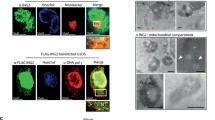
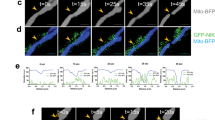
Abstract
The insulin-like growth factor (IGF-I) signalling pathway is essential for metabolism, cell growth and survival. It induces expression of the mitochondrial pyrimidine nucleotide carrier 1 (PNC1) in transformed cells, but the consequences of this for cell phenotype are unknown. Here we show that PNC1 is necessary to maintain mitochondrial function by controlling mitochondrial DNA replication and the ratio of transcription of mitochondrial genes relative to nuclear genes. PNC1 suppression causes reduced oxidative phosphorylation and leakage of reactive oxygen species (ROS), which activates the AMPK-PGC1α signalling pathway and promotes mitochondrial biogenesis. Overexpression of PNC1 suppresses mitochondrial biogenesis. Suppression of PNC1 causes a profound ROS-dependent epithelial–mesenchymal transition (EMT), whereas overexpression of PNC1 suppresses both basal EMT and induction of EMT by TGF-β. Overall, our findings indicate that PNC1 is essential for mitochondria maintenance and suggest that its induction by IGF-I facilitates cell growth whereas protecting cells from an ROS-promoted differentiation programme that arises from mitochondrial dysfunction.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Ayllon V, O'Connor R . (2007). PBK/TOPK promotes tumour cell proliferation through p38 MAPK activity and regulation of the DNA damage response. Oncogene 26: 3451–3461.
Brandon M, Baldi P, Wallace DC . (2006). Mitochondrial mutations in cancer. Oncogene 25: 4647–4662.
Butow RA, Avadhani NG . (2004). Mitochondrial signaling: the retrograde response. Mol Cell 14: 1–15.
Chatterjee A, Mambo E, Sidransky D . (2006). Mitochondrial DNA mutations in human cancer. Oncogene 25: 4663–4674.
Copeland WC . (2008). Inherited mitochondrial diseases of DNA replication. Annu Rev Med 59: 131–146.
Deberardinis RJ, Sayed N, Ditsworth D, Thompson CB . (2008). Brick by brick: metabolism and tumor cell growth. Curr Opin Genet Dev 18: 54–61.
Elstrom RL, Bauer DE, Buzzai M, Karnauskas R, Harris MH, Plas DR et al. (2004). Akt stimulates aerobic glycolysis in cancer cells. Cancer Res 64: 3892–3899.
Falkenberg M, Larsson NG, Gustafsson CM . (2007). DNA replication and transcription in mammalian mitochondria. Annu Rev Biochem 76: 679–699.
Ferraro D, Corso S, Fasano E, Panieri E, Santangelo R, Borrello S et al. (2006). Pro-metastatic signaling by c-Met through RAC-1 and reactive oxygen species (ROS). Oncogene 25: 3689–3698.
Finkel T . (2000). Redox-dependent signal transduction. FEBS Lett 476: 52–54.
Floyd S, Favre C, Lasorsa FM, Leahy M, Trigiante G, Stroebel P et al. (2007). The insulin-like growth factor-I mTOR signaling pathway induces the mitochondrial pyrimidine nucleotide carrier to promote cell growth. Mol Biol Cell 18: 3545–3555.
Gatenby RA, Gillies RJ . (2004). Why do cancers have high aerobic glycolysis? Nat Rev Cancer 4: 891–899.
Hajduch E, Alessi DR, Hemmings BA, Hundal HS . (1998). Constitutive activation of protein kinase B alpha by membrane targeting promotes glucose and system A amino acid transport, protein synthesis, and inactivation of glycogen synthase kinase 3 in L6 muscle cells. Diabetes 47: 1006–1013.
Huang C, Thirone AC, Huang X, Klip A . (2005). Differential contribution of insulin receptor substrates 1 versus 2 to insulin signaling and glucose uptake in l6 myotubes. J Biol Chem 280: 19426–19435.
Hynes J, O′Riordan TC, Zhdanov AV, Uray G, Will Y, Papkovsky DB . (2009). In vitro analysis of cell metabolism using a long-decay pH-sensitive lanthanide probe and extracellular acidification assay. Anal Biochem 390: 21–28.
Irrcher I, Ljubicic V, Kirwan AF, Hood DA . (2008). AMP-activated protein kinase-regulated activation of the PGC-1alpha promoter in skeletal muscle cells. PLoS ONE 3: e3614.
Ishikawa K, Takenaga K, Akimoto M, Koshikawa N, Yamaguchi A, Imanishi H et al. (2008). ROS-generating mitochondrial DNA mutations can regulate tumor cell metastasis. Science 320: 661–664.
Jager S, Handschin C, St-Pierre J, Spiegelman BM . (2007). AMP-activated protein kinase (AMPK) action in skeletal muscle via direct phosphorylation of PGC-1alpha. Proc Natl Acad Sci USA 104: 12017–12022.
Jazayeri M, Andreyev A, Will Y, Ward M, Anderson CM, Clevenger W . (2003). Inducible expression of a dominant negative DNA polymerase-gamma depletes mitochondrial DNA and produces a rho0 phenotype. J Biol Chem 278: 9823–9830.
Jeng JY, Yeh TS, Lee JW, Lin SH, Fong TH, Hsieh RH . (2008). Maintenance of mitochondrial DNA copy number and expression are essential for preservation of mitochondrial function and cell growth. J Cell Biochem 103: 347–357.
Jones RG, Plas DR, Kubek S, Buzzai M, Mu J, Xu Y et al. (2005). AMP-activated protein kinase induces a p53-dependent metabolic checkpoint. Mol Cell 18: 283–293.
Jones RG, Thompson CB . (2009). Tumor suppressors and cell metabolism: a recipe for cancer growth. Genes Dev 23: 537–548.
Klaunig JE, Xu Y, Isenberg JS, Bachowski S, Kolaja KL, Jiang J et al. (1998). The role of oxidative stress in chemical carcinogenesis. Environ Health Perspect 106 (Suppl 1): 289–295.
Korhonen JA, Gaspari M, Falkenberg M . (2003). TWINKLE Has 5′->3′ DNA helicase activity and is specifically stimulated by mitochondrial single-stranded DNA-binding protein. J Biol Chem 278: 48627–48632.
Lage R, Dieguez C, Vidal-Puig A, Lopez M . (2008). AMPK: a metabolic gauge regulating whole-body energy homeostasis. Trends Mol Med 14: 539–549.
Marroquin LD, Hynes J, Dykens JA, Jamieson JD, Will Y . (2007). Circumventing the Crabtree effect: replacing media glucose with galactose increases susceptibility of HepG2 cells to mitochondrial toxicants. Toxicol Sci 97: 539–547.
Marshall S . (2006). Role of insulin, adipocyte hormones, and nutrient-sensing pathways in regulating fuel metabolism and energy homeostasis: a nutritional perspective of diabetes, obesity, and cancer. Sci STKE 2006: re7.
Miller SA, Dykes DD, Polesky HF . (1988). A simple salting out procedure for extracting DNA from human nucleated cells. Nucleic Acids Res 16: 1215.
Mori K, Shibanuma M, Nose K . (2004). Invasive potential induced under long-term oxidative stress in mammary epithelial cells. Cancer Res 64: 7464–7472.
Naito A, Cook CC, Mizumachi T, Wang M, Xie CH, Evans TT et al. (2008). Progressive tumor features accompany epithelial–mesenchymal transition induced in mitochondrial DNA-depleted cells. Cancer Sci 99: 1584–1588.
Owusu-Ansah E, Yavari A, Mandal S, Banerjee U . (2008). Distinct mitochondrial retrograde signals control the G1–S cell cycle checkpoint. Nat Genet 40: 356–361.
Pankratz SL, Tan EY, Fine Y, Mercurio AM, Shaw LM . (2009). Insulin receptor substrate-2 regulates aerobic glycolysis in mouse mammary tumor cells via glucose transporter 1. J Biol Chem 284: 2031–2037.
Pedersen PL . (1978). Tumor mitochondria and the bioenergetics of cancer cells. Prog Exp Tumor Res 22: 190–274.
Penta JS, Johnson FM, Wachsman JT, Copeland WC . (2001). Mitochondrial DNA in human malignancy. Mutat Res 488: 119–133.
Perez R, Garcia-Fernandez M, Diaz-Sanchez M, Puche JE, Delgado G, Conchillo M et al. (2008). Mitochondrial protection by low doses of insulin-like growth factor-I in experimental cirrhosis. World J Gastroenterol 14: 2731–2739.
Pollak M . (2008). Insulin and insulin-like growth factor signalling in neoplasia. Nat Rev Cancer 8: 915–928.
Quon MJ, Chen H, Ing BL, Liu ML, Zarnowski MJ, Yonezawa K et al. (1995). Roles of 1-phosphatidylinositol 3-kinase and ras in regulating translocation of GLUT4 in transfected rat adipose cells. Mol Cell Biol 15: 5403–5411.
Rhyu DY, Yang Y, Ha H, Lee GT, Song JS, Uh ST et al. (2005). Role of reactive oxygen species in TGF-beta1-induced mitogen-activated protein kinase activation and epithelial–mesenchymal transition in renal tubular epithelial cells. J Am Soc Nephrol 16: 667–675.
Terada S, Goto M, Kato M, Kawanaka K, Shimokawa T, Tabata I . (2002). Effects of low-intensity prolonged exercise on PGC-1 mRNA expression in rat epitrochlearis muscle. Biochem Biophys Res Commun 296: 350–354.
Unterluggauer H, Hutter E, Viertler HP, Jansen-Durr P . (2008). Insulin-like growth factor-induced signals activate mitochondrial respiration. Biotechnol J 3: 813–816.
Van Dyck E, Jank B, Ragnini A, Schweyen RJ, Duyckaerts C, Sluse F et al. (1995). Overexpression of a novel member of the mitochondrial carrier family rescues defects in both DNA and RNA metabolism in yeast mitochondria. Mol Gen Genet 246: 426–436.
Warburg O . (1956). On the origin of cancer cells. Science 123: 309–314.
Will Y, Hynes J, Ogurtsov VI, Papkovsky DB . (2006). Analysis of mitochondrial function using phosphorescent oxygen-sensitive probes. Nat Protoc 1: 2563–2572.
Witczak CA, Sharoff CG, Goodyear LJ . (2008). AMP-activated protein kinase in skeletal muscle: from structure and localization to its role as a master regulator of cellular metabolism. Cell Mol Life Sci 65: 3737–3755.
Wu WS . (2006). The signaling mechanism of ROS in tumor progression. Cancer Metastasis Rev 25: 695–705.
Zhdanov AV, Ward MW, Prehn JH, Papkovsky DB . (2008). Dynamics of intracellular oxygen in PC12 cells upon stimulation of neurotransmission. J Biol Chem 283: 5650–5661.
Acknowledgements
We thank Kurt Tidmore for assistance with illustrations and to our colleagues in the Cell Biology Laboratory for helpful discussions. This work was funded by Science Foundation Ireland and the Health Research Board.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on the Oncogene website
Supplementary information
Rights and permissions
About this article
Cite this article
Favre, C., Zhdanov, A., Leahy, M. et al. Mitochondrial pyrimidine nucleotide carrier (PNC1) regulates mitochondrial biogenesis and the invasive phenotype of cancer cells. Oncogene 29, 3964–3976 (2010). https://doi.org/10.1038/onc.2010.146
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2010.146
Keywords
This article is cited by
-
Novel genetic variants associated with brain functional networks in 18,445 adults from the UK Biobank
Scientific Reports (2021)
-
Cellular pyrimidine imbalance triggers mitochondrial DNA–dependent innate immunity
Nature Metabolism (2021)
-
Insulin-like growth factor receptor signaling in breast tumor epithelium protects cells from endoplasmic reticulum stress and regulates the tumor microenvironment
Breast Cancer Research (2018)
-
Control of mitochondrial function and cell growth by the atypical cadherin Fat1
Nature (2016)
-
TUFM downregulation induces epithelial–mesenchymal transition and invasion in lung cancer cells via a mechanism involving AMPK-GSK3β signaling
Cellular and Molecular Life Sciences (2016)