Abstract
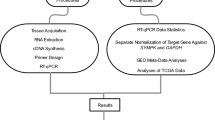
Differentiation is central to development, while dedifferentiation is central to cancer progression. Hence, a quantitative assessment of differentiation would be most useful. We propose an unbiased method to derive organ-specific differentiation indices from gene expression data and demonstrate its usefulness in thyroid cancer diagnosis. We derived a list of thyroid-specific genes by selecting automatically those genes that are expressed at higher level in the thyroid than in any other organ in a normal tissue's genome-wide gene expression compendium. The thyroid index of a tissue was defined as the median expression of these thyroid-specific genes in that tissue. As expected, the thyroid index was inversely correlated with meta-PCNA, a proliferation metagene, across a wide range of thyroid tumors. By contrast, the two indices were positively correlated in a time course of thyroid-stimulating hormone (TSH) activation of primary thyrocytes. Thus, the thyroid index captures biological information not integrated by proliferation rates. The differential diagnostic of follicular thyroid adenomas and follicular thyroid carcinoma is a notorious challenge for pathologists. The thyroid index discriminated them as accurately as did machine-learning classifiers trained on the genome-wide cancer data. Hence, although it was established exclusively from normal tissue data, the thyroid index integrates the relevant diagnostic information contained in tumoral transcriptomes. Similar results were obtained for the classification of the follicular vs classical variants of papillary thyroid cancers, that is, tumors dedifferentiating along a different route. The automated procedures demonstrated in the thyroid are applicable to other organs.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Accession codes
References
Aldred MA, Huang Y, Liyanarachchi S, Pellegata NS, Gimm O, Jhiang S et al. (2004). Papillary and follicular thyroid carcinomas show distinctly different microarray expression profiles and can be distinguished by a minimum of five genes. J Clin Oncol 22: 3531–3539.
Baloch ZW, Hendreen S, Gupta PK, LiVolsi VA, Mandel SJ, Weber R et al. (2001). Interinstitutional review of thyroid fine-needle aspirations: impact on clinical management of thyroid nodules. Diagn Cytopathol 25: 231–234.
Clary KM, Condel JL, Liu Y, Johnson DR, Grzybicki DM, Raab SS . (2005). Interobserver variability in the fine needle aspiration biopsy diagnosis of follicular lesions of the thyroid gland. Acta Cytol 49: 378–382.
DeLong ER, DeLong DM, Clarke-Pearson DL . (1988). Comparing the areas under two or more correlated receiver operating characteristic curves: a nonparametric approach. Biometrics 44: 837–845.
Delys L, Detours V, Franc B, Thomas G, Bogdanova T, Tronko M et al. (2007). Gene expression and the biological phenotype of papillary thyroid carcinomas. Oncogene 26: 7894–7903.
Detours V, Delys L, Libert F, Weiss Solís D, Bogdanova T, Dumont JE et al. (2007). Genome-wide gene expression profiling suggests distinct radiation susceptibilities in sporadic and post-Chernobyl papillary thyroid cancers. Br J Cancer 97: 818–825.
Finley DJ, Zhu B, Barden CB, Fahey TJ . (2004). Discrimination of benign and malignant thyroid nodules by molecular profiling. Ann Surg 240: 425–436; discussion 436–437.
Ge X, Yamamoto S, Tsutsumi S, Midorikawa Y, Ihara S, Wang SM et al. (2005). Interpreting expression profiles of cancers by genome-wide survey of breadth of expression in normal tissues. Genomics 86: 127–141.
Gentleman RC, Carey VJ, Bates DM, Bolstad B, Dettling M, Dudoit S et al. (2004). Bioconductor: open software development for computational biology and bioinformatics. Genome Biol 5: R80.
Hegedüs L . (2004). Clinical practice. The thyroid nodule. N Engl J Med 351: 1764–1771.
Irizarry RA, Hobbs B, Collin F, Beazer-Barclay YD, Antonellis KJ, Scherf U et al. (2003). Exploration, normalization, and summaries of high density oligonucleotide array probe level data. Biostatistics 4: 249–264.
Jongeneel CV, Delorenzi M, Iseli C, Zhou D, Haudenschild CD, Khrebtukova I et al. (2005). An atlas of human gene expression from massively parallel signature sequencing (MPSS). Genome Res 15: 1007–1014.
Kebebew E, Greenspan FS, Clark OH, Woeber KA, McMillan A . (2005). Anaplastic thyroid carcinoma. Treatment outcome and prognostic factors. Cancer 103: 1330–1335.
Kim D, Salzberg SL . (2011). TopHat-Fusion: an algorithm for discovery of novel fusion transcripts. Genome Biol 12: R72.
Kohlmann A, Kipps TJ, Rassenti LZ, Downing JR, Shurtleff SA, Mills KI et al. (2008). An international standardization programme towards the application of gene expression profiling in routine leukaemia diagnostics: the Microarray Innovations in LEukemia study prephase. Br J Haematol 142: 802–807.
Kondo T, Ezzat S, Asa SL . (2006). Pathogenetic mechanisms in thyroid follicular-cell neoplasia. Nat Rev Cancer 6: 292–306.
Lang BH-H, Lo C-Y, Chan W-F, Lam AK-Y, Wan K-Y . (2006). Classical and follicular variant of papillary thyroid carcinoma: a comparative study on clinicopathologic features and long-term outcome. World J Surg 30: 752–758.
Lin HW, Bhattacharyya N . (2010). Clinical behavior of follicular variant of papillary thyroid carcinoma: presentation and survival. Laryngoscope 120 (Suppl 4): S163.
Lubitz CC, Gallagher LA, Finley DJ, Zhu B, Fahey TJ . (2005). Molecular analysis of minimally invasive follicular carcinomas by gene profiling. Surgery 138: 1042–1048; discussion 1048–1049.
Mills KI, Kohlmann A, Williams PM, Wieczorek L, Liu W-min, Li R et al. (2009). Microarray-based classifiers and prognosis models identify subgroups with distinct clinical outcomes and high risk of AML transformation of myelodysplastic syndrome. Blood 114: 1063–1072.
Moreno JC, Klootwijk W, van Toor H, Pinto G, D'Alessandro M, Lèger A et al. (2008). Mutations in the iodotyrosine deiodinase gene and hypothyroidism. N Engl J Med 358: 1811–1818.
Mullighan CG, Miller CB, Radtke I, Phillips LA, Dalton J, Ma J et al. (2008). BCR-ABL1 lymphoblastic leukaemia is characterized by the deletion of Ikaros. Nature 453: 110–114.
Novershtern N, Subramanian A, Lawton LN, Mak RH, Haining WN, McConkey ME et al. (2011). Densely interconnected transcriptional circuits control cell states in human hematopoiesis. Cell 144: 296–309.
R Development Core Team, R: A Language and Environment for Statistical Computing. 1: ISBN 3-900051-07-0.
Rhodes DR, Yu J, Shanker K, Deshpande N, Varambally R, Ghosh D et al. (2004). Large-scale meta-analysis of cancer microarray data identifies common transcriptional profiles of neoplastic transformation and progression. Proc Natl Acad Sci USA 101: 9309–9314.
Roberts A, Pimentel H, Trapnell C, Pachter L . (2011). Identification of novel transcripts in annotated genomes using RNA-Seq. Bioinformatics 27: 2325–2329.
Roth RB, Hevezi P, Lee J, Willhite D, Lechner SM, Foster AC et al. (2006). Gene expression analyses reveal molecular relationships among 20 regions of the human CNS. Neurogenetics 7: 67–80.
Ruschhaupt M, Huber W, Poustka A, Mansmann U . (2004). A compendium to ensure computational reproducibility in high-dimensional classification tasks. Stat Appl Genet Mol Biol 3: Article37.
van Staveren WCG, Solís DW, Delys L, Venet D, Cappello M, Andry G et al. (2006). Gene expression in human thyrocytes and autonomous adenomas reveals suppression of negative feedbacks in tumorigenesis. Proc Natl Acad Sci USA 103: 413–418.
Su AI, Cooke MP, Ching KA, Hakak Y, Walker JR, Wiltshire T et al. (2002). Large-scale analysis of the human and mouse transcriptomes. Proc Natl Acad Sci USA 99: 4465–4470.
Taminau J, Steenhoff D, Coletta A, Meganck S, Lazar C, de Schaetzen V et al. (2011). inSilicoDb: an R/Bioconductor package for accessing human Affymetrix expert-curated datasets from GEO. Bioinformatics [Internet]. Available from:http://www.ncbi.nlm.nih.gov/pubmed/21937664.
Venet D, Dumont JE, Detours V . (2011). Most random gene expression signatures are significantly associated with breast cancer outcome. PLoS Comput Biol 7: e1002240.
Wang Z, Gerstein M, Snyder M . (2009). RNA-Seq: a revolutionary tool for transcriptomics. Nat Rev Genet 10: 57–63.
Wickham H . (2009). ggplot2: Elegant Graphics for Data Analysis 2nd edn. Springer (http://cran.r-project.org/).
Wu Z, Irizarry RA, Gentleman R, Martinez-Murillo F, Spencer F . (2004). A model-based background adjustment for oligonucleotide expression arrays. J Am Stat Assoc 99: 909–917.
Zurzolo C, Gentile R, Mascia A, Garbi C, Polistina C, Aloj L et al. (1991). The polarized epithelial phenotype is dominant in hybrids between polarized and unpolarized rat thyroid cell lines. J Cell Sci 98 (Part 1): 65–73.
Acknowledgements
This research was partially funded by the Brussels-Capital IRSIB project ICT-impulse 2006, In Silico Wet Lab. GT is supported by the Wallonie–Bruxelles International grant (7450/AMG/VDL/IN,WBI/doh/2009/21649). MT is supported by a FRIA fellowship from FNRS.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on the Oncogene website
Supplementary information
Rights and permissions
About this article
Cite this article
Tomás, G., Tarabichi, M., Gacquer, D. et al. A general method to derive robust organ-specific gene expression-based differentiation indices: application to thyroid cancer diagnostic. Oncogene 31, 4490–4498 (2012). https://doi.org/10.1038/onc.2011.626
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2011.626
Keywords
This article is cited by
-
Robustness evaluations of pathway activity inference methods on gene expression data
BMC Bioinformatics (2024)
-
PFKFB3 facilitates cell proliferation and migration in anaplastic thyroid carcinoma via the WNT/β‐catenin signaling pathway
Endocrine (2024)
-
Estrogen-related genes for thyroid cancer prognosis, immune infiltration, staging, and drug sensitivity
BMC Cancer (2023)
-
Adding pieces to the puzzle of differentiated-to-anaplastic thyroid cancer evolution: the oncogene E2F7
Cell Death & Disease (2023)
-
LncRNA H19 is a potential biomarker and correlated with immune infiltration in thyroid carcinoma
Clinical and Experimental Medicine (2022)