Abstract
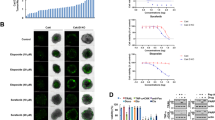
Proteasome inhibitors (PIs) are promising new therapeutic agents for treating non-small cell lung carcinoma (NSCLC). To investigate the mechanisms of action of PIs, we analyzed the proapoptotic activities of PIs (MG132 or Bortezomib) in NSCLC cells. We found that both MG132 (>1 μM) and Bortezomib (>0.025 μM) induced a significant apoptosis in NCI-H1703, a PI-sensitive NSCLC cell line, through initially activating the intrinsic apoptosis pathway, leading to the activation of a positive feedback mechanism (PFM), which then conveyed apoptosis signaling from the intrinsic pathway to the extrinsic pathway with formation of a signaling loop for maximal caspase activation. Mcl-1 and Noxa were identified to be the major anti-apoptotic and proapoptotic proteins, respectively, in PI-induced apoptosis and mutually exclusive in protein stability. Although the Mcl-1 protein was upregulated by proteasome inhibition, it was also subjected to caspase 3-dependent cleavage governed by the PFM. Moreover, it was revealed that Mcl-1 protein cleavage contributed to PFM-governed apoptosis in following inter-related ways: reducing the anti-apoptotic Mcl-1; generating the truncated proapoptotic Mcl-1S; and inducing a shift of balance between Mcl-1 and Noxa. It was further manifested that tumor necrosis factor-related apoptosis-inducing ligand boosted MG132's proapoptotic activity through strengthening the PFM in both NCI-H1703 and NCI-H358, a PI-resistant NSCLC cell line. Therefore, this study provides a basis for enhancing the efficacy of PIs in treating NSCLC.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout









Similar content being viewed by others
References
Adams J . (2004). The development of proteasome inhibitors as anticancer drugs. Cancer Cell 5: 417–421.
Certo M, Del Gaizo Moore V, Nishino M, Wei G, Korsmeyer S, Armstrong SA et al. (2006). Mitochondria primed by death signals determine cellular addiction to antiapoptotic BCL-2 family members. Cancer Cell 9: 351–365.
Cregan SP, Dawson VL, Slack RS . (2004). Role of AIF in caspase-dependent and caspase-independent cell death. Oncogene 23: 2785–2796.
Cryns V, Yuan J . (1998). Proteases to die for. Genes Dev 12: 1551–1570.
Cuconati A, Mukherjee C, Perez D, White E . (2003). DNA damage response and MCL-1 destruction initiate apoptosis in adenovirus-infected cells. Genes Dev 17: 2922–2932.
Das T, Sa G, Sinha P, Ray PK . (1999). Induction of cell proliferation and apoptosis: dependence on the dose of the inducer. Biochem Biophys Res Commun 260: 105–110.
Davies AM, Lara Jr PN, Mack PC, Gandara DR . (2007). Incorporating bortezomib into the treatment of lung cancer. Clin Cancer Res 13: s4647–s4651.
Fiskum G, Craig SW, Decker GL, Lehninger AL . (1980). The cytoskeleton of digitonin-treated rat hepatocytes. Proc Natl Acad Sci USA 77: 3430–3434.
Green DR . (2006). At the gates of death. Cancer Cell 9: 328–330.
Hacker G . (2000). The morphology of apoptosis. Cell Tissue Res 301: 5–17.
Hassan M, Alaoui A, Feyen O, Mirmohammadsadegh A, Essmann F, Tannapfel A et al. (2008). The BH3-only member Noxa causes apoptosis in melanoma cells by multiple pathways. Oncogene 27: 4557–4568.
Henderson CJ, Aleo E, Fontanini A, Maestro R, Paroni G, Brancolini C . (2005). Caspase activation and apoptosis in response to proteasome inhibitors. Cell Death Differ 12: 1240–1254.
Hunter AM, LaCasse EC, Korneluk RG . (2007). The inhibitors of apoptosis (IAPs) as cancer targets. Apoptosis 12: 1543–1568.
Jemal A, Murray T, Ward E, Samuels A, Tiwari RC, Ghafoor A et al. (2005). Cancer statistics, 2005. CA Cancer J Clin 55: 10–30.
Jing N, Tweardy DJ . (2005). Targeting Stat3 in cancer therapy. Anticancer Drugs 16: 601–607.
Kruyt FA . (2008). TRAIL and cancer therapy. Cancer Lett 263: 14–25.
Letai A, Bassik MC, Walensky LD, Sorcinelli MD, Weiler S, Korsmeyer SJ . (2002). Distinct BH3 domains either sensitize or activate mitochondrial apoptosis, serving as prototype cancer therapeutics. Cancer Cell 2: 183–192.
Liu X, Yue P, Schonthal AH, Khuri FR, Sun SY . (2006). Cellular FLICE-inhibitory protein down-regulation contributes to celecoxib-induced apoptosis in human lung cancer cells. Cancer Res 66: 11115–11119.
MacFarlane M, Merrison W, Bratton SB, Cohen GM . (2002). Proteasome-mediated degradation of Smac during apoptosis: XIAP promotes Smac ubiquitination in vitro. J Biol Chem 277: 36611–36616.
Milhas D, Cuvillier O, Therville N, Clave P, Thomsen M, Levade T et al. (2005). Caspase-10 triggers Bid cleavage and caspase cascade activation in FasL-induced apoptosis. J Biol Chem 280: 19836–19842.
Molina JR, Yang P, Cassivi SD, Schild SE, Adjei AA . (2008). Non-small cell lung cancer: epidemiology, risk factors, treatment, and survivorship. Mayo Clin Proc 83: 584–594.
Muzio M, Chinnaiyan AM, Kischkel FC, O’Rourke K, Shevchenko A, Ni J et al. (1996). FLICE, a novel FADD-homologous ICE/CED-3-like protease, is recruited to the CD95 (Fas/APO-1) death-inducing signaling complex. Cell 85: 817–827.
Nagy K, Szekely-Szuts K, Izeradjene K, Douglas L, Tillman M, Barti-Juhasz H et al. (2006). Proteasome inhibitors sensitize colon carcinoma cells to TRAIL-induced apoptosis via enhanced release of Smac/DIABLO from the mitochondria. Pathol Oncol Res 12: 133–142.
Rogakou EP, Pilch DR, Orr AH, Ivanova VS, Bonner WM . (1998). DNA double-stranded breaks induce histone H2AX phosphorylation on serine 139. J Biol Chem 273: 5858–5868.
Saulle E, Petronelli A, Pasquini L, Petrucci E, Mariani G, Biffoni M et al. (2007). Proteasome inhibitors sensitize ovarian cancer cells to TRAIL-induced apoptosis. Apoptosis 12: 635–655.
Steenland K . (1996). Epidemiology of occupation and coronary heart disease: research agenda. Am J Ind Med 30: 495–499.
Strasser A . (2005). The role of BH3-only proteins in the immune system. Nat Rev Immunol 5: 189–200.
Voortman J, Checinska A, Giaccone G, Rodriguez JA, Kruyt FA . (2007). Bortezomib, but not cisplatin, induces mitochondria-dependent apoptosis accompanied by up-regulation of noxa in the non-small cell lung cancer cell line NCI-H460. Mol Cancer Ther 6: 1046–1053.
Wang S, El-Deiry WS . (2003). TRAIL and apoptosis induction by TNF-family death receptors. Oncogene 22: 8628–8633.
Weng C, Li Y, Xu D, Shi Y, Tang H . (2005). Specific cleavage of Mcl-1 by caspase-3 in tumor necrosis factor-related apoptosis-inducing ligand (TRAIL)-induced apoptosis in Jurkat leukemia T cells. J Biol Chem 280: 10491–10500.
Willis SN, Chen L, Dewson G, Wei A, Naik E, Fletcher JI et al. (2005). Proapoptotic Bak is sequestered by Mcl-1 and Bcl-xL, but not Bcl-2, until displaced by BH3-only proteins. Genes Dev 19: 1294–1305.
Yuan B-Z, Chapman JA, Reynolds SH . (2008). Proteasome inhibitor MG132 induces apoptosis and inhibits invasion of human malignant pleural mesothelioma cells. Transl Oncol 1: 129–140.
Yuan BZ, Jefferson AM, Millecchia L, Popescu NC, Reynolds SH . (2007). Morphological changes and nuclear translocation of DLC1 tumor suppressor protein precede apoptosis in human non-small cell lung carcinoma cells. Exp Cell Res 313: 3868–3880.
Zhang HG, Wang J, Yang X, Hsu HC, Mountz JD . (2004). Regulation of apoptosis proteins in cancer cells by ubiquitin. Oncogene 23: 2009–2015.
Acknowledgements
We thank Dr Robert Lanciotti (National Institute for Occupational Safety and Health) for his assistance with the flow cytometry assay.
Author information
Authors and Affiliations
Corresponding author
Additional information
Disclaimer
The findings and conclusions in this report are those of the authors and do not necessarily represent the views of the National Institute for Occupational Safety and Health of the Centers for Disease Control and Prevention.
Supplementary Information accompanies the paper on the Oncogene website (http://www.nature.com/onc)
Rights and permissions
About this article
Cite this article
Yuan, BZ., Chapman, J. & Reynolds, S. Proteasome inhibitors induce apoptosis in human lung cancer cells through a positive feedback mechanism and the subsequent Mcl-1 protein cleavage. Oncogene 28, 3775–3786 (2009). https://doi.org/10.1038/onc.2009.240
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2009.240
Keywords
This article is cited by
-
Cullin-5 neddylation-mediated NOXA degradation is enhanced by PRDX1 oligomers in colorectal cancer
Cell Death & Disease (2021)
-
The DLC-1 tumor suppressor is involved in regulating immunomodulation of human mesenchymal stromal /stem cells through interacting with the Notch1 protein
BMC Cancer (2020)
-
The ubiquitin system: orchestrating cellular signals in non-small-cell lung cancer
Cellular & Molecular Biology Letters (2020)
-
TRAIL and proteasome inhibitors combination induces a robust apoptosis in human malignant pleural mesothelioma cells through Mcl-1 and Akt protein cleavages
BMC Cancer (2013)
-
Small interfering RNA targeting mcl-1 enhances proteasome inhibitor-induced apoptosis in various solid malignant tumors
BMC Cancer (2011)