Abstract
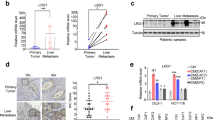
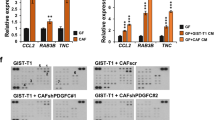
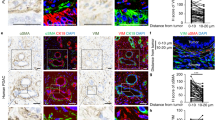
The interaction between epithelial cancer cells and cancer-associated fibroblasts (CAFs) has a major role in cancer progression and eventually in metastasis. In colorectal cancer (CRC), CAFs are present in high abundance, but their origin and functional interaction with epithelial tumor cells has not been elucidated. In this study we observed strong activation of the transforming growth factor-β (TGF-β)/Smad signaling pathway in CRC CAFs, accompanied by decreased signaling in epithelial tumor cells. We evaluated the TGF-β1 response and the expression of target genes including matrix metalloproteinases (MMPs) and plasminogen activator inhibitor (PAI)-1 of various epithelial CRC cell lines and primary CAFs in vitro. TGF-β1 stimulation caused high upregulation of MMPs, PAI-1 and TGF-β1 itself. Next we showed that incubation of CAFs with conditioned medium (CM) from epithelial cancer cells led to hyperactivation of the TGF-β signaling pathway, enhanced expression of target genes like PAI-1, and the expression of α-smooth muscle actin (α-SMA). We propose that the interaction of tumor cells with resident fibroblasts results in hyperactivated TGF-β1 signaling and subsequent transdifferentiation of the fibroblasts into α-SMA-positive CAFs. In turn this leads to cumulative production of TGF-β and proteinases within the tumor microenvironment, creating a cancer-promoting feedback loop.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
De Wever O, Mareel M . Role of tissue stroma in cancer cell invasion. J Pathol 2003; 200: 429–447.
Ruiter D, Bogenrieder T, Elder D, Herlyn M . Melanoma-stroma interactions: structural and functional aspects. Lancet Oncol 2002; 3: 35–43.
Micke P, Ostman A . Tumour-stroma interaction: cancer-associated fibroblasts as novel targets in anti-cancer therapy? Lung Cancer 2004; 45 (Suppl 2): S163–S175.
Park CC, Bissell MJ, Barcellos-Hoff MH . The influence of the microenvironment on the malignant phenotype. Mol Med Today 2000; 6: 324–329.
Powell DW, Adegboyega PA, Di Mari JF, Mifflin RC . Epithelial cells and their neighbors I. Role of intestinal myofibroblasts in development, repair, and cancer. Am J Physiol Gastrointest Liver Physiol 2005; 289: G2–G7.
Kalluri R, Zeisberg M . Fibroblasts in cancer. Nat Rev Cancer 2006; 6: 392–401.
Adegboyega PA, Mifflin RC, DiMari JF, Saada JI, Powell DW . Immunohistochemical study of myofibroblasts in normal colonic mucosa, hyperplastic polyps, and adenomatous colorectal polyps. Arch Pathol Lab Med 2002; 126: 829–836.
Lieubeau B, Heymann MF, Henry F, Barbieux I, Meflah K, Gregoire M . Immunomodulatory effects of tumor-associated fibroblasts in colorectal-tumor development. Int J Cancer 1999; 81: 629–636.
Desmouliere A, Geinoz A, Gabbiani F, Gabbiani G . Transforming growth factor-β1 induces alpha-smooth muscle actin expression in granulation tissue myofibroblasts and in quiescent and growing cultured fibroblasts. J Cell Biol 1993; 122: 103–111.
Blobe GC, Schiemann WP, Lodish HF . Role of transforming growth factor-β in human disease. N Engl J Med 2000; 342: 1350–1358.
Derynck R, Akhurst RJ, Balmain A . TGF-β signaling in tumor suppression and cancer progression. Nat Genet 2001; 29: 117–129.
Akhurst RJ, Derynck R . TGF-β signaling in cancer-a double-edged sword. Trends Cell Biol 2001; 11: S44–S51.
ten Dijke P, Arthur HM . Extracellular control of TGF-β signalling in vascular development and disease. Nat Rev Mol Cell Biol 2007; 8: 857–869.
Yoshinaga K, Obata H, Jurukovski V, Mazzieri R, Chen Y, Zilberberg L et al. Perturbation of transforming growth factor (TGF)-β1 association with latent TGF-β binding protein yields inflammation and tumors. Proc Natl Acad Sci USA 2008; 105: 18758–18763.
Lyons RM, Gentry LE, Purchio AF, Moses HL . Mechanism of activation of latent recombinant transforming growth factor β1 by plasmin. J Cell Biol 1990; 110: 1361–1367.
Jenkins G . The role of proteases in transforming growth factor-β activation. Int J Biochem Cell Biol 2008; 40: 1068–1078.
Annes JP, Munger JS, Rifkin DB . Making sense of latent TGF-β activation. J Cell Sci 2003; 116: 217–224.
Meulmeester E, ten Dijke P . The dynamic roles of TGF-β in cancer. J Pathol 2011; 223: 205–218.
Mesker WE, Junggeburt JM, Szuhai K, de Heer P, Morreau H, Tanke HJ et al. The carcinoma-stromal ratio of colon carcinoma is an independent factor for survival compared to lymph node status and tumor stage. Cell Oncol 2007; 29: 387–398.
Pourreyron C, Dumortier J, Ratineau C, Nejjari M, Beatrix O, Jacquier MF et al. Age-dependent variations of human and rat colon myofibroblasts in culture: Influence on their functional interactions with colon cancer cells. Int J Cancer 2003; 104: 28–35.
Huet E, Vallee B, Szul D, Verrecchia F, Mourah S, Jester JV et al. Extracellular matrix metalloproteinase inducer/CD147 promotes myofibroblast differentiation by inducing α-smooth muscle actin expression and collagen gel contraction: implications in tissue remodeling. FASEB J 2008; 22: 1144–1154.
Kankuri E, Cholujova D, Comajova M, Vaheri A, Bizik J . Induction of hepatocyte growth factor/scatter factor by fibroblast clustering directly promotes tumor cell invasiveness. Cancer Res 2005; 65: 9914–9922.
Kankuri E, Babusikova O, Hlubinova K, Salmenpera P, Boccaccio C, Lubitz W et al. Fibroblast nemosis arrests growth and induces differentiation of human leukemia cells. Int J Cancer 2008; 122: 1243–1252.
Elenbaas B, Weinberg RA . Heterotypic signaling between epithelial tumor cells and fibroblasts in carcinoma formation. Exp Cell Res 2001; 264: 169–184.
Cirri P, Chiarugi P . Cancer-associated-fibroblasts and tumour cells: a diabolic liaison driving cancer progression. Cancer Metastasis Rev 2011; 31: 195–208.
Ronnov-Jessen L, Petersen OW, Koteliansky VE, Bissell MJ . The origin of the myofibroblasts in breast cancer. recapitulation of tumor environment in culture unravels diversity and implicates converted fibroblasts and recruited smooth muscle cells. J Clin Invest 1995; 95: 859–873.
Lewis MP, Lygoe KA, Nystrom ML, Anderson WP, Speight PM, Marshall JF et al. Tumour-derived TGF-β1 modulates myofibroblast differentiation and promotes HGF/SF-dependent invasion of squamous carcinoma cells. Br J Cancer 2004; 90: 822–832.
Leivonen SK, Kahari VM . Transforming growth factor-β signaling in cancer invasion and metastasis. Int J Cancer 2007; 121: 2119–2124.
Bierie B, Moses HL . Tumour microenvironment: TGF-β: the molecular Jekyll and Hyde of cancer. Nat Rev Cancer 2006; 6: 506–520.
Munoz NM, Upton M, Rojas A, Washington MK, Lin L, Chytil A et al. Transforming growth factor β receptor type II inactivation induces the malignant transformation of intestinal neoplasms initiated by Apc mutation. Cancer Res 2006; 66: 9837–9844.
Woodford-Richens KL, Rowan AJ, Gorman P, Halford S, Bicknell DC, Wasan HS et al. SMAD4 mutations in colorectal cancer probably occur before chromosomal instability, but after divergence of the microsatellite instability pathway. Proc Natl Acad Sci USA 2001; 98: 9719–9723.
Hawinkels LJ, Verspaget HW, van Duijn W, van der Zon JM, Zuidwijk K, Kubben FJ et al. Tissue level, activation and cellular localisation of TGF-β1 and association with survival in gastric cancer patients. Br J Cancer 2007; 97: 398–404.
Barcellos-Hoff MH, Ewan KB . Transforming growth factor-β and breast cancer: Mammary gland development. Breast Cancer Res 2000; 2: 92–99.
Hawinkels L, Verspaget HW, van der Reijden JJ, Van der Zon J, Verheijen JH, Hommes D et al. Active TGF-β1 correlates with myofibroblasts and malignancy in the colorectal adenoma-carcinoma sequence. Cancer Sci 2009; 100: 663–670.
Shephard P, Martin G, Smola-Hess S, Brunner G, Krieg T, Smola H . Myofibroblast differentiation is induced in keratinocyte-fibroblast co-cultures and is antagonistically regulated by endogenous transforming growth factor-β and interleukin-1. Am J Pathol 2004; 164: 2055–2066.
Kunz-Schughart LA, Heyder P, Schroeder J, Knuechel R . A heterologous 3-D coculture model of breast tumor cells and fibroblasts to study tumor-associated fibroblast differentiation. Exp Cell Res 2001; 266: 74–86.
Ge G, Greenspan DS . BMP1 controls TGFβ1 activation via cleavage of latent TGFβ-binding protein. J Cell Biol 2006; 175: 111–120.
Wipff PJ, Hinz B . Integrins and the activation of latent transforming growth factor β1 - An intimate relationship. Eur J Cell Biol 2008; 87: 601–615.
Nishimura SL . Integrin-mediated transforming growth factor-β activation, a potential therapeutic target in fibrogenic disorders. Am J Pathol 2009; 175: 1362–1370.
Barcellos-Hoff MH, Derynck R, Tsang ML, Weatherbee JA . Transforming growth factor-β activation in irradiated murine mammary gland. J Clin Invest 1994; 93: 892–899.
Hawinkels LJ, ten Dijke P . Exploring anti-TGF-β therapies in cancer and fibrosis. Growth Factors 2011; 29: 140–152.
Hawinkels LJ, Zuidwijk K, Verspaget HW, Jonge-Muller ES, Duijn W, Ferreira V et al. VEGF release by MMP-9 mediated heparan sulphate cleavage induces colorectal cancer angiogenesis. Eur J Cancer 2008; 44: 1904–1913.
Dennler S, Itoh S, Vivien D, ten Dijke P, Huet S, Gauthier JM . Direct binding of Smad3 and Smad4 to critical TGF β-inducible elements in the promoter of human plasminogen activator inhibitor-type 1 gene. EMBO J 1998; 17: 3091–3100.
Dooley S, Hamzavi J, Breitkopf K, Wiercinska E, Said HM, Lorenzen J et al. Smad7 prevents activation of hepatic stellate cells and liver fibrosis in rats. Gastroenterology 2003; 125: 178–191.
Hawinkels LJ, Kuiper P, Wiercinska E, Verspaget HW, Liu Z, Pardali E et al. Matrix metalloproteinase-14 (MT1-MMP)-mediated endoglin shedding inhibits tumor angiogenesis. Cancer Res 2010; 70: 4141–4150.
Persson U, Izumi H, Souchelnytskyi S, Itoh S, Grimsby S, Engstrom U et al. The L45 loop in type I receptors for TGF-β family members is a critical determinant in specifying Smad isoform activation. FEBS Lett 1998; 434: 83–87.
Lindeman JH, Hanemaaijer R, Mulder A, Dijkstra PD, Szuhai K, Bromme D et al. Cathepsin K is the principal protease in giant cell tumor of bone. Am J Pathol 2004; 165: 593–600.
Sier CF, Hawinkels LJ, Zijlmans HJ, Zuidwijk K, de Jonge-Muller ES, Ferreira V et al. Endothelium specific matrilysin (MMP-7) expression in human cancers. Matrix Biol 2008; 27: 267–271.
Pardali E, van der Schaft DW, Wiercinska E, Gorter A, Hogendoorn PC, Griffioen AW et al. Critical role of endoglin in tumor cell plasticity of Ewing sarcoma and melanoma. Oncogene 2011; 30: 334–345.
Stolle K, Schnoor M, Fuellen G, Spitzer M, Engel T, Spener F et al. Cloning, cellular localization, genomic organization, and tissue-specific expression of the TGFβ1-inducible SMAP-5 gene. Gene 2005; 351: 119–130.
Hawinkels LJ, van Rossenberg SM, de Jonge-Muller ES, Molenaar TJ, Appeldoorn CC, van Berkel TJ et al. Efficient degradation-aided selection of protease inhibitors by phage display. Biochem Biophys Res Commun 2007; 364: 549–555.
Acknowledgements
We thank Dr R Hanemaaijer (TNO Quality of Life BioSciences, Leiden, The Netherlands) for helpful suggestions and reagents. Eveline de Jonge-Muller (Department of Gastroenterology-Hepatology, LUMC), Adri Mulder-Stapel (TNO) and Gabi van Pelt (Department Surgery, LUMC) are acknowledged for excellent technical support. This work was supported by the EC Tumor-host genomic project, the Centre for Biomedical Genetics, the Swedish Cancer Fonden 090773 (EW, PtD) and the Bas Mulder Award 2011 (LJACH, MP, UL2011-5051).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on the Oncogene website
Supplementary information
Rights and permissions
About this article
Cite this article
Hawinkels, L., Paauwe, M., Verspaget, H. et al. Interaction with colon cancer cells hyperactivates TGF-β signaling in cancer-associated fibroblasts. Oncogene 33, 97–107 (2014). https://doi.org/10.1038/onc.2012.536
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2012.536
Keywords
This article is cited by
-
Brevilin A is a potent anti-metastatic CRC agent that targets the VEGF-IL6-STAT3 axis in the HSCs-CRC interplay
Journal of Translational Medicine (2023)
-
Intimate communications within the tumor microenvironment: stromal factors function as an orchestra
Journal of Biomedical Science (2023)
-
Colitis-associated carcinogenesis: crosstalk between tumors, immune cells and gut microbiota
Cell & Bioscience (2023)
-
Circ_0020256 induces fibroblast activation to drive cholangiocarcinoma development via recruitment of EIF4A3 protein to stabilize KLF4 mRNA
Cell Death Discovery (2023)
-
A Novel TGF-β-Related Signature for Predicting Prognosis, Tumor Microenvironment, and Therapeutic Response in Colorectal Cancer
Biochemical Genetics (2023)