Abstract
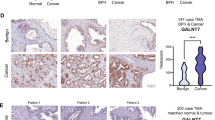
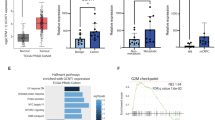
Prostate cancer is the second most common cause of cancer-associated deaths in men, and signaling via a transcription factor called androgen receptor (AR) is an important driver of the disease. Consequently, AR target genes are prominent candidates to be specific for prostate cancer and also important for the survival of the cancer cells. Here we assess the levels of all hexosamine biosynthetic pathway (HBP) enzymes in 15 separate clinical gene expression data sets and identify the last enzyme in the pathway, UDP-N-acetylglucosamine pyrophosphorylase 1 (UAP1), to be highly overexpressed in prostate cancer. We analyzed 3261 prostate cancers on a tissue microarray and found that UAP1 staining correlates negatively with Gleason score (P=0.0039) and positively with high AR expression (P<0.0001). Cells with high UAP1 expression have 10-fold increased levels of the HBP end-product, UDP-N-acetylglucosamine (UDP-GlcNAc). UDP-GlcNAc is essential for N-linked glycosylation occurring in the endoplasmic reticulum (ER) and high UAP1 expression associates with resistance against inhibitors of N-linked glycosylation (tunicamycin and 2-deoxyglucose) but not with a general ER stress-inducing agent, the calcium ionophore A23187. Knockdown of UAP1 expression re-sensitized cells towards inhibitors of N-linked glycosylation, as measured by proliferation and activation of ER stress markers. Taken together, we have identified an enzyme, UAP1, which is highly overexpressed in prostate cancer and protects cancer cells from ER stress conferring a growth advantage.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Harris WP, Mostaghel EA, Nelson PS, Montgomery B . Androgen deprivation therapy: progress in understanding mechanisms of resistance and optimizing androgen depletion. Nat Clin Pract Urol 2009; 6: 76–85.
Massie CE, Lynch A, Ramos-Montoya A, Boren J, Stark R, Fazli L et al. The androgen receptor fuels prostate cancer by regulating central metabolism and biosynthesis. EMBO J 2011; 30: 2719–2733.
Moon JS, Jin WJ, Kwak JH, Kim HJ, Yun MJ, Kim JW et al. Androgen stimulates glycolysis for de novo lipid synthesis by increasing the activities of hexokinase 2 and 6-phosphofructo-2-kinase/fructose-2,6-bisphosphatase 2 in prostate cancer cells. Biochem J 2011; 433: 225–233.
Humphrey PA . Diagnosis of adenocarcinoma in prostate needle biopsy tissue. J Clin Pathol 2007; 60: 35–42.
Luo J, Zha S, Gage WR, Dunn TA, Hicks JL, Bennett CJ et al. Alpha-methylacyl-CoA racemase: a new molecular marker for prostate cancer. Cancer Res 2002; 62: 2220–2226.
Kridel SJ, Axelrod F, Rozenkrantz N, Smith JW . Orlistat is a novel inhibitor of fatty acid synthase with antitumor activity. Cancer Res 2004; 64: 2070–2075.
Sartor O, Pal SK . Abiraterone and its place in the treatment of metastatic CRPC. Nat Rev Clin Oncol 2013; 10: 6–8.
Itkonen HM, Minner S, Guldvik IJ, Sandmann MJ, Tsourlakis MC, Berge V et al. O-GlcNAc transferase integrates metabolic pathways to regulate the stability of c-MYC in human prostate cancer. Cancer Res 2013; 73: 5277–5287.
Slawson C, Copeland RJ, Hart GW . O-GlcNAc signaling: a metabolic link between diabetes and cancer? Trends Biochem Sci 2010; 35: 547–555.
Wellen KE, Thompson CB . A two-way street: reciprocal regulation of metabolism and signalling. Nat Rev Mol Cell Biol 2012; 13: 270–276.
Butkinaree C, Park K, Hart GW . O-linked beta-N-acetylglucosamine (O-GlcNAc): extensive crosstalk with phosphorylation to regulate signaling and transcription in response to nutrients and stress. Biochim Biophys Acta 2010; 1800: 96–106.
Schwarz F, Aebi M . Mechanisms and principles of N-linked protein glycosylation. Curr Opin Struct Biol 2011; 21: 576–582.
Dennis JW, Lau KS, Demetriou M, Nabi IR . Adaptive regulation at the cell surface by N-glycosylation. Traffic 2009; 10: 1569–1578.
Lau KS, Partridge EA, Grigorian A, Silvescu CI, Reinhold VN, Demetriou M et al. Complex N-glycan number and degree of branching cooperate to regulate cell proliferation and differentiation. Cell 2007; 129: 123–134.
Chen R, Li J, Feng CH, Chen SK, Liu YP, Duan CY et al. c-Met function requires N-linked glycosylation modification of pro-Met. J Cell Biochem 2013; 114: 816–822.
Dricu A, Carlberg M, Wang M, Larsson O . Inhibition of N-linked glycosylation using tunicamycin causes cell death in malignant cells: role of down-regulation of the insulin-like growth factor 1 receptor in induction of apoptosis. Cancer Res 1997; 57: 543–548.
Itkonen HM, Mills IG . N-linked glycosylation supports cross-talk between receptor tyrosine kinases and androgen receptor. PLoS ONE 2013; 8: e65016.
Hagisawa S, Ohyama C, Takahashi T, Endoh M, Moriya T, Nakayama J et al. Expression of core 2 beta1,6-N-acetylglucosaminyltransferase facilitates prostate cancer progression. Glycobiology 2005; 15: 1016–1024.
Krzeslak A, Forma E, Bernaciak M, Romanowicz H, Brys M . Gene expression of O-GlcNAc cycling enzymes in human breast cancers. Clin Exp Med 2012; 12: 61–65.
Lynch TP, Ferrer CM, Jackson SR, Shahriari KS, Vosseller K, Reginato MJ . Critical role of O-Linked beta-N-acetylglucosamine transferase in prostate cancer invasion, angiogenesis, and metastasis. J Biol Chem 2012; 287: 11070–11081.
Mi W, Gu Y, Han C, Liu H, Fan Q, Zhang X et al. O-GlcNAcylation is a novel regulator of lung and colon cancer malignancy. Biochim Biophys Acta 2011; 1812: 514–519.
Rhodes DR, Yu J, Shanker K, Deshpande N, Varambally R, Ghosh D et al. ONCOMINE: a cancer microarray database and integrated data-mining platform. Neoplasia 2004; 6: 1–6.
Kilpinen S, Autio R, Ojala K, Iljin K, Bucher E, Sara H et al. Systematic bioinformatic analysis of expression levels of 17,330 human genes across 9,783 samples from 175 types of healthy and pathological tissues. Genome Biol 2008; 9: R139.
Diekman AB, Olson G, Goldberg E . Expression of the human antigen SPAG2 in the testis and localization to the outer dense fibers in spermatozoa. Mol Reprod Dev 1998; 50: 284–293.
Mio T, Yabe T, Arisawa M, Yamada-Okabe H . The eukaryotic UDP-N-acetylglucosamine pyrophosphorylases. Gene cloning, protein expression, and catalytic mechanism. J Biol Chem 1998; 273: 14392–14397.
Peneff C, Ferrari P, Charrier V, Taburet Y, Monnier C, Zamboni V et al. Crystal structures of two human pyrophosphorylase isoforms in complexes with UDPGlc(Gal)NAc: role of the alternatively spliced insert in the enzyme oligomeric assembly and active site architecture. EMBO J 2001; 20: 6191–6202.
Bello D, Webber MM, Kleinman HK, Wartinger DD, Rhim JS . Androgen responsive adult human prostatic epithelial cell lines immortalized by human papillomavirus 18. Carcinogenesis 1997; 18: 1215–1223.
Berthon P, Cussenot O, Hopwood L, Leduc A, Maitland N . Functional expression of sv40 in normal human prostatic epithelial and fibroblastic cells—differentiation pattern of nontumorigenic cell-lines. Int J Oncol 1995; 6: 333–343.
Nakajima K, Kitazume S, Angata T, Fujinawa R, Ohtsubo K, Miyoshi E et al. Simultaneous determination of nucleotide sugars with ion-pair reversed-phase HPLC. Glycobiology 2010; 20: 865–871.
Sreekumar A, Poisson LM, Rajendiran TM, Khan AP, Cao Q, Yu J et al. Metabolomic profiles delineate potential role for sarcosine in prostate cancer progression. Nature 2009; 457: 910–914.
Monauni T, Zenti MG, Cretti A, Daniels MC, Targher G, Caruso B et al. Effects of glucosamine infusion on insulin secretion and insulin action in humans. Diabetes 2000; 49: 926–935.
Wellen KE, Lu C, Mancuso A, Lemons JM, Ryczko M, Dennis JW et al. The hexosamine biosynthetic pathway couples growth factor-induced glutamine uptake to glucose metabolism. Genes Dev 2010; 24: 2784–2799.
Buse MG . Hexosamines, insulin resistance, and the complications of diabetes: current status. Am J Physiol Endocrinol Metab 2006; 290: E1–E8.
Bosch RR, Pouwels MJ, Span PN, Olthaar AJ, Tack CJ, Hermus AR et al. Hexosamines are unlikely to function as a nutrient-sensor in 3T3-L1 adipocytes: a comparison of UDP-hexosamine levels after increased glucose flux and glucosamine treatment. Endocrine 2004; 23: 17–24.
Patti ME, Virkamaki A, Landaker EJ, Kahn CR, Yki-Jarvinen H . Activation of the hexosamine pathway by glucosamine in vivo induces insulin resistance of early postreceptor insulin signaling events in skeletal muscle. Diabetes 1999; 48: 1562–1571.
Kurtoglu M, Gao N, Shang J, Maher JC, Lehrman MA, Wangpaichitr M et al. Under normoxia, 2-deoxy-D-glucose elicits cell death in select tumor types not by inhibition of glycolysis but by interfering with N-linked glycosylation. Mol Cancer Ther 2007; 6: 3049–3058.
Xi H, Kurtoglu M, Liu H, Wangpaichitr M, You M, Liu X et al. 2-Deoxy-D-glucose activates autophagy via endoplasmic reticulum stress rather than ATP depletion. Cancer Chemother Pharmacol 2011; 67: 899–910.
Gross BJ, Kraybill BC, Walker S . Discovery of O-GlcNAc transferase inhibitors. J Am Chem Soc 2005; 127: 14588–14589.
Bassik MC, Kampmann M . Knocking out the door to tunicamycin entry. Proc Natl Acad Sci USA 2011; 108: 11731–11732.
Keller RK, Boon DY, Crum FC . N-Acetylglucosamine- 1 -phosphate transferase from hen oviduct: solubilization, characterization, and inhibition by tunicamycin. Biochemistry 1979; 18: 3946–3952.
Grassian AR, Coloff JL, Brugge JS . Extracellular matrix regulation of metabolism and implications for tumorigenesis. Cold Spring Harb Symp Quant Biol 2011; 76: 313–324.
Ding WX, Ni HM, Gao W, Hou YF, Melan MA, Chen X et al. Differential effects of endoplasmic reticulum stress-induced autophagy on cell survival. J Biol Chem 2007; 282: 4702–4710.
Johansen T, Lamark T . Selective autophagy mediated by autophagic adapter proteins. Autophagy 2011; 7: 279–296.
Musgrove EA, Caldon CE, Barraclough J, Stone A, Sutherland RL . Cyclin D as a therapeutic target in cancer. Nat Rev Cancer 2011; 11: 558–572.
Wang ZV, Deng Y, Gao N, Pedrozo Z, Li DL, Morales CR et al. Spliced x-box binding protein 1 couples the unfolded protein response to hexosamine biosynthetic pathway. Cell 2014; 156: 1179–1192.
Ying H, Kimmelman AC, Lyssiotis CA, Hua S, Chu GC, Fletcher-Sananikone E et al. Oncogenic Kras maintains pancreatic tumors through regulation of anabolic glucose metabolism. Cell 2012; 149: 656–670.
Braga-Basaria M, Dobs AS, Muller DC, Carducci MA, John M, Egan J et al. Metabolic syndrome in men with prostate cancer undergoing long-term androgen-deprivation therapy. J Clin Oncol 2006; 24: 3979–3983.
Gunter JH, Sarkar PL, Lubik AA, Nelson CC . New players for advanced prostate cancer and the rationalisation of insulin-sensitising medication. Int J Cell Biol 2013; 2013: 834684.
Derweesh IH, Diblasio CJ, Kincade MC, Malcolm JB, Lamar KD, Patterson AL et al. Risk of new-onset diabetes mellitus and worsening glycaemic variables for established diabetes in men undergoing androgen-deprivation therapy for prostate cancer. BJU Int 2007; 100: 1060–1065.
Acknowledgements
HMI is funded by an Early Stage Researcher fellowship as part of the EU FP7 Marie Curie Integrated Training Network, PRO-NEST (Prostate Research Organizations – Network Early Stage Training) Norwegian Cancer Society and Finnish Cultural Foundation. IGM is supported by funding from the Norwegian Research Council, Helse and the University of Oslo through the Centre for Molecular Medicine (Norway), which is a part of the Nordic EMBL (European Molecular Biology Laboratory) partnership. IGM is also supported by the Norwegian Cancer Society and by EU FP7 funding. IGM holds a visiting scientist position with Cancer Research UK through the Cambridge Research Institute and a Senior Visiting Research Fellowship with Cambridge University through the Department of Oncology. We thank Professor Olli Kallioniemi’s group (Institute for Molecular Medicine Finland, Helsinki, Finland) for technical support.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on the Oncogene website
Supplementary information
Rights and permissions
About this article
Cite this article
Itkonen, H., Engedal, N., Babaie, E. et al. UAP1 is overexpressed in prostate cancer and is protective against inhibitors of N-linked glycosylation. Oncogene 34, 3744–3750 (2015). https://doi.org/10.1038/onc.2014.307
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2014.307
This article is cited by
-
O-GlcNAc transferase couples MRE11 to transcriptionally active chromatin to suppress DNA damage
Journal of Biomedical Science (2022)
-
O-GlcNAcylation links oncogenic signals and cancer epigenetics
Discover Oncology (2021)
-
Fbxl17 is rearranged in breast cancer and loss of its activity leads to increased global O-GlcNAcylation
Cellular and Molecular Life Sciences (2020)
-
Fueling the fire: emerging role of the hexosamine biosynthetic pathway in cancer
BMC Biology (2019)
-
EZH2 is overexpressed in transitional preplasmablasts and is involved in human plasma cell differentiation
Leukemia (2019)