Abstract
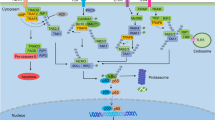
The oncogenic fusion protein RET/PTC3 (RP3) that is expressed in papillary thyroid carcinoma (PTC) and thyroid epithelia in Hashimoto's thyroiditis activates nuclear factor-kappa B (NF-κB) and induces pro-inflammatory gene expression; however, the mechanism of this activation is unknown. To address this, we expressed RP3 in murine embryonic fibroblasts (MEFs) lacking key classical and noncanonical NF-κB signaling components. In wild-type MEFs, RP3 upregulated CCL2, CXCL1, granulocyte-macrophage colony-stimulating factor and tumor necrosis factor expression and activated classical but not noncanonical NF-κB. RP3-activated NF-κB in IκB kinase (IKK)β−/− MEFs but not IKKα- or NF-κB essential modulator (NEMO)-deficient cells and activation was inhibited by a peptide that blocks NEMO binding to the IKKs. RP3 increased the levels of NF-κB-inducing kinase (NIK) and did not activate NF-κB in NIK-deficient MEFs. Notably, NIK stabilization was not accompanied by TRAF3 degradation demonstrating that RP3 disrupts normal basal NIK regulation. Dominant-negative NIK blocked RP3-induced NF-κB activation and an RP3 signaling mutant (RP3Y588F) did not stabilize NIK. Finally, examination of PTC specimens revealed strong positive staining for NIK. We therefore conclude that RP3 activates classical NF-κB via NIK, NEMO and IKKα. Importantly, our findings reveal a novel mechanism for oncogene-induced NF-κB activation via stabilization of NIK.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Annunziata CM, Davis RE, Demchenko Y, Bellamy W, Gabrea A, Zhan F et al. (2007). Frequent engagement of the classical and alternative NF-κB pathways by diverse genetic abnormalities in multiple myeloma. Cancer Cell 12: 115–130.
Basseres DS, Baldwin AS . (2006). NF-κB and IκB kinase pathways in oncogenic initiation and progression. Oncogene 25: 6817–6830.
Birbach A, Bailey ST, Ghosh S, Schmid JA . (2004). Cytosolic, nuclear and nucleolar localization signals determine subcellular distribution and activity of the NF-κB inducing kinase NIK. J Cell Sci 117: 3615–3624.
Borrello MG, Alberti L, Fischer A, Degl'innocenti D, Ferrario C, Gariboldi M et al. (2005). Induction of a proinflammatory program in normal human thyrocytes by the RET/PTC1 oncogene. Proc Natl Acad Sci USA 102: 14825–14830.
Bunone G, Uggeri M, Mondellini P, Pierotti MA, Bongarzone I . (2000). RET receptor expression in thyroid follicular epithelial cell-derived tumors. Cancer Res 60: 2845–2849.
Ciampi R, Nikiforov YE . (2007). RET/PTC rearrangements and BRAF mutations in thyroid tumorigenesis. Endocrinology 148: 936–941.
Dejardin E, Droin NM, Delhase M, Haas E, Cao Y, Makris C et al. (2002). The lymphotoxin-β receptor induces different patterns of gene expression via two NF-κB pathways. Immunity 17: 525–535.
Derudder E, Dejardin E, Pritchard LL, Green DR, Korner M, Baud V . (2003). RelB/p50 dimers are differentially regulated by tumor necrosis factor-α and lymphotoxin-β receptor activation: critical roles for p100. J Biol Chem 278: 23278–23284.
Eisenlohr LC, Rothstein JL . (2006). Oncogenic inflammation and autoimmune disease. Autoimmun Rev 6: 107–114.
Gilmore TD, Herscovitch M . (2006). Inhibitors of NF-κB signaling: 785 and counting. Oncogene 25: 6887–6899.
Hayashi H, Ichihara M, Iwashita T, Murakami H, Shimono Y, Kawai K et al. (2000). Characterization of intracellular signals via tyrosine 1062 in RET activated by glial cell line-derived neurotrophic factor. Oncogene 19: 4469–4475.
Hayden MS, Ghosh S . (2008). Shared principles in NF-κB signaling. Cell 132: 344–362.
He JQ, Saha SK, Kang JR, Zarnegar B, Cheng G . (2007). Specificity of TRAF3 in its negative regulation of the noncanonical NF-κB pathway. J Biol Chem 282: 3688–3694.
Karin M . (2006). NF-κB in cancer development and progression. Nature 441: 431–436.
Keats JJ, Fonseca R, Chesi M, Schop R, Baker A, Chng WJ et al. (2007). Promiscuous mutations activate the noncanonical NF-κB pathway in multiple myeloma. Cancer Cell 12: 131–144.
Lam LT, Davis RE, Ngo VN, Lenz G, Wright G, Xu W et al. (2008). Compensatory IKKα activation of classical NF-κB signaling during IKKβ inhibition identified by an RNA interference sensitization screen. Proc Natl Acad Sci USA 105: 20798–20803.
Lodyga M, De Falco V, Bai XH, Kapus A, Melillo RM, Santoro M et al. (2009). XB130, a tissue-specific adaptor protein that couples the RET/PTC oncogenic kinase to PI 3-kinase pathway. Oncogene 28: 937–949.
Ludwig L, Kessler H, Wagner M, Hoang-Vu C, Dralle H, Adler G et al. (2001). NF-κB is constitutively active in C-cell carcinoma and required for RET-induced transformation. Cancer Res 61: 4526–4535.
Madge LA, Kluger MS, Orange JS, May MJ . (2008). Lymphotoxin-α1β2 and LIGHT induce classical and noncanonical NF-κB-dependent proinflammatory gene expression in vascular endothelial cells. J Immunol 180: 3467–3477.
May MJ, D'Acquisto F, Madge LA, Glockner J, Pober JS, Ghosh S . (2000). Selective inhibition of NF-κB activation by a peptide that blocks the interaction of NEMO with the IκB kinase complex. Science 289: 1550–1554.
Monaco C, Visconti R, Barone MV, Pierantoni GM, Berlingieri MT, De Lorenzo C et al. (2001). The RFG oligomerization domain mediates kinase activation and re-localization of the RET/PTC3 oncoprotein to the plasma membrane. Oncogene 20: 599–608.
Muzza M, Degl'innocenti D, Colombo C, Perrino M, Ravasi E, Rossi S et al. (2009). The tight relationship between papillary thyroid cancer, autoimmunity and inflammation: clinical and molecular studies. Clin Endocrinol (Oxf) 72: 702–708.
Nellore A, Paziana K, Ma C, Tsygankova OM, Wang Y, Puttaswamy K et al. (2009). Loss of Rap1GAP in papillary thyroid cancer. J Clin Endocrinol Metab 94: 1026–1032.
Nibu K, Otsuki N, Nakao K, Sugasawa M, Rothstein JL . (2005). RET/PTC fusion gene rearrangements in Japanese thyroid carcinomas. Eur Arch Otorhinolaryngol 262: 368–373.
Nishina T, Yamaguchi N, Gohda J, Semba K, Inoue J . (2009). NIK is involved in constitutive activation of the alternative NF-κB pathway and proliferation of pancreatic cancer cells. Biochem Biophys Res Commun 388: 96–101.
Pacifico F, Leonardi A . (2010). Role of NF-κB in thyroid cancer. Mol Cell Endocrinol 321: 29–35.
Pacifico F, Mauro C, Barone C, Crescenzi E, Mellone S, Monaco M et al. (2004). Oncogenic and anti-apoptotic activity of NF-κB in human thyroid carcinomas. J Biol Chem 279: 54610–54619.
Pufnock JS, Rothstein JL . (2009). Oncoprotein signaling mediates tumor-specific inflammation and enhances tumor progression. J Immunol 182: 5498–5506.
Razani B, Zarnegar B, Ytterberg AJ, Shiba T, Dempsey PW, Ware CF et al. (2010). Negative feedback in noncanonical NF-κB signaling modulates NIK stability through IKKα-mediated phosphorylation. Sci Signal 3: ra41.
Rhoden KJ, Unger K, Salvatore G, Yilmaz Y, Vovk V, Chiappetta G et al. (2006). RET/papillary thyroid cancer rearrangement in nonneoplastic thyrocytes: follicular cells of Hashimoto's thyroiditis share low-level recombination events with a subset of papillary carcinoma. J Clin Endocrinol Metab 91: 2414–2423.
Russell JP, Shinohara S, Melillo RM, Castellone MD, Santoro M, Rothstein JL . (2003). Tyrosine kinase oncoprotein, RET/PTC3, induces the secretion of myeloid growth and chemotactic factors. Oncogene 22: 4569–4577.
Santoro M, Melillo RM, Grieco M, Berlingieri MT, Vecchio G, Fusco A . (1993). The TRK and RET tyrosine kinase oncogenes cooperate with ras in the neoplastic transformation of a rat thyroid epithelial cell line. Cell Growth Differ 4: 77–84.
Senftleben U, Cao Y, Xiao G, Greten FR, Krahn G, Bonizzi G et al. (2001). Activation by IKKα of a second, evolutionary conserved, NF-κB signaling pathway. Science 293: 1495–1499.
Sheils OM, O'Eary JJ, Uhlmann V, Lattich K, Sweeney EC . (2000). RET/PTC-1 activation in Hashimoto thyroiditis. Int J Surg Pathol 8: 185–189.
Solt LA, Madge LA, Orange JS, May MJ . (2007). Interleukin-1-induced NF-κB activation is NEMO-dependent but does not require IKKβ. J Biol Chem 282: 8724–8733.
Vallabhapurapu S, Matsuzawa A, Zhang W, Tseng PH, Keats JJ, Wang H et al. (2008). Nonredundant and complementary functions of TRAF2 and TRAF3 in a ubiquitination cascade that activates NIK-dependent alternative NF-κB signaling. Nat Immunol 9: 1364–1370.
Veiga-Fernandes H, Coles MC, Foster KE, Patel A, Williams A, Natarajan D et al. (2007). Tyrosine kinase receptor RET is a key regulator of Peyer's patch organogenesis. Nature 446: 547–551.
Visconti R, Cerutti J, Battista S, Fedele M, Trapasso F, Zeki K et al. (1997). Expression of the neoplastic phenotype by human thyroid carcinoma cell lines requires NF-κB p65 protein expression. Oncogene 15: 1987–1994.
Wells Jr SA, Santoro M . (2009). Targeting the RET pathway in thyroid cancer. Clin Cancer Res 15: 7119–7123.
Wharry CE, Haines KM, Carroll RG, May MJ . (2009). Constitutive non-canonical NF-κB signaling in pancreatic cancer cells. Cancer Biol Ther 8: 1567–1576.
Wirtschafter A, Schmidt R, Rosen D, Kundu N, Santoro M, Fusco A et al. (1997). Expression of the RET/PTC fusion gene as a marker for papillary carcinoma in Hashimoto's thyroiditis. Laryngoscope 107: 95–100.
Xiao G, Harhaj EW, Sun SC . (2001). NF-κB-inducing kinase regulates the processing of NF-κB2 p100. Mol Cell 7: 401–409.
Zarnegar B, Yamazaki S, He JQ, Cheng G . (2008). Control of canonical NF-κB activation through the NIK-IKK complex pathway. Proc Natl Acad Sci USA 105: 3503–3508.
Acknowledgements
This work was supported by grants from the National Institutes of Health (RO1HL080612, R21CA135602 and T32CA009683, T32GM07229) and the Mari Lowe Center for Comparative Oncology.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict interest.
Additional information
Supplementary Information accompanies the paper on the Oncogene website
Supplementary information
Rights and permissions
About this article
Cite this article
Neely, R., Brose, M., Gray, C. et al. The RET/PTC3 oncogene activates classical NF-κB by stabilizing NIK. Oncogene 30, 87–96 (2011). https://doi.org/10.1038/onc.2010.396
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2010.396
Keywords
This article is cited by
-
NF-κB signaling activation and roles in thyroid cancers: implication of MAP3K14/NIK
Oncogenesis (2023)
-
Long non-coding RNA DIO3OS/let-7d/NF-κB2 axis regulates cells proliferation and metastasis of thyroid cancer cells
Journal of Cell Communication and Signaling (2021)
-
NF-κB inducing kinase (NIK) modulates melanoma tumorigenesis by regulating expression of pro-survival factors through the β-catenin pathway
Oncogene (2012)