Abstract
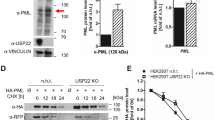
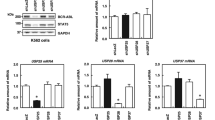
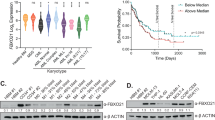
Acute promyelocytic leukemia (APL) is predominantly characterized by chromosomal translocations between the retinoic acid receptor, alpha (RARA) gene and the promyelocytic leukemia (PML) or promyelocytic leukemia zinc finger (PLZF) gene. In APL cells with PML/RARA fusions, arsenic trioxide and all-trans retinoic acid treatments specifically target the fusion protein for proteasome-dependent degradation, thereby promoting cellular differentiation and clinical remission of disease. In contrast, APL cells expressing PLZF/RARA fusion proteins are largely resistant to similar treatments and prognosis for patients with this translocation is poor. Understanding the molecular mechanisms regulating PLZF/RARA protein stability would provide novel therapeutic targets for PLZF/RARA-associated APL. Toward this end, we have performed an RNAi-based screen to identify factors affecting PLZF/RARA stability. Among the factors identified was the ubiquitin-specific peptidase 37 (USP37). We showed that USP37 interacted with PLZF/RARA through the PLZF moiety and sustained PLZF/RARA steady state levels. Domain mapping study revealed that N-terminal domain of USP37 is required for the PLZF/RARA interaction and protein regulation. Furthermore, overexpression or depletion of USP37 caused an increase or decrease of PLZF/RARA protein half-life, correlating with down- or upregulation of PLZF/RARA poly-ubiquitination, respectively. By PLZF/RARA-transduced primary mouse hematopoietic progenitor cells, we demonstrated that Usp37 knockdown alleviated PLZF/RARA-mediated target gene suppression and cell transformation potential. Altogether, our findings of USP37-modulating PLZF/RARA stability and cell transformation suggest that USP37 is a potential therapeutic target for PLZF/RARA-associated APL.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Mistry AR, Pedersen EW, Solomon E, Grimwade D . The molecular pathogenesis of acute promyelocytic leukaemia: implications for the clinical management of the disease. Blood Rev 2003; 17: 71–97.
Melnick A, Licht JD . Deconstructing a disease: RARalpha, its fusion partners, and their roles in the pathogenesis of acute promyelocytic leukemia. Blood 1999; 93: 3167–3215.
Guibal FC, Alberich-Jorda M, Hirai H, Ebralidze A, Levantini E, Di Ruscio A et al. Identification of a myeloid committed progenitor as the cancer-initiating cell in acute promyelocytic leukemia. Blood 2009; 114: 5415–5425.
Nasr R, Guillemin MC, Ferhi O, Soilihi H, Peres L, Berthier C et al. Eradication of acute promyelocytic leukemia-initiating cells through PML-RARA degradation. Nat Med 2008; 14: 1333–1342.
Nasr R, Lallemand-Breitenbach V, Zhu J, Guillemin MC, de The H . Therapy-induced PML/RARA proteolysis and acute promyelocytic leukemia cure. Clin Cancer Res 2009; 15: 6321–6326.
Wang ZY, Chen Z . Acute promyelocytic leukemia: from highly fatal to highly curable. Blood 2008; 111: 2505–2515.
Lallemand-Breitenbach V, Jeanne M, Benhenda S, Nasr R, Lei M, Peres L et al. Arsenic degrades PML or PML-RARalpha through a SUMO-triggered RNF4/ubiquitin-mediated pathway. Nat Cell Biol 2008; 10: 547–555.
Zhang XW, Yan XJ, Zhou ZR, Yang FF, Wu ZY, Sun HB et al. Arsenic trioxide controls the fate of the PML-RARalpha oncoprotein by directly binding PML. Science 2010; 328: 240–243.
Glickman MH, Ciechanover A . The ubiquitin-proteasome proteolytic pathway: destruction for the sake of construction. Physiol Rev 2002; 82: 373–428.
Ventii KH, Wilkinson KD . Protein partners of deubiquitinating enzymes. Biochem J 2008; 414: 161–175.
Deshaies RJ, Joazeiro CA . RING domain E3 ubiquitin ligases. Annu Rev Biochem 2009; 78: 399–434.
Nijman SM, Luna-Vargas MP, Velds A, Brummelkamp TR, Dirac AM, Sixma TK et al. A genomic and functional inventory of deubiquitinating enzymes. Cell 2005; 123: 773–786.
Sowa ME, Bennett EJ, Gygi SP, Harper JW . Defining the human deubiquitinating enzyme interaction landscape. Cell 2009; 138: 389–403.
Lin RJ, Sternsdorf T, Tini M, Evans RM . Transcriptional regulation in acute promyelocytic leukemia. Oncogene 2001; 20: 7204–7215.
Duprez E, Wagner K, Koch H, Tenen DG . C/EBPbeta: a major PML-RARA-responsive gene in retinoic acid-induced differentiation of APL cells. EMBO J 2003; 22: 5806–5816.
Lavau C, Szilvassy SJ, Slany R, Cleary ML . Immortalization and leukemic transformation of a myelomonocytic precursor by retrovirally transduced HRX-ENL. EMBO J 1997; 16: 4226–4237.
Li M, Brooks CL, Kon N, Gu W . A dynamic role of HAUSP in the p53-Mdm2 pathway. Mol Cell 2004; 13: 879–886.
Schwickart M, Huang X, Lill JR, Liu J, Ferrando R, French DM et al. Deubiquitinase USP9X stabilizes MCL1 and promotes tumour cell survival. Nature 2010; 463: 103–107.
Popov N, Wanzel M, Madiredjo M, Zhang D, Beijersbergen R, Bernards R et al. The ubiquitin-specific protease USP28 is required for MYC stability. Nat Cell Biol 2007; 9: 765–774.
Colland F, Formstecher E, Jacq X, Reverdy C, Planquette C, Conrath S et al. Small-molecule inhibitor of USP7/HAUSP ubiquitin protease stabilizes and activates p53 in cells. Mol Cancer Ther 2009; 8: 2286–2295.
Lee BH, Lee MJ, Park S, Oh DC, Elsasser S, Chen PC et al. Enhancement of proteasome activity by a small-molecule inhibitor of USP14. Nature 2010; 467: 179–184.
Huang X, Summers MK, Pham V, Lill JR, Liu J, Lee G et al. Deubiquitinase USP37 is activated by CDK2 to antagonize APC(CDH1) and promote S phase entry. Mol Cell 2011; 42: 511–523.
Muller C, Yang R, Park DJ, Serve H, Berdel WE, Koeffler HP . The aberrant fusion proteins PML-RAR alpha and PLZF-RAR alpha contribute to the overexpression of cyclin A1 in acute promyelocytic leukemia. Blood 2000; 96: 3894–3899.
Yang R, Nakamaki T, Lubbert M, Said J, Sakashita A, Freyaldenhoven BS et al. Cyclin A1 expression in leukemia and normal hematopoietic cells. Blood 1999; 93: 2067–2074.
Ji P, Agrawal S, Diederichs S, Baumer N, Becker A, Cauvet T et al. Cyclin A1, the alternative A-type cyclin, contributes to G1/S cell cycle progression in somatic cells. Oncogene 2005; 24: 2739–2744.
Doulatov S, Notta F, Rice KL, Howell L, Zelent A, Licht JD et al. PLZF is a regulator of homeostatic and cytokine-induced myeloid development. Genes Dev 2009; 23: 2076–2087.
Chen Z, Brand NJ, Chen A, Chen SJ, Tong JH, Wang ZY et al. Fusion between a novel Kruppel-like zinc finger gene and the retinoic acid receptor-alpha locus due to a variant t(11;17) translocation associated with acute promyelocytic leukaemia. EMBO J 1993; 12: 1161–1167.
Shaknovich R, Yeyati PL, Ivins S, Melnick A, Lempert C, Waxman S et al. The promyelocytic leukemia zinc finger protein affects myeloid cell growth, differentiation, and apoptosis. Mol Cell Biol 1998; 18: 5533–5545.
Buaas FW, Kirsh AL, Sharma M, McLean DJ, Morris JL, Griswold MD et al. Plzf is required in adult male germ cells for stem cell self-renewal. Nat Genet 2004; 36: 647–652.
Costoya JA, Hobbs RM, Barna M, Cattoretti G, Manova K, Sukhwani M et al. Essential role of Plzf in maintenance of spermatogonial stem cells. Nat Genet 2004; 36: 653–659.
Chang CC, Naik MT, Huang YS, Jeng JC, Liao PH, Kuo HY et al. Structural and functional roles of Daxx SIM phosphorylation in SUMO paralog-selective binding and apoptosis modulation. Mol Cell 2011; 42: 62–74.
Lin DY, Huang YS, Jeng JC, Kuo HY, Chang CC, Chao TT et al. Role of SUMO-interacting motif in Daxx SUMO modification, subnuclear localization, and repression of sumoylated transcription factors. Mol Cell 2006; 24: 341–354.
Acknowledgements
We thank Dr. J Wade Harper for USP37, OTUD6B and USP2 cDNA constructs, Dr Sheng-Chung Lee for anti-ubiquitin antibody, and Dr Yen-Sung Huang for mouse preparation. We also thank for the NRPGM Core RNAi by providing lentiviral constructs for TetOn system and shRNAs of DUBs. We are grateful to Dr Michael J Matunis for comments on the paper. The work was supported by NSC Grants (NSC100-2321-B-001-004; NSC100-3112-B-001-004) and an Academia Sinica Investigator Award to H-M Shih.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on the Oncogene website
Supplementary information
Rights and permissions
About this article
Cite this article
Yang, WC., Shih, HM. The deubiquitinating enzyme USP37 regulates the oncogenic fusion protein PLZF/RARA stability. Oncogene 32, 5167–5175 (2013). https://doi.org/10.1038/onc.2012.537
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2012.537
Keywords
This article is cited by
-
Ubiquitin specific peptidase 37 and PCNA interaction promotes osteosarcoma pathogenesis by modulating replication fork progression
Journal of Translational Medicine (2023)
-
Deubiquitinases in hematological malignancies
Biomarker Research (2021)
-
Ubiquitin-specific peptidase 37: an important cog in the oncogenic machinery of cancerous cells
Journal of Experimental & Clinical Cancer Research (2021)
-
Current views on the genetic landscape and management of variant acute promyelocytic leukemia
Biomarker Research (2021)
-
Targeting USP47 overcomes tyrosine kinase inhibitor resistance and eradicates leukemia stem/progenitor cells in chronic myelogenous leukemia
Nature Communications (2021)