Abstract
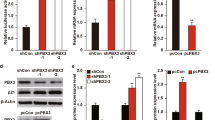
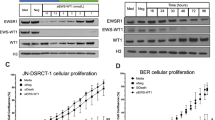
Aberrant nuclear factor (NF)-κB activation is frequently observed in human cancers. Genome characterization efforts have identified genetic alterations in multiple components of the NF-κB pathway, some of which have been shown to be essential for cancer initiation and tumor maintenance. Here, using patient tumors and cancer cell lines, we identify the NF-κB regulator, TRAF2 (tumor necrosis factor (TNF) receptor-associated factor 2), as an oncogene that is recurrently amplified and rearranged in 15% of human epithelial cancers. Suppression of TRAF2 in cancer cells harboring TRAF2 copy number gain inhibits proliferation, NF-κB activation, anchorage-independent growth and tumorigenesis. Cancer cells that are dependent on TRAF2 also require NF-κB for survival. The phosphorylation of TRAF2 at serine 11 is essential for the survival of cancer cells harboring TRAF2 amplification. Together, these observations identify TRAF2 as a frequently amplified oncogene.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Baud V, Karin M . Is NF-kappaB a good target for cancer therapy? Hopes and pitfalls. Nat Rev Drug Discov 2009; 8: 33–40.
Greten FR, Eckmann L, Greten TF, Park JM, Li ZW, Egan LJ et al. IKKbeta links inflammation and tumorigenesis in a mouse model of colitis-associated cancer. Cell 2004; 118: 285–296.
Karin M . Nuclear factor-kappaB in cancer development and progression. Nature 2006; 441: 431–436.
Karin M, Greten FR . NF-kappaB: linking inflammation and immunity to cancer development and progression. Nat Rev Immunol 2005; 5: 749–759.
Pikarsky E, Porat RM, Stein I, Abramovitch R, Amit S, Kasem S et al. NF-kappaB functions as a tumour promoter in inflammation-associated cancer. Nature 2004; 431: 461–466.
Aggarwal BB, Sung B . NF-kappaB in cancer: a matter of life and death. Cancer Discov 2011; 1: 469–471.
Schwitalla S, Fingerle AA, Cammareri P, Nebelsiek T, Goktuna SI, Ziegler PK et al. Intestinal tumorigenesis initiated by dedifferentiation and acquisition of stem-cell-like properties. Cell 2013; 152: 25–38.
Compagno M, Lim WK, Grunn A, Nandula SV, Brahmachary M, Shen Q et al. Mutations of multiple genes cause deregulation of NF-kappaB in diffuse large B-cell lymphoma. Nature 2009; 459: 717–721.
Kovalenko A, Chable-Bessia C, Cantarella G, Israel A, Wallach D, Courtois G . The tumour suppressor CYLD negatively regulates NF-kappaB signalling by deubiquitination. Nature 2003; 424: 801–805.
Lenz G, Davis RE, Ngo VN, Lam L, George TC, Wright GW et al. Oncogenic CARD11 mutations in human diffuse large B cell lymphoma. Science 2008; 319: 1676–1679.
Novak U, Rinaldi A, Kwee I, Nandula SV, Rancoita PM, Compagno M et al. The NF-{kappa}B negative regulator TNFAIP3 (A20) is inactivated by somatic mutations and genomic deletions in marginal zone lymphomas. Blood 2009; 113: 4918–4921.
Brummelkamp TR, Nijman SM, Dirac AM, Bernards R . Loss of the cylindromatosis tumour suppressor inhibits apoptosis by activating NF-kappaB. Nature 2003; 424: 797–801.
Annunziata CM, Davis RE, Demchenko Y, Bellamy W, Gabrea A, Zhan F et al. Frequent engagement of the classical and alternative NF-kappaB pathways by diverse genetic abnormalities in multiple myeloma. Cancer Cell 2007; 12: 115–130.
Keats JJ, Fonseca R, Chesi M, Schop R, Baker A, Chng WJ et al. Promiscuous mutations activate the noncanonical NF-kappaB pathway in multiple myeloma. Cancer Cell 2007; 12: 131–144.
Kan Z, Jaiswal BS, Stinson J, Janakiraman V, Bhatt D, Stern HM et al. Diverse somatic mutation patterns and pathway alterations in human cancers. Nature 2010; 466: 869–873.
Stratton MR, Campbell PJ, Futreal PA . The cancer genome. Nature 2009; 458: 719–724.
Pflueger D, Terry S, Sboner A, Habegger L, Esgueva R, Lin PC et al. Discovery of non-ETS gene fusions in human prostate cancer using next-generation RNA sequencing. Genome Res 2011; 21: 56–67.
Boehm JS, Zhao JJ, Yao J, Kim SY, Firestein R, Dunn IF et al. Integrative genomic approaches identify IKBKE as a breast cancer oncogene. Cell 2007; 129: 1065–1079.
Meylan E, Dooley AL, Feldser DM, Shen L, Turk E, Ouyang C et al. Requirement for NF-kappaB signalling in a mouse model of lung adenocarcinoma. Nature 2009; 462: 104–107.
Basseres DS, Ebbs A, Levantini E, Baldwin AS . Requirement of the NF-kappaB subunit p65/RelA for K-Ras-induced lung tumorigenesis. Cancer Res 2010; 70: 3537–3546.
Barbie DA, Tamayo P, Boehm JS, Kim SY, Moody SE, Dunn IF et al. Systematic RNA interference reveals that oncogenic KRAS-driven cancers require TBK1. Nature 2009; 462: 108–112.
Ling J, Kang Y, Zhao R, Xia Q, Lee DF, Chang Z et al. KrasG12D-induced IKK2/beta/NF-kappaB activation by IL-1alpha and p62 feedforward loops is required for development of pancreatic ductal adenocarcinoma. Cancer Cell 2012; 21: 105–120.
Starczynowski DT, Lockwood WW, Delehouzee S, Chari R, Wegrzyn J, Fuller M et al. TRAF6 is an amplified oncogene bridging the RAS and NF-kappaB pathways in human lung cancer. J Clin Invest 2011; 121: 4095–4105.
Min J, Zaslavsky A, Fedele G, McLaughlin SK, Reczek EE, De Raedt T et al. An oncogene-tumor suppressor cascade drives metastatic prostate cancer by coordinately activating Ras and nuclear factor-kappaB. Nat Med 2010; 16: 286–294.
Au PY, Yeh WC . Physiological roles and mechanisms of signaling by TRAF2 and TRAF5. Adv Exp Med Biol 2007; 597: 32–47.
Pomerantz JL, Baltimore D . NF-kappaB activation by a signaling complex containing TRAF2, TANK and TBK1, a novel IKK-related kinase. EMBO J 1999; 18: 6694–6704.
Chen ZJ . Ubiquitin signalling in the NF-kappaB pathway. Nat Cell Biol 2005; 7: 758–765.
Thomas GS, Zhang L, Blackwell K, Habelhah H . Phosphorylation of TRAF2 within its RING domain inhibits stress-induced cell death by promoting IKK and suppressing JNK activation. Cancer Res 2009; 69: 3665–3672.
Wood KC, Konieczkowski DJ, Johannessen CM, Boehm JS, Tamayo P, Botvinnik OB et al. MicroSCALE screening reveals genetic modifiers of therapeutic response in melanoma. Sci Signal 2012; 5: rs4.
Bivona TG, Hieronymus H, Parker J, Chang K, Taron M, Rosell R et al. FAS and NF-kappaB signalling modulate dependence of lung cancers on mutant EGFR. Nature 2011; 471: 523–526.
Hutti JE, Shen RR, Abbott DW, Zhou AY, Sprott KM, Asara JM et al. Phosphorylation of the tumor suppressor CYLD by the breast cancer oncogene IKKepsilon promotes cell transformation. Mol Cell 2009; 34: 461–472.
Beroukhim R, Mermel CH, Porter D, Wei G, Raychaudhuri S, Donovan J et al. The landscape of somatic copy-number alteration across human cancers. Nature 2010; 463: 899–905.
Cancer Genome Atlas Network. Comprehensive molecular portraits of human breast tumours. Nature 2012; 490: 61–70.
Cancer Genome Atlas Research Network. Integrated genomic analyses of ovarian carcinoma. Nature 2011; 474: 609–615.
Cancer Genome Atlas Network. Comprehensive molecular characterization of human colon and rectal cancer. Nature 2012; 487: 330–337.
Blackwell K, Zhang L, Thomas GS, Sun S, Nakano H, Habelhah H . TRAF2 phosphorylation modulates tumor necrosis factor alpha-induced gene expression and cell resistance to apoptosis. Mol Cell Biol 2009; 29: 303–314.
Karin M, Gallagher E . TNFR signaling: ubiquitin-conjugated TRAFfic signals control stop-and-go for MAPK signaling complexes. Immunol Rev 2009; 228: 225–240.
Li S, Wang L, Dorf ME . PKC phosphorylation of TRAF2 mediates IKKalpha/beta recruitment and K63-linked polyubiquitination. Mol Cell 2009; 33: 30–42.
Staudt LM . Oncogenic activation of NF-kappaB. Cold Spring Harb Perspect Biol 2010; 2: a000109.
Zhou AY, Shen RR, Kim E, Lock YJ, Xu M, Chen ZJ et al. IKKepsilon-mediated tumorigenesis requires K63-linked polyubiquitination by a cIAP1/cIAP2/TRAF2 E3 ubiquitin ligase complex. Cell Rep 2013; 3: 724–733.
Bassing CH, Suh H, Ferguson DO, Chua KF, Manis J, Eckersdorff M et al. Histone H2AX: a dosage-dependent suppressor of oncogenic translocations and tumors. Cell 2003; 114: 359–370.
Fuerer C, Nusse R . Lentiviral vectors to probe and manipulate the Wnt signaling pathway. PloS One 2010; 5: e9370.
Barretina J, Caponigro G, Stransky N, Venkatesan K, Margolin AA, Kim S et al. The Cancer Cell Line Encyclopedia enables predictive modelling of anticancer drug sensitivity. Nature 2012; 483: 603–607.
Acknowledgements
We thank members of the Hahn lab and the Cichowski lab for thoughtful discussion, reagents and technical assistance. We also thank Hasem Habelhah for providing the pTRAF2 (Ser11) antibody. We further thank Shumei Wang, Anita Hawkins, Chengzeng Zhang and Cynthia Morton at the DFCI Cytogenetics Core for TRAF2 FISH analysis and discussion. This work was supported in part by R01 CA130988 (to WCH), a Ruth L. Kirschstein National Research Service Award F32 CA128265 (to RRS) and The Aid for Cancer Research (to RRS).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
RB and WCH are consultants for Novartis. The remaining authors declare no conflicts of interest.
Additional information
Supplementary Information accompanies this paper on the Oncogene website
Rights and permissions
About this article
Cite this article
Shen, R., Zhou, A., Kim, E. et al. TRAF2 is an NF-κB-activating oncogene in epithelial cancers. Oncogene 34, 209–216 (2015). https://doi.org/10.1038/onc.2013.543
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2013.543
Keywords
This article is cited by
-
The TRAF2-p62 axis promotes proliferation and survival of liver cancer by activating mTORC1 pathway
Cell Death & Differentiation (2023)
-
Molecular insight into isoform specific inhibition of PI3K-α and PKC-η with dietary agents through an ensemble pharmacophore and docking studies
Scientific Reports (2021)
-
The ubiquitin ligase CHIP modulates cellular behaviors of gastric cancer cells by regulating TRAF2
Cancer Cell International (2019)
-
TRAF2 in osteotropic breast cancer cells enhances skeletal tumour growth and promotes osteolysis
Scientific Reports (2018)
-
Downregulation of miR-141-3p promotes bone metastasis via activating NF-κB signaling in prostate cancer
Journal of Experimental & Clinical Cancer Research (2017)