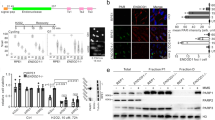
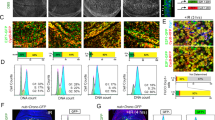
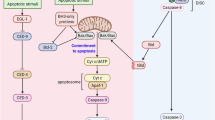
Abstract
We previously identified Caliban (Clbn) as the Drosophila homolog of human Serologically defined colon cancer antigen 1 gene and demonstrated that it could function as a tumor suppressor in human non-small-cell lung cancer (NSCLC) cells, although its mode of action was unknown. Herein, we identify roles for Clbn in DNA damage response. We generate clbn knockout flies using homologous recombination and demonstrate that they have a heightened sensitivity to irradiation. We show that normal Clbn function facilitates both p53-dependent and -independent DNA damage-induced apoptosis. Clbn coordinates different apoptosis pathways, showing a two-stage upregulation following DNA damage. Clbn has proapoptotic functions, working with both caspase and the proapoptotic gene Hid. Finally, ecotopic expression of clbn+ in NSCLC cells suppresses tumor formation in athymic nude mice. We conclude that Caliban is a regulator of DNA damage-induced apoptosis, functioning as a tumor suppressor in both p53-dependent and -independent pathways.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Chen Z, Trotman LC, Shaffer D, Lin HK, Dotan ZA, Niki M et al. Crucial role of p53-dependent cellular senescence in suppression of Pten-deficient tumorigenesis. Nature 2005; 436: 725–730.
Kemp CJ, Donehower LA, Bradley A, Balmain A . Reduction of p53 gene dosage does not increase initiation or promotion but enhances malignant progression of chemically induced skin tumors. Cell 1993; 74: 813–822.
Xue W, Zender L, Miething C, Dickins RA, Hernando E, Krizhanovsky V et al. Senescence and tumour clearance is triggered by p53 restoration in murine liver carcinomas. Nature 2007; 445: 656–660.
Ventura A, Kirsch DG, McLaughlin ME, Tuveson DA, Grimm J, Lintault L et al. Restoration of p53 function leads to tumour regression in vivo. Nature 2007; 445: 661–665.
Xie G, Habbersett RC, Jia Y, Peterson SR, Lehnert BE, Bradbury EM et al. Requirements for p53 and the ATM gene product in the regulation of G1/S and S phase checkpoints. Oncogene 1998; 16: 721–736.
Bode AM, Dong Z . Post-translational modification of p53 in tumorigenesis. Nat Rev Cancer 2004; 4: 793–805.
Bartek J, Lukas J . Mammalian G1- and S-phase checkpoints in response to DNA damage. Curr Opin Cell Biol 2001; 13: 738–747.
Attardi LD . The role of p53-mediated apoptosis as a crucial anti-tumor response to genomic instability: lessons from mouse models. Mutat Res 2005; 569: 145–157.
Donehower LA, Harvey M, Slagle BL, McArthur MJ, Montgomery CA, Butel JS et al. Mice deficient for p53 are developmentally normal but susceptible to spontaneous tumours. Nature 1992; 356: 215–221.
Ollmann M, Young LM, Di Como CJ, Karim F, Belvin M, Robertson S et al. Drosophila p53 is a structural and functional homolog of the tumor suppressor p53. Cell 2000; 101: 91–101.
Brodsky MH, Nordstrom W, Tsang G, Kwan E, Rubin GM, Abrams JM . Drosophila p53 binds a damage response element at the reaper locus. Cell 2000; 101: 103–113.
Sogame N, Kim M, Abrams JM . Drosophila p53 preserves genomic stability by regulating cell death. Proc Natl Acad Sci USA 2003; 100: 4696–4701.
Matsuoka S, Ballif BA, Smogorzewska A, McDonald ER, Hurov KE, Luo J et al. ATM and ATR substrate analysis reveals extensive protein networks responsive to DNA damage. Science 2007; 316: 1160–1166.
Sabourin M, Zakian VA . ATM-like kinases and regulation of telomerase: lessons from yeast and mammals. Trends Cell Biol 2008; 18: 337–346.
McKinnon PJ . ATM and ataxia telangiectasia. EMBO Rep 2004; 5: 772–776.
Song YH, Mirey G, Betson M, Haber DA, Settleman J . The Drosophila ATM ortholog, dATM, mediates the response to ionizing radiation and to spontaneous DNA damage during development. Curr Biol 2004; 14: 1354–1359.
Bi X, Wei SC, Rong YS . Telomere protection without a telomerase; the role of ATM and Mre11 in Drosophila telomere maintenance. Curr Biol 2004; 14: 1348–1353.
Silva E, Tiong S, Pedersen M, Homola E, Royou A, Fasulo B et al. ATM is required for telomere maintenance and chromosome stability during Drosophila development. Curr Biol 2004; 14: 1341–1347.
Oikemus SR, McGinnis N, Queiroz-Machado J, Tukachinsky H, Takada S, Sunkel CE et al. Drosophila atm/telomere fusion is required for telomeric localization of HP1 and telomere position effect. Genes Dev 2004; 18: 1850–1861.
Bi X, Jones T, Abbasi F, Lee H, Stultz B, Hursh DA et al. Drosophila caliban, a nuclear export mediator, can function as a tumor suppressor in human lung cancer cells. Oncogene 2005; 24: 8229–8239.
Scanlan MJ, Chen YT, Williamson B, Gure AO, Stockert E, Gordan JD et al. Characterization of human colon cancer antigens recognized by autologous antibodies. Int J Cancer 1998; 76: 652–658.
Carbonnelle D, Jacquot C, Lanco X, Le Dez G, Tomasoni C, Briand G et al. Up-regulation of a novel mRNA (NY-CO-1) involved in the methyl 4-methoxy-3-(3-methyl-2-butenoyl) benzoate (VT1)-induced proliferation arrest of a non-small-cell lung carcinoma cell line (NSCLC-N6). Int J Cancer 2001; 92: 388–397.
Chalastanis A, Penard-Lacronique V, Svrcek M, Defaweux V, Antoine N, Buhard O et al. Azathioprine-induced carcinogenesis in mice according to Msh2 genotype. J Natl Cancer Inst 2010; 102: 1731–1740.
Akdemir F, Christich A, Sogame N, Chapo J, Abrams JM . p53 directs focused genomic responses in Drosophila. Oncogene 2007; 26: 5184–5193.
Gong WJ, Golic KG . Ends-out or replacement, gene targeting in Drosophila. Proc Natl Acad Sci USA 2003; 100: 2556–2561.
Wichmann A, Jaklevic B, Su TT . Ionizing radiation induces caspase-dependent but Chk2- and p53-independent cell death in Drosophila melanogaster. Proc Natl Acad Sci USA 2006; 103: 9952–9957.
Lee JH, Lee E, Park J, Kim E, Kim J, Chung J . In vivo p53 function is indispensable for DNA damage-induced apoptotic signaling in Drosophila. FEBS Lett 2003; 550: 5–10.
Kornbluth S, White K . Apoptosis in Drosophila: neither fish nor fowl (nor man, nor worm). J Cell Sci 2005; 118 (Part 9): 1779–1787.
Steller H . Regulation of apoptosis in Drosophila. Cell Death Differ 2008; 15: 1132–1138.
McNamee LM, Brodsky MH . p53-Independent apoptosis limits DNA damage-induced aneuploidy. Genetics 2009; 182: 423–435.
Moon NS, Di Stefano L, Morris EJ, Patel R, White K, Dyson NJ . E2F and p53 induce apoptosis independently during Drosophila development but intersect in the context of DNA damage. PLoS Genet 2008; 4: e1000153.
Fan Y, Lee TV, Xu D, Chen Z, Lamblin AF, Steller H et al. Dual roles of Drosophila p53 in cell death and cell differentiation. Cell Death Differ 2010; 17: 912–921.
Duffy JB . GAL4 system in Drosophila: a fly geneticist’s Swiss army knife. Genesis 2002; 34: 1–15.
Baum JS, Arama E, Steller H, McCall K . The Drosophila caspases Strica and Dronc function redundantly in programmed cell death during oogenesis. Cell Death Differ 2007; 14: 1508–1517.
Xue D, Horvitz HR . Inhibition of the Caenorhabditis elegans cell-death protease CED-3 by a CED-3 cleavage site in baculovirus p35 protein. Nature 1995; 377: 248–251.
Lannan E, Vandergaast R, Friesen PD . Baculovirus caspase inhibitors P49 and P35 block virus-induced apoptosis downstream of effector caspase DrICE activation in Drosophila melanogaster cells. J Virol 2007; 81: 9319–9330.
Titen SW, Golic KG . Telomere loss provokes multiple pathways to apoptosis and produces genomic instability in Drosophila melanogaster. Genetics 2008; 180: 1821–1832.
Sherr CJ . Principles of tumor suppression. Cell 2004; 116: 235–246.
Wichmann A, Uyetake L, Su TT . E2F1 and E2F2 have opposite effects on radiation-induced p53-independent apoptosis in Drosophila. Dev Biol 2010; 346: 80–89.
Bennett K, Callard R, Heywood W, Harper J, Jayakumar A, LC G et al. New role for LEKTI in skin barrier formation: label-free quantitative proteomic identification of caspase 14 as a novel target for the protease inhibitor LEKTI. J Proteome Res 2010; 9: 4289–4294.
Long SW, Ooi JY, Yau PM, Jones PL . A brain-derived MeCP2 complex supports a role for MeCP2 in RNA processing. Biosci Rep 2011; 31: 333–343.
Squillaro T, Alessio N, Cipollaro M, Renieri A, Giordano A, Galderisi U . Partial silencing of methyl cytosine protein binding 2 (MECP2) in mesenchymal stem cells induces senescence with an increase in damaged DNA. FASEB J 2010; 24: 1593–1603.
Papait R, Magrassi L, Rigamonti D, Cattaneo E . Temozolomide and carmustine cause large-scale heterochromatin reorganization in glioma cells. Biochem Bioph Res Co 2009; 379: 434–439.
Rasti M, Arabsolghar R, Khatooni Z, Mostafavi-Pour Z . p53 Binds to estrogen receptor 1 promoter in human breast cancer cells. Pathol Oncol Res 2011; 18: 169–175.
Deuring R, Fanti L, Armstrong JA, Sarte M, Papoulas O, Prestel M et al. The ISWI chromatin-remodeling protein is required for gene expression and the maintenance of higher order chromatin structure in vivo. Mol Cell 2000; 5: 355–365.
Stultz BG, Lee H, Ramon K, Hursh DA . Decapentaplegic head capsule mutations disrupt novel peripodial expression controlling the morphogenesis of the Drosophila ventral head. Dev Biol 2006; 296: 329–339.
Acknowledgements
We thank Drs Hermann Steller, Laura Johnston and Tin Tin Su; the Bloomington Stock Center for fly stocks; Dr Dong Han at National Center for Nanoscience and Technology for help with the scanning electron microscope; and the members of the Bi laboratory for advice and discussions. We also thank Dr Mark Mortin for generating the knockout clbn flies and for critically reading this manuscript. We are grateful for comments on this manuscript from Tehyen Chu and Brent McCright. This work was supported by grants from National Basic Research Program of China (973 Program grant no. 2010CB934004), National Natural Science Foundation of China (grant no. 30871388) and CAS Knowledge Innovation Program to XB and by the Food and Drug Administration, Center for Biologics Evaluation and Research.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on the Oncogene website
Supplementary information
Rights and permissions
About this article
Cite this article
Wang, Y., Wang, Z., Joshi, B. et al. The tumor suppressor Caliban regulates DNA damage-induced apoptosis through p53-dependent and -independent activity. Oncogene 32, 3857–3866 (2013). https://doi.org/10.1038/onc.2012.395
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2012.395