, Navarro S, Jiménez-Sáinz J, Roglá I, Ripoll F, Cervera J et al. PINK1 displays tissue-specific subcellular location and regulates apoptosis and cell growth in breast cancer cells. Human Pathol 2011; 42: 75–87.
Martin SA, Hewish M, Sims D, Lord CJ, Ashworth A . Parallel high-throughput RNA interference screens identify PINK1 as a potential therapeutic target for the treatment of DNA mismatch repair-deficient cancers. Cancer Res 2011; 71: 1836–1848.
MacKeigan JP, Murphy LO, Blenis J . Sensitized RNAi screen of human kinases and phosphatases identifies new regulators of apoptosis and chemoresistance. Nat Cell Biol 2005; 7: 591–600.
Mei Y, Zhang Y, Yamamoto K, Xie W, Mak TW, You H . FOXO3a-dependent regulation of Pink1 (Park6) mediates survival signaling in response to cytokine deprivation. Proc Natl Acad Sci USA 2009; 106: 5153–5158.
Jin SM, Youle RJ . PINK1- and Parkin-mediated mitophagy at a glance. J Cell Sci 2012; 125: 795–799.
Wilhelmus M, Nijland P . Involvement and interplay of Parkin, PINK1 and DJ1 in neurodegenerative and neuroinflammatory disorders. Free Radic Biol Med 2012; 53: 983–992.
Fujiwara M, Marusawa H, Wang H-Q, Iwai a, Ikeuchi K, Imai Y et al. Parkin as a tumor suppressor gene for hepatocellular carcinoma. Oncogene 2008; 27: 6002–6011.
Veeriah S, Taylor BS, Meng S, Fang F, Yilmaz E, Vivanco I et al. Somatic mutations of the Parkinson’s disease-associated gene PARK2 in glioblastoma and other human malignancies. Nat Genet 2010; 42: 77–82.
Sun X, Liu M, Hao J, Li D, Luo Y, Wang X et al. Parkin deficiency contributes to pancreatic tumorigenesis by inducing spindle multipolarity and misorientation. Cell Cycle 2013; 12: 1133–1141.
Bagchi A, Mills AA . The quest for the 1p36 tumor suppressor. Cancer Res 2008; 68: 2551–2256.
Bernard D, Monte D, Vandenbunder B, Abbadie C . The c-Rel transcription factor can both induce and inhibit apoptosis in the same cells via the upregulation of MnSOD. Oncogene 2002; 21: 4392–4402.
Chaiwatanasirikul KA, Sala A . The tumour-suppressive function of CLU is explained by its localisation and interaction with HSP60. Cell Death Dis 2011; 2: e219.
Youle RJ, Karbowski M . Mitochondrial fission in apoptosis. Nat Rev Mol Cell Biol 2005; 6: 657–663.
Youle RJ, Van der Bliek AM . Mitochondrial fission, fusion, and stress. Science 2012; 337: 1062–1065.
Chatterjee A, Mambo E, Sidransky D . Mitochondrial DNA mutations in human cancer. Oncogene 2006; 25: 4663–4674.
Grandemange S, Herzig S, Martinou J-C . Mitochondrial dynamics and cancer. Semin Cancer Biol 2009; 19: 50–56.
Favre C, Zhdanov a, Leahy M, Papkovsky D, O’Connor R . Mitochondrial pyrimidine nucleotide carrier (PNC1) regulates mitochondrial biogenesis and the invasive phenotype of cancer cells. Oncogene 2010; 29: 3964–3976.
Barbosa IA, Machado NG, Skildum AJ, Scott PM, Oliveira PJ . Mitochondrial remodeling in cancer metabolism and survival: Potential for new therapies. Biochim Biophy Acta 2012; 1826: 238–254.
Poole AC, Thomas RE, Andrews LA, McBride HM, Whitworth AJ, Pallanck LJ . The PINK1/Parkin pathway regulates mitochondrial morphology. Proc Natl Acad Sci USA 2008; 105: 1638–1643.
Deng H, Dodson MW, Huang H, Guo M . The Parkinson’s disease genes pink1 and parkin promote mitochondrial fission and/or inhibit fusion in Drosophila. Proc Natl Acad Sci USA 2008; 105: 14503–14508.
Poole AC, Thomas RE, Yu S, Vincow ES, Pallanck L . The mitochondrial fusion-promoting factor mitofusin is a substrate of the PINK1/parkin pathway. PLoS One 2010; 5: e10054.
Lutz AK, Exner N, Fett ME, Schlehe JS, Kloos K, Lämmermann K et al. Loss of parkin or PINK1 function increases Drp1-dependent mitochondrial fragmentation. J Biol Chem 2009; 284: 22938–22951.
Taguchi N, Ishihara N, Jofuku A, Oka T, Mihara K . Mitotic phosphorylation of dynamin-related GTPase Drp1 participates in mitochondrial fission. J Biol Chem 2007; 282: 11521–11529.
Kashatus DF, Lim K-H, Brady DC, Pershing NLK, Cox AD, Counter CM . RALA and RALBP1 regulate mitochondrial fission at mitosis. Nature Cell Biol 2011; 13: 1108–1115.
Horn SR, Thomenius MJ, Johnson ES, Freel CD, Wu JQ, Coloff JL et al. Regulation of mitochondrial morphology by APC/CCdh1-mediated control of Drp1 stability. Mol Biol Cell 2011; 22: 1207–1216.
Mitra K, Wunder C, Roysam B, Lin G, Lippincott-Schwartz J . A hyperfused mitochondrial state achieved at G1-S regulates cyclin E buildup and entry into S phase. Proc Natl Acad Sci USA 2009; 106: 11960–11965.
Rambold AS, Kostelecky B, Elia N, Lippincott-Schwartz J . Tubular network formation protects mitochondria from autophagosomal degradation during nutrient starvation. Proc Natl Acad Sci USA 2011; 108: 10190–10195.
Gomes LC, Di Benedetto G, Scorrano L . During autophagy mitochondria elongate, are spared from degradation and sustain cell viability. Nat Cell Biol 2011; 13: 589–598.
Beilina A, Van Der Brug M, Ahmad R, Kesavapany S, Miller DW, Petsko G et al. Mutations in PTEN-induced putative kinase 1 associated with recessive parkinsonism have differential effects on protein stability. Proc Natl Acad Sci USA 2005; 102: 5703–5708.
Cho K, Ryu SJ, Oh YS, Park JH, Lee JW, Kim H-P et al. Morphological adjustment of senescent cells by modulating caveolin-1 status. J Biol Chem 2004; 279: 42270–42278.
Dagda RK, Cherra SJ, Kulich SM, Tandon A, Park D, Chu CT . Loss of PINK1 function promotes mitophagy through effects on oxidative stress and mitochondrial fission. J Biol Chem 2009; 284: 13843–13855.
Smirnova E, Griparic L, Shurland DL, Van der Blieka M . Dynamin-related protein Drp1 is required for mitochondrial division in mammalian cells. Mol Biol Cell 2001; 12: 2245–2256.
Yamano K, Youle RJ . Coupling mitochondrial and cell division. Nat Cell Biol 2011; 13 (9): 1026–1027.
Marongiu R, Spencer B, Crews L, Adame A, Patrick C, Trejo M et al. Mutant Pink1 induces mitochondrial dysfunction in a neuronal cell model of Parkinson’s disease by disturbing calcium flux. J Neurochem 2009; 108: 1561–1574.
Heeman B, Van den Haute C, Aelvoet S-A, Valsecchi F, Rodenburg RJ, Reumers V et al. Depletion of PINK1 affects mitochondrial metabolism, calcium homeostasis and energy maintenance. J Cell Sci 2011; 124: 1115–1125.
Akundi RS, Huang Z, Eason J, Pandya JD, Zhi L, Cass WA et al. Increased mitochondrial calcium sensitivity and abnormal expression of innate immunity genes precede dopaminergic defects in Pink1-deficient mice. PLoS One 2011; 6: e16038.
Park J, Lee G, Chung J . The PINK1-Parkin pathway is involved in the regulation of mitochondrial remodeling process. Biochem Biophys Res Comm 2009; 378: 518–523.
Hanahan D, Weinberg RA . Hallmarks of cancer: the next generation. Cell 2011; 144: 646–674.
Knudsen KE, Diehl JA, Haiman CA, Knudsen ES . Cyclin D1: polymorphism, aberrant splicing and cancer risk. Oncogene 2006; 25: 1620–1628.
Bharadwaj R, Yu H . The spindle checkpoint, aneuploidy, and cancer. Oncogene 2004; 23: 2016–2027.
Burgess DJ . Aneuploidy stokes the fire. Nat Rev Cancer 2011; 11: 692.
Bonda DJ, Bajić VP, Spremo-Potparevic B, Casadesus G, Zhu X, Smith M a et al. Review: cell cycle aberrations and neurodegeneration. Neuropathol Appl Neurobiol 2010; 36: 157–163.
Herrup K . The involvement of cell cycle events in the pathogenesis of Alzheimer’s disease. Alzheimers Res Ther 2010; 2: 13.
Herrup K, Yang Y . Cell cycle regulation in the postmitotic neuron: oxymoron or new biology? Nat Rev Neurosci 2007; 8: 368–378.
Staropoli JF . Tumorigenesis and neurodegeneration: two sides of the same coin? BioEssays 2008; 30: 719–727.
Ayllón V, O’connor R . PBK/TOPK promotes tumour cell proliferation through p38 MAPK activity and regulation of the DNA damage response. Oncogene 2007; 26: 3451–3461.
Liang C-C, Park AY, Guan J-L . In vitro scratch assay: a convenient and inexpensive method for analysis of cell migration in vitro. Nat Protoc 2007; 2: 329–333.
Acknowledgements
We are grateful to Mark Cookson and Alexandra Beilina, National Institute on Aging, Bethesda, MD, USA for providing us with BE(2)-M17 cells transduced with control and PINK1 shRNA. We thank Rosemary O’Connor, School of Biochemistry and Cell Biology, University College Cork (UCC) for many helpful discussions, Sandra Yeomans UCC for technical assistance and Daniela Vogt-Weisenhorn, Helmholtz Centrum München, for helpful input. This work was funded by the Health Research Board of Ireland, PhD Scholars’ Programme in Cancer Biology. Support from Science Foundation Ireland (SFI) (RFP), the FWO Foundation for Scientific Research Belgium, a Methusalem grant of the Flemish Government and the KU Leuven, and the Helmholtz Alliance for Mental Health in an Ageing Society is also gratefully acknowledged. BDS is the Arthur Bax and Anna Vanluffelen Chair for Alzheimer’s disease. The Molecular Cell Biology group, UCC, provided access to a Zeiss 510 Confocal microscope, funded by an SFI Programme Grant to Mary W McCaffrey.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on the Oncogene website
Supplementary information
Rights and permissions
About this article
Cite this article
O'Flanagan, C., Morais, V., Wurst, W. et al. The Parkinson’s gene PINK1 regulates cell cycle progression and promotes cancer-associated phenotypes. Oncogene 34, 1363–1374 (2015). https://doi.org/10.1038/onc.2014.81
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2014.81
This article is cited by
-
PINK1 regulates apoptosis of osteosarcoma as the target gene of cisplatin
Journal of Orthopaedic Surgery and Research (2023)
-
MicroRNAs in Parkinson’s disease: a systematic review and diagnostic accuracy meta-analysis
Scientific Reports (2023)
-
Mitochondrial adaptation in cancer drug resistance: prevalence, mechanisms, and management
Journal of Hematology & Oncology (2022)
-
BRCA1 degradation in response to mitochondrial damage in breast cancer cells
Scientific Reports (2021)
-
Endosomes, lysosomes, and the role of endosomal and lysosomal biogenesis in cancer development
Molecular Biology Reports (2020)