Abstract
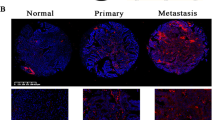
MicroRNAs (miRNAs) play important roles in tumorigenesis by regulating oncogenes and tumor-suppressor genes. In this study, miR-187 and miR-200a were found to be expressed at higher levels in ovarian cancers than in benign tumors. In patients with ovarian cancer, however, higher levels of miR-187 and miR-200a expression were paradoxically associated with better OS and recurrence-free survival. Further, multivariate analysis showed that miR-187 served as an independent prognostic factor for patients with ovarian cancer (n=176). Computational prediction and microarray results indicated that miR-187 directly targeted Disabled homolog-2 (Dab2), and luciferase reporter assays confirmed that the target site of miR-187 was located at the 3′-UTR of the Dab2 gene. Generally considered as a tumor-suppressor gene, Dab2 may actually promote tumor progression in advanced cancers through epithelial-to-mesenchymal transition (EMT). Ectopic expression of miR-187 in cancer cells promoted cell proliferation, but continued overexpression of miR-187 suppressed Dab2 and inhibited migration. Suppression of miR-187 upregulated Dab2, which, by inhibiting E-cadherin levels while stimulating vimentin and phospho-FAK levels, promoted EMT. Reduced ovarian cancer Dab2 histoscores correlated with high miR-187 levels and improved outcomes of patients. Collectively, these results demonstrate distinct dual roles of Dab2 in cell proliferation and tumor progression. In the initial steps of tumorigenesis, upregulated miR-187 suppresses Dab2, promoting cell proliferation. During the later stages, however, continued increased levels of miR-187 inhibits the Dab2-dependent EMT that is associated with tumor invasiveness, which is presumed to be the reason why cancers with high miR-187 levels were associated with better survivals.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Bartel DP . (2004). MicroRNAs: genomics, biogenesis, mechanism, and function. Cell 116: 281–297.
Bast Jr RC, Feeney M, Lazarus H, Nadler LM, Colvin RB, Knapp RC . (1981). Reactivity of a monoclonal antibody with human ovarian carcinoma. J Clin Invest 68: 1331–1337.
Berek JS, Friedlander M, Hacker NF . (2010). Epithelial ovarian, fallopian tube, and peritoneal cancer. In: Berek JS, Hacker NF (eds). Berek & Hacker's Gynecologic Oncology. Lippincott Williams & Wilkins, pp 444–508.
Braun J, Hoang-Vu C, Dralle H, Huttelmaier S . (2010). Downregulation of microRNAs directs the EMT and invasive potential of anaplastic thyroid carcinomas. Oncogene 29: 4237–4244.
Chao A, Wang TH, Lee YS, Hsueh S, Chao AS, Chang TC et al. (2006). Molecular characterization of adenocarcinoma and squamous carcinoma of the uterine cervix using microarray analysis of gene expression. Int J Cancer 119: 91–98.
Chao A, Tsai CL, Wei PC, Hsueh S, Chao AS, Wang CJ et al. (2010). Decreased expression of microRNA-199b increases protein levels of SET (protein phosphatase 2A inhibitor) in human choriocarcinoma. Cancer Lett 291: 99–107.
Chaudhury A, Hussey GS, Ray PS, Jin G, Fox PL, Howe PH . (2010). TGF-beta-mediated phosphorylation of hnRNP E1 induces EMT via transcript-selective translational induction of Dab2 and ILEI. Nat Cell Biol 12: 286–293.
Choi JH, Choi KC, Auersperg N, Leung PC . (2006). Differential regulation of two forms of gonadotropin-releasing hormone messenger ribonucleic acid by gonadotropins in human immortalized ovarian surface epithelium and ovarian cancer cells. Endocr Relat Cancer 13: 641–651.
Chung CM, Man C, Jin Y, Jin C, Guan XY, Wang Q et al. (2005). Amplification and overexpression of aurora kinase A (AURKA) in immortalized human ovarian epithelial (HOSE) cells. Mol Carcinog 43: 165–174.
Eitan R, Kushnir M, Lithwick-Yanai G, David MB, Hoshen M, Glezerman M et al. (2009). Tumor microRNA expression patterns associated with resistance to platinum based chemotherapy and survival in ovarian cancer patients. Gynecol Oncol 114: 253–259.
Fazili Z, Sun W, Mittelstaedt S, Cohen C, Xu XX . (1999). Disabled-2 inactivation is an early step in ovarian tumorigenicity. Oncogene 18: 3104–3113.
Gregory PA, Bert AG, Paterson EL, Barry SC, Tsykin A, Farshid G et al. (2008). The miR-200 family and miR-205 regulate epithelial to mesenchymal transition by targeting ZEB1 and SIP1. Nat Cell Biol 10: 593–601.
Griffiths-Jones S . (2004). The microRNA registry. Nucleic Acids Res 32: D109–D111.
Hannigan A, Smith P, Kalna G, Lo Nigro C, Orange C, O'Brien DI et al. (2010). Epigenetic downregulation of human disabled homolog 2 switches TGF-beta from a tumor suppressor to a tumor promoter. J Clin Invest 120: 2842–2857.
Hu X, Macdonald DM, Huettner PC, Feng Z, El Naqa IM, Schwarz JK et al. (2009). A miR-200 microRNA cluster as prognostic marker in advanced ovarian cancer. Gynecol Oncol 114: 457–464.
Iorio MV, Visone R, Di Leva G, Donati V, Petrocca F, Casalini P et al. (2007). MicroRNA signatures in human ovarian cancer. Cancer Res 67: 8699–8707.
Jemal A, Siegel R, Ward E, Murray T, Xu J, Thun MJ . (2007). Cancer statistics, 2007. CA Cancer J Clin 57: 43–66.
Laios A, O'Toole S, Flavin R, Martin C, Kelly L, Ring M et al. (2008). Potential role of miR-9 and miR-223 in recurrent ovarian cancer. Mol Cancer 7: 35.
Lee YS, Chen CH, Tsai CN, Tsai CL, Chao A, Wang TH . (2009). Microarray labeling extension values: laboratory signatures for Affymetrix GeneChips. Nucleic Acids Res 37: e61.
Liao CJ, Wu TI, Huang YH, Chang TC, Wang CS, Tsai MM et al. (2011). Overexpression of gelsolin in human cervical carcinoma and its clinicopathological significance. Gynecol Oncol 120: 135–144.
Lu L, Katsaros D, de la Longrais IA, Sochirca O, Yu H . (2007). Hypermethylation of let-7a-3 in epithelial ovarian cancer is associated with low insulin-like growth factor-II expression and favorable prognosis. Cancer Res 67: 10117–10122.
Metzler M, Wilda M, Busch K, Viehmann S, Borkhardt A . (2004). High expression of precursor microRNA-155/BIC RNA in children with Burkitt lymphoma. Genes Chromosomes Cancer 39: 167–169.
Mok SC, Wong KK, Chan RK, Lau CC, Tsao SW, Knapp RC et al. (1994). Molecular cloning of differentially expressed genes in human epithelial ovarian cancer. Gynecol Oncol 52: 247–252.
Mok SC, Chan WY, Wong KK, Cheung KK, Lau CC, Ng SW et al. (1998). DOC-2, a candidate tumor suppressor gene in human epithelial ovarian cancer. Oncogene 16: 2381–2387.
Nam EJ, Yoon H, Kim SW, Kim H, Kim YT, Kim JH et al. (2008). MicroRNA expression profiles in serous ovarian carcinoma. Clin Cancer Res 14: 2690–2695.
Peter ME . (2009). Let-7 and miR-200 microRNAs: guardians against pluripotency and cancer progression. Cell Cycle 8: 843–852.
Prunier C, Howe PH . (2005). Disabled-2 (Dab2) is required for transforming growth factor beta-induced epithelial to mesenchymal transition (EMT). J Biol Chem 280: 17540–17548.
Singh A, Settleman J . (2010). EMT, cancer stem cells and drug resistance: an emerging axis of evil in the war on cancer. Oncogene 29: 4741–4751.
Takamizawa J, Konishi H, Yanagisawa K, Tomida S, Osada H, Endoh H et al. (2004). Reduced expression of the let-7 microRNAs in human lung cancers in association with shortened postoperative survival. Cancer Res 64: 3753–3756.
Targetscan4.0 (2007). Prediction of microRNA targets. http://www.targetscanorg/cgi-bin/targetscan/vert_40/view_genecgi?taxid=9606&gs=DAB2&showcnc=0&shownc=0.
Thiery JP . (2002). Epithelial–mesenchymal transitions in tumour progression. Nat Rev Cancer 2: 442–454.
Tsai MS, Hwang SM, Chen KD, Lee YS, Hsu LW, Chang YJ . et al. (2007). Functional network analysis of the transcriptomes of mesenchymal stem cells derived from amniotic fluid, amniotic membrane, cord blood, and bone marrow. Stem Cells 25: 2511–2523.
Tusher VG, Tibshirani R, Chu G . (2001). Significance analysis of microarrays applied to the ionizing radiation response. Proc Natl Acad Sci USA 98: 5116–5121.
Vetter G, Saumet A, Moes M, Vallar L, Le Bechec A, Laurini C et al. (2010). miR-661 expression in SNAI1-induced epithelial to mesenchymal transition contributes to breast cancer cell invasion by targeting Nectin-1 and StarD10 messengers. Oncogene 29: 4436–4448.
Volinia S, Calin GA, Liu CG, Ambs S, Cimmino A, Petrocca F et al. (2006). A microRNA expression signature of human solid tumors defines cancer gene targets. Proc Natl Acad Sci USA 103: 2257–2261.
Wang TH, Wang HS, Ichijo H, Giannakakou P, Foster JS, Fojo T et al. (1998). Microtubule-interfering agents activate c-Jun N-terminal kinase/stress-activated protein kinase through both Ras and apoptosis signal-regulating kinase pathways. J Biol Chem 273: 4928–4936.
Yanaihara N, Caplen N, Bowman E, Seike M, Kumamoto K, Yi M et al. (2006). Unique microRNA molecular profiles in lung cancer diagnosis and prognosis. Cancer Cell 9: 189–198.
Yang DH, Fazili Z, Smith ER, Cai KQ, Klein-Szanto A, Cohen C et al. (2006). Disabled-2 heterozygous mice are predisposed to endometrial and ovarian tumorigenesis and exhibit sex-biased embryonic lethality in a p53-null background. Am J Pathol 169: 258–267.
Yang H, Kong W, He L, Zhao JJ, O'Donnell JD, Wang J et al. (2008). MicroRNA expression profiling in human ovarian cancer: miR-214 induces cell survival and cisplatin resistance by targeting PTEN. Cancer Res 68: 425–433.
Yang WL, Godwin AK, Xu XX . (2004). Tumor necrosis factor-alpha-induced matrix proteolytic enzyme production and basement membrane remodeling by human ovarian surface epithelial cells: molecular basis linking ovulation and cancer risk. Cancer Res 64: 1534–1540.
Acknowledgements
We thank Ching-Ling Wang, Jung-Erh Yang and Ying-Yu Lin for technical assistance; the Division of Gynecologic Oncology of Chang Gung Memorial Hospital for clinical data retrieval; Shih-Yee Mimi Wang (University of Illinois College of Medicine, Rockford) for language editing and Professor Ching-Ping Tseng (Chang Gung University) for discussion and advices. This study was supported by grants from the National Science Council (NSC99-2314-B-182A-087-MY3 to AC), the National Research Program for Genomic Medicine (NSC97-3112-B-001-020 to Y-SL), the Chang Gung Medical Foundation (CMRPG360953/4, 391451 to AC; IRB No. 95-1364B, 97-1444C, 99-2072C, 99-2411C) and the Department of Health (DOH99-TD-B-111-005 to C-HL; DOH99-TD-C-111-006 to AC and T-HW).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on the Oncogene website
Supplementary information
Rights and permissions
About this article
Cite this article
Chao, A., Lin, CY., Lee, YS. et al. Regulation of ovarian cancer progression by microRNA-187 through targeting Disabled homolog-2. Oncogene 31, 764–775 (2012). https://doi.org/10.1038/onc.2011.269
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2011.269
Keywords
This article is cited by
-
Disable 2, A Versatile Tissue Matrix Multifunctional Scaffold Protein with Multifaceted Signaling: Unveiling Role in Breast Cancer for Therapeutic Revolution
Cell Biochemistry and Biophysics (2024)
-
Disabled-2, a versatile tissue matrix multifunctional scaffold protein with multifaceted signaling: Unveiling its potential in the cancer battle
Naunyn-Schmiedeberg's Archives of Pharmacology (2024)
-
Identification of novel miRNAs potentially involved in the pathogenesis of adult T-cell leukemia/lymphoma using WGCNA followed by RT-qPCR test of hub genes
Infectious Agents and Cancer (2023)
-
High-throughput sequencing identification of differentially expressed microRNAs in metastatic ovarian cancer with experimental validations
Cancer Cell International (2020)
-
A novel microRNA signature predicts survival in liver hepatocellular carcinoma after hepatectomy
Scientific Reports (2018)