Abstract
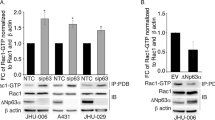
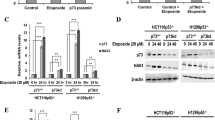
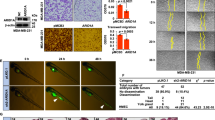
The DEAD-box RNA helicase DDX3 is a versatile protein involved in multiple steps of gene expression and various cellular signaling pathways. DDX3 mutations have been implicated in the wingless (Wnt) type of medulloblastoma. We show here that small interfering RNA-mediated DDX3 knockdown in various cell lines increased cell–cell adhesion but decreased cell–extracellular matrix adhesion. Moreover, DDX3 depletion suppressed cell motility and impaired directional migration in the wound-healing assay. Accordingly, DDX3-depleted cells exhibited reduced invasive capacities in vitro as well as reduced metastatic potential in mice. We also examined the mechanism underlying DDX3-regulated cell migration. DDX3 knockdown reduced the levels of both Rac1 and β-catenin proteins, and consequentially downregulated the expression of several β-catenin target genes. Moreover, we demonstrated that DDX3-regulated Rac1 mRNA translation, possibly through an interaction with its 5′-untranslated region, and affected β-catenin protein stability in an Rac1-dependent manner. Taken together, our results indicate the DDX3–Rac1–β-catenin regulatory axis in modulating the expression of Wnt/β-catenin target genes. Therefore, this report provides a mechanistic context for the role of DDX3 in Wnt-type tumors.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Tarn WY, Chang TH . The current understanding of Ded1p/DDX3 homologs from yeast to human. RNA Biol 2009; 6: 17–20.
Schroder M, Baran M, Bowie AG . Viral targeting of DEAD box protein 3 reveals its role in TBK1/IKKepsilon-mediated IRF activation. EMBO J 2008; 27: 2147–2157.
Soulat D, Burckstummer T, Westermayer S, Goncalves A, Bauch A, Stefanovic A et al. The DEAD-box helicase DDX3X is a critical component of the TANK-binding kinase 1-dependent innate immune response. EMBO J 2008; 27: 2135–2146.
Cruciat CM, Dolde C, de Groot RE, Ohkawara B, Reinhard C, Korswagen HC et al. RNA helicase DDX3 is a regulatory subunit of casein kinase 1 in Wnt-beta-catenin signaling. Science 2013; 339: 1436–1441.
Chuang RY, Weaver PL, Liu Z, Chang TH . Requirement of the DEAD-Box protein ded1p for messenger RNA translation. Science 1997; 275: 1468–1471.
Lai MC, Lee YH, Tarn WY . The DEAD-box RNA helicase DDX3 associates with export messenger ribonucleoproteins as well as tip-associated protein and participates in translational control. Mol Biol Cell 2008; 19: 3847–3858.
Lai MC, Chang WC, Shieh SY, Tarn WY . DDX3 regulates cell growth through translational control of cyclin E1. Mol Cell Biol 2010; 30: 5444–5453.
Soto-Rifo R, Rubilar PS, Limousin T, de Breyne S, Decimo D, Ohlmann T . DEAD-box protein DDX3 associates with eIF4F to promote translation of selected mRNAs. EMBO J 2012; 31: 3745–3756.
Yang Q, Jankowsky E . ATP- and ADP-dependent modulation of RNA unwinding and strand annealing activities by the DEAD-box protein DED1. Biochemistry 2005; 44: 13591–13601.
Marsden S, Nardelli M, Linder P, McCarthy JE . Unwinding single RNA molecules using helicases involved in eukaryotic translation initiation. J Mol Biol 2006; 361: 327–335.
Grallert B, Kearsey SE, Lenhard M, Carlson CR, Nurse P, Boye E et al. A fission yeast general translation factor reveals links between protein synthesis and cell cycle controls. J Cell Sci 2000; 113((Part 8)): 1447–1458.
Fukumura J, Noguchi E, Sekiguchi T, Nishimoto T . A temperature-sensitive mutant of the mammalian RNA helicase, DEAD-BOX X isoform, DBX, defective in the transition from G1 to S phase. J Biochem 2003; 134: 71–82.
Miao X, Yang ZL, Xiong L, Zou Q, Yuan Y, Li J et al. Nectin-2 and DDX3 are biomarkers for metastasis and poor prognosis of squamous cell/adenosquamous carcinomas and adenocarcinoma of gallbladder. Int J Clin Exp Pathol 2013; 6: 179–190.
Botlagunta M, Vesuna F, Mironchik Y, Raman A, Lisok A, Winnard P Jr et al. Oncogenic role of DDX3 in breast cancer biogenesis. Oncogene 2008; 27: 3912–3922.
Huang JS, Chao CC, Su TL, Yeh SH, Chen DS, Chen CT et al. Diverse cellular transformation capability of overexpressed genes in human hepatocellular carcinoma. Biochem Biophys Res Commun 2004; 315: 950–958.
Jones DT, Jager N, Kool M, Zichner T, Hutter B, Sultan M et al. Dissecting the genomic complexity underlying medulloblastoma. Nature 2012; 488: 100–105.
Pugh TJ, Weeraratne SD, Archer TC, Pomeranz Krummel DA, Auclair D, Bochicchio J et al. Medulloblastoma exome sequencing uncovers subtype-specific somatic mutations. Nature 2012; 488: 106–110.
Robinson G, Parker M, Kranenburg TA, Lu C, Chen X, Ding L et al. Novel mutations target distinct subgroups of medulloblastoma. Nature 2012; 488: 43–48.
Koshio J, Kagamu H, Nozaki K, Saida Y, Tanaka T, Shoji S et al. DEAD/H (Asp-Glu-Ala-Asp/His) box polypeptide 3, X-linked is an immunogenic target of cancer stem cells. Cancer Immunol Immunother 2013; 62: 1619–1628.
Sun M, Song L, Zhou T, Gillespie GY, Jope RS . The role of DDX3 in regulating Snail. Biochim Biophys Acta 2011; 1813: 438–447.
Wu DW, Lee MC, Wang J, Chen CY, Cheng YW, Lee H . DDX3 loss by p53 inactivation promotes tumor malignancy via the MDM2/Slug/E-cadherin pathway and poor patient outcome in non-small-cell lung cancer. Oncogene 2013; 33: 1515–1526.
Parri M, Chiarugi P . Rac and Rho GTPases in cancer cell motility control. Cell Commun Signal 2010; 8: 23.
Li J, Luo R, Kowluru A, Li G . Novel regulation by Rac1 of glucose- and forskolin-induced insulin secretion in INS-1 beta-cells. Am J Physiol Endocrinol Metab 2004; 286: E818–E827.
Chang F, Lemmon C, Lietha D, Eck M, Romer L . Tyrosine phosphorylation of Rac1: a role in regulation of cell spreading. PLoS ONE 2011; 6: e28587.
Cheon SS, Wei Q, Gurung A, Youn A, Bright T, Poon R et al. Beta-catenin regulates wound size and mediates the effect of TGF-beta in cutaneous healing. FASEB J 2006; 20: 692–701.
Hao J, Li TG, Qi X, Zhao DF, Zhao GQ . WNT/beta-catenin pathway up-regulates Stat3 and converges on LIF to prevent differentiation of mouse embryonic stem cells. Dev Biol 2006; 290: 81–91.
Kang DW, Lee SH, Yoon JW, Park WS, Choi KY, Min do S . Phospholipase D1 drives a positive feedback loop to reinforce the Wnt/beta-catenin/TCF signaling axis. Cancer Res 2010; 70: 4233–4242.
Lee CH, Hung HW, Hung PH, Shieh YS . Epidermal growth factor receptor regulates beta-catenin location, stability, and transcriptional activity in oral cancer. Mol Cancer 2010; 9: 64.
Zhu G, Wang Y, Huang B, Liang J, Ding Y, Xu A et al. A Rac1/PAK1 cascade controls beta-catenin activation in colon cancer cells. Oncogene 2012; 31: 1001–1012.
Soto-Rifo R, Rubilar PS, Ohlmann T . The DEAD-box helicase DDX3 substitutes for the cap-binding protein eIF4E to promote compartmentalized translation initiation of the HIV-1 genomic RNA. Nucleic Acids Res 2013; 41: 6286–6299.
Yedavalli VS, Neuveut C, Chi YH, Kleiman L, Jeang KT . Requirement of DDX3 DEAD box RNA helicase for HIV-1 Rev-RRE export function. Cell 2004; 119: 381–392.
Chen B, Gao Y, Jiang T, Ding J, Zeng Y, Xu R et al. Inhibition of tumor cell migration and invasion through knockdown of Rac1 expression in medulloblastoma cells. Cell Mol Neurobiol 2011; 31: 251–257.
Vidali L, Chen F, Cicchetti G, Ohta Y, Kwiatkowski DJ . Rac1-null mouse embryonic fibroblasts are motile and respond to platelet-derived growth factor. Mol Biol Cell 2006; 17: 2377–2390.
Akhtar N, Hotchin NA . RAC1 regulates adherens junctions through endocytosis of E-cadherin. Mol Biol Cell 2001; 12: 847–862.
Muller T, Bain G, Wang X, Papkoff J . Regulation of epithelial cell migration and tumor formation by beta-catenin signaling. Exp Cell Res 2002; 280: 119–133.
Schlessinger K, Hall A, Tolwinski N . Wnt signaling pathways meet Rho GTPases. Genes Dev 2009; 23: 265–277.
Zavarella S, Nakada M, Belverud S, Coniglio SJ, Chan A, Mittler MA et al. Role of Rac1-regulated signaling in medulloblastoma invasion. Laboratory investigation. J Neurosurg Pediatr 2009; 4: 97–104.
Acknowledgements
We thank Dr Yi-Shing Shieh for the TOPflash and FOPflash vectors, and Cho-Ying Chiang for technical assistance. This work was supported by a grant (NHRI-EX101-10046NI to WYT) from the National Health Research Institutes of Taiwan.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on the Oncogene website
Rights and permissions
About this article
Cite this article
Chen, HH., Yu, HI., Cho, WC. et al. DDX3 modulates cell adhesion and motility and cancer cell metastasis via Rac1-mediated signaling pathway. Oncogene 34, 2790–2800 (2015). https://doi.org/10.1038/onc.2014.190
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2014.190
This article is cited by
-
Disease-related protein co-expression networks are associated with the prognosis of resectable node-positive pancreatic ductal adenocarcinoma
Scientific Reports (2022)
-
Inhibition of DDX3 and COX-2 by forskolin and evaluation of anti-proliferative, pro-apoptotic effects on cervical cancer cells: molecular modelling and in vitro approaches
Medical Oncology (2022)
-
DDX3X: structure, physiologic functions and cancer
Molecular Cancer (2021)
-
The DEAD-box protein family of RNA helicases: sentinels for a myriad of cellular functions with emerging roles in tumorigenesis
International Journal of Clinical Oncology (2021)
-
Cytoplasmic DDX3 as prognosticator in male breast cancer
Virchows Archiv (2021)