Abstract
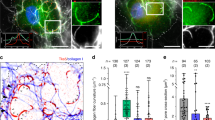
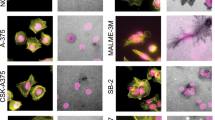
During metastasis, invading cells produce various actin-based membrane protrusions that promote directional migration and proteolysis of extracellular matrix (ECM). Observations of actin staining within thin, tubulin-based microtentacle (McTN) protrusions in suspended MDA-MB-231 tumor cells, prompted an investigation of whether McTNs are structural or functional analogs of invadopodia. We show here that MDA-MB-231 cells are capable of producing invadopodia and McTNs, both of which contain F-actin. Invadopodium formation was enhanced by the expression of a constitutively active c-Src kinase, and repressed by the expression of dominant-negative, catalytically inactive form of c-Src. In contrast, expression of inactive c-Src significantly increased McTN formation. Direct inhibition of c-Src with the SU6656 inhibitor compound also significantly enhanced McTN formation, but suppressed invadopodia, including the appearance of F-actin cores and phospho-cortactin foci, as well as completely blocking focal degradation of ECM. In addition, silencing of Tks5 in Src-transformed fibroblasts blocked invadopodia without affecting McTNs. Genetic modification of c-Src activity that promoted McTN formation augmented capillary retention of circulating tumor cells in vivo and rapid re-attachment of suspended cells in vitro, even though invadopodia were strongly suppressed. These results indicate that McTNs are capable of enhancing tumor cell reattachment, even in the absence of Tks5 and active Src, and define separate cytoskeletal mechanisms and functions for McTNs and invadopodia.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Abbreviations
- ECM:
-
extracellular matrix
- McTN:
-
microtentacle
- F-actin:
-
filamentous actin
- PDGFR:
-
platelet derived growth factor receptor
References
Balzer EM, Whipple RA, Cho EH, Matrone MA, Martin SS . (2010). Antimitotic chemotherapeutics promote adhesive responses in detached and circulating tumor cells. Breast Cancer Res Treat 121: 65–78.
Blake RA, Broome MA, Liu X, Wu J, Gishizky M, Sun L et al. (2000). SU6656, a selective src family kinase inhibitor, used to probe growth factor signaling. Mol Cell Biol 20: 9018–9027.
Buccione R, Caldieri G, Ayala I . (2009). Invadopodia: specialized tumor cell structures for the focal degradation of the extracellular matrix. Cancer Metastasis Rev 28: 137–149.
Buccione R, Orth JD, McNiven MA . (2004). Foot and mouth: podosomes, invadopodia and circular dorsal ruffles. Nat Rev Mol Cell Biol 5: 647–657.
Chambers AF, Groom AC, MacDonald IC . (2002). Dissemination and growth of cancer cells in metastatic sites. Nat Rev Cancer 2: 563–572.
Clark ES, Weaver AM . (2008). A new role for cortactin in invadopodia: regulation of protease secretion. Eur J Cell Biol 87: 581–590.
Frame MC, Fincham VJ, Carragher NO, Wyke JA . (2002). v-Src's hold over actin and cell adhesions. Nat Rev Mol Cell Biol 3: 233–245.
Gupton SL, Gertler FB . (2007). Filopodia: the fingers that do the walking. Sci STKE 2007: re5.
Korb T, Schlüter K, Enns A, Spiegel HU, Senninger N, Nicolson GL et al. (2004). Integrity of actin fibers and microtubules influences metastatic tumor cell adhesion. Exp Cell Res 299: 236–247.
Linder S . (2007). The matrix corroded: podosomes and invadopodia in extracellular matrix degradation. Trends Cell Biol 17: 107–117.
Linder S, Aepfelbacher M . (2003). Podosomes: adhesion hot-spots of invasive cells. Trends Cell Biol 13: 376–385.
Machesky LM . (2008). Lamellipodia and filopodia in metastasis and invasion. FEBS Lett 582: 2102–2111.
Mehlen P, Puisieux A . (2006). Metastasis: a question of life or death. Nat Rev Cancer 6: 449–458.
Mendoza M, Khanna C . (2009). Revisiting the seed and soil in cancer metastasis. Int J Biochem Cell Biol 41: 1452–1462.
Miles FL, Pruitt FL, van Golen KL, Cooper CR . (2008). Stepping out of the flow: capillary extravasation in cancer metastasis. Clin Exp Metastasis 25: 305–324.
Mitra SK, Schlaepfer DD . (2006). Integrin-regulated FAK-Src signaling in normal and cancer cells. Curr Opin Cell Biol 18: 516–523.
Seals DF, Azucena Jr EF, Pass I, Tesfay L, Gordon R, Woodrow M et al. (2005). The adaptor protein Tks5/Fish is required for podosome formation and function, and for the protease-driven invasion of cancer cells. Cancer Cell 7: 155–165.
Whipple RA, Balzer EM, Cho EH, Matrone MA, Yoon JR, Martin SS . (2008). Vimentin filaments support extension of tubulin-based microtentacles in detached breast tumor cells. Cancer Res 68: 5678–5688.
Whipple RA, Cheung AM, Martin SS . (2007). Detyrosinated microtubule protrusions in suspended mammary epithelial cells promote reattachment. Exp Cell Res 313: 1326–1336.
Yilmaz M, Christofori G . (2009). EMT, the cytoskeleton, and cancer cell invasion. Cancer Metastasis Rev 28: 15–33.
Acknowledgements
This study was supported by R01-CA124704 grant from the National Cancer Institute, Breast Cancer Idea Award from USA Medical Research and Materiel Command (BC061047) and a Susan G Komen investigator-initiated grant (KG100240). We are very grateful to Darren Seals and Sara Courtneidge for generously providing cell lines and reagents to examine the role of Tks5.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on the Oncogene website
Rights and permissions
About this article
Cite this article
Balzer, E., Whipple, R., Thompson, K. et al. c-Src differentially regulates the functions of microtentacles and invadopodia. Oncogene 29, 6402–6408 (2010). https://doi.org/10.1038/onc.2010.360
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2010.360
Keywords
This article is cited by
-
Microtubule disruption reduces metastasis more effectively than primary tumor growth
Breast Cancer Research (2022)
-
Aspartate β-hydroxylase promotes pancreatic ductal adenocarcinoma metastasis through activation of SRC signaling pathway
Journal of Hematology & Oncology (2019)
-
Evaluation of α-tubulin, detyrosinated α-tubulin, and vimentin in CTCs: identification of the interaction between CTCs and blood cells through cytoskeletal elements
Breast Cancer Research (2018)
-
Data-Driven Discovery of Extravasation Pathway in Circulating Tumor Cells
Scientific Reports (2017)
-
Mechanical interplay between invadopodia and the nucleus in cultured cancer cells
Scientific Reports (2015)