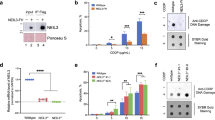
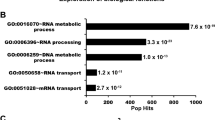
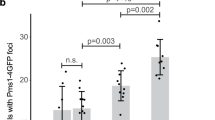
Abstract
O6-Methylguanine produced in DNA induces mutation due to its ambiguous base-pairing properties during DNA replication. To suppress such an outcome, organisms possess a mechanism to eliminate cells carrying O6-methylguanine by inducing apoptosis that requires the function of mismatch repair proteins. To identify other factors involved in this apoptotic process, we performed retrovirus-mediated gene-trap mutagenesis and isolated a mutant that acquired resistance to a simple alkylating agent, N-methyl-N-nitrosourea (MNU). However, it was still sensitive to methyl methanesulfonate, 1-(4-amino-2-methyl-5-pyrimidinyl)methyl-3-(2-chloroethyl)-3-nitrosourea, etoposide and ultraviolet irradiation. Moreover, the mutant exhibited an increased mutant frequency after exposure to MNU. The gene responsible was identified and designated Mapo1 (O6-methylguanine-induced apoptosis 1). When the expression of the gene was inhibited by small interfering RNA, MNU-induced apoptosis was significantly suppressed. In the Mapo1-defective mutant cells treated with MNU, the mitochondrial membrane depolarization and caspase-3 activation were severely suppressed, although phosphorylation of p53, CHK1 and histone H2AX was observed. The orthologs of the Mapo1 gene are present in various organisms from nematode to humans. Both mouse and human MAPO1 proteins expressed in cells localize in the cytoplasm. We therefore propose that MAPO1 may play a role in the signal-transduction pathway of apoptosis induced by O6-methylguanine-mispaired lesions.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Beranek DT . (1990). Distribution of methyl and ethyl adducts following alkylation with monofunctional alkylating agents. Mutat Res 231: 11–30.
Branch P, Aquilina G, Bignami M, Karran P . (1993). Defective mismatch binding and a mutator phenotype in cells tolerant to DNA damage. Nature 362: 652–654.
Caporali S, Falcinelli S, Starace G, Russo MT, Bonmassar E, Jiricny J et al. (2004). DNA damage induced by temozolomide signals to both ATM and ATR: role of the mismatch repair system. Mol Pharmacol 66: 478–491.
Cejka P, Stojic L, Mojas N, Russell AM, Heinimann K, Cannavo E et al. (2003). Methylation-induced G(2)/M arrest requires a full complement of the mismatch repair protein hMLH1. EMBO J 22: 2245–2254.
Christmann M, Kaina B . (2000). Nuclear translocation of mismatch repair proteins MSH2 and MSH6 as a response of cells to alkylating agents. J Biol Chem 275: 36256–36262.
Cossarizza A, Kalashnikova G, Grassilli E, Chiappelli F, Salvioli S, Capri M et al. (1994). Mitochondrial modifications during rat thymocyte apoptosis: a study at the single cell level. Exp Cell Res 214: 323–330.
Coulondre C, Miller JH . (1977). Genetic studies of the lac repressor. IV. Mutagenic specificity in the lacI gene of Escherichia coli. J Mol Biol 117: 577–606.
Fishel R . (1998). Mismatch repair, molecular switches, and signal transduction. Genes Dev 12: 2096–2101.
Glaab WE, Risinger JI, Umar A, Barrett JC, Kunkel TA, Tindall KR . (1998). Cellular resistance and hypermutability in mismatch repair-deficient human cancer cell lines following treatment with methyl methanesulfonate. Mutat Res 398: 197–207.
Glassner BJ, Weeda G, Allan JM, Broekhof JL, Carls NH, Donker I et al. (1999). DNA repair methyltransferase (Mgmt) knockout mice are sensitive to the lethal effects of chemotherapeutic alkylating agents. Mutagenesis 14: 339–347.
Guo G, Wang W, Bradley A . (2004). Mismatch repair genes identified using genetic screens in Blm-deficient embryonic stem cells. Nature 429: 891–985.
Haracska L, Prakash S, Prakash L . (2000). Replication past O6-methylguanine by yeast and human DNA polymerase eta. Mol Cell Biol 20: 8001–8007.
Hickman MJ, Samson LD . (1999). Role of DNA mismatch repair and p53 in signaling induction of apoptosis by alkylating agents. Proc Natl Acad Sci USA 96: 10764–10769.
Hickman MJ, Samson LD . (2004). Apoptotic signaling in response to a single type of DNA lesion, O6-methylguanine. Mol Cell 14: 105–116.
Hidaka M, Takagi Y, Takano TY, Sekiguchi M . (2005). PCNA-MutSalpha-mediated binding of MutLalpha to replicative DNA with mismatched bases to induce apoptosis in human cells. Nucleic Acids Res 33: 5703–5712.
Ishida Y, Leder P . (1999). RET: a poly A-trap retrovirus vector for reversible disruption and expression monitoring of genes in living cells. Nucleic Acids Res 27: e35.
Ito T, Nakamura T, Maki H, Sekiguchi M . (1994). Roles of transcription and repair in alkylation mutagenesis. Mutat Res 314: 273–285.
Kaina B, Christmann M, Naumann S, Roos WP . (2007). MGMT: key node in the battle against genotoxicity, carcinogenicity and apoptosis induced by alkylating agents. DNA Repair (Amst) 6: 1079–1099.
Kaina B, Fritz G, Mitra S, Coquerelle T . (1991). Transfection and expression of human O6-methylguanine-DNA methyltransferase (MGMT) cDNA in Chinese hamster cells: the role of MGMT in protection against the genotoxic effects of alkylating agents. Carcinogenesis 12: 1857–1867.
Karran P . (2001). Mechanisms of tolerance to DNA damaging therapeutic drugs. Carcinogenesis 22: 1931–1937.
Kat A, Thilly WG, Fang WH, Longley MJ, Li GM, Modrich P . (1993). An alkylation-tolerant, mutator human cell line is deficient in strand-specific mismatch repair. Proc Natl Acad Sci USA 90: 6424–6428.
Kawate H, Itoh R, Sakumi K, Nakabeppu Y, Tsuzuki T, Ide F et al. (2000). A defect in a single allele of the Mlh1 gene causes dissociation of the killing and tumorigenic actions of an alkylating carcinogen in methyltransferase-deficient mice. Carcinogenesis 21: 301–305.
Kawate H, Sakumi K, Tsuzuki T, Nakatsuru Y, Ishikawa T, Takahashi S et al. (1998). Separation of killing and tumorigenic effects of an alkylating agent in mice defective in two of the DNA repair genes. Proc Natl Acad Sci USA 95: 5116–5120.
Li G, Modrich P . (1995). Restoration of mismatch repair to nuclear extracts of H6 colorectal tumor cells by a heterodimer of human MutL homologs. Proc Natl Acad Sci USA 92: 1950–1954.
Loechler EL, Green CL, Essigmann JM . (1984). In vivo mutagenesis by O6-methylguanine built into a unique site in a viral genome. Proc Natl Acad Sci USA 81: 6271–6275.
Margison GP, Santibanez-Koref MF . (2002). O6-alkylguanine-DNA alkyltransferase: role in carcinogenesis and chemotherapy. Bioessays 24: 255–266.
Meikrantz W, Bergom MA, Memisoglu A, Samson L . (1998). O6-alkylguanine DNA lesions trigger apoptosis. Carcinogenesis 19: 369–372.
Ochs K, Kaina B . (2000). Apoptosis induced by DNA damage O6-methylguanine is Bcl-2 and caspase-9/3 regulated and Fas/caspase-8 independent. Cancer Res 60: 5815–5824.
Pegg AE . (2000). Repair of O(6)-alkylguanine by alkyltransferases. Mutat Res 462: 83–100.
Peng M, Litman R, Xie J, Sharma S, Brosh Jr RM, Cantor SB . (2007). The FANCJ/MutLalpha interaction is required for correction of the cross-link response in FA-J cells. EMBO J 26: 3238–3249.
Pepponi R, Marra G, Fuggetta MP, Falcinelli S, Pagani E, Bonmassar E et al. (2003). The effect of O6-alkylguanine-DNA alkyltransferase and mismatch repair activities on the sensitivity of human melanoma cells to temozolomide, 1,3-bis(2-chloroethyl)1-nitrosourea, and cisplatin. J Pharmacol Exp Ther 304: 661–668.
Sakumi K, Shiraishi A, Shimizu S, Tsuzuki T, Ishikawa T, Sekiguchi M . (1997). Methylnitrosourea-induced tumorigenesis in MGMT gene knockout mice. Cancer Res 57: 2415–2418.
Sanada M, Hidaka M, Takagi Y, Takano TY, Nakatsu Y, Tsuzuki T et al. (2007). Modes of actions of two types of anti-neoplastic drugs, dacarbazine and ACNU, to induce apoptosis. Carcinogenesis 28: 2657–2663.
Singer B, Chavez F, Goodman MF, Essigmann JM, Dosanjh MK . (1989). Effect of 3′ flanking neighbors on kinetics of pairing of dCTP or dTTP opposite O6-methylguanine in a defined primed oligonucleotide when Escherichia coli DNA polymerase I is used. Proc Natl Acad Sci USA 86: 8271–8274.
Stojic L, Mojas N, Cejka P, Di Pietro M, Ferrari S, Marra G et al. (2004). Mismatch repair-dependent G2 checkpoint induced by low doses of SN1 type methylating agents requires the ATR kinase. Genes Dev 18: 1331–1344.
Takagi Y, Hidaka M, Sanada M, Yoshida H, Sekiguchi M . (2008). Different initial steps of apoptosis induced by two types of antineoplastic drugs. Biochem Pharmacol 76: 303–311.
Takagi Y, Takahashi M, Sanada M, Ito R, Yamaizumi M, Sekiguchi M . (2003). Roles of MGMT and MLH1 proteins in alkylation-induced apoptosis and mutagenesis. DNA Repair (Amst) 2: 1135–1146.
Tominaga Y, Tsuzuki T, Shiraishi A, Kawate H, Sekiguchi M . (1997). Alkylation-induced apoptosis of embryonic stem cells in which the gene for DNA-repair, methyltransferase, had been disrupted by gene targeting. Carcinogenesis 18: 889–896.
Tsuzuki T, Sakumi K, Shiraishi A, Kawate H, Igarashi H, Iwakuma T et al. (1996). Targeted disruption of the DNA repair methyltransferase gene renders mice hypersensitive to alkylating agent. Carcinogenesis 17: 1215–1220.
Wiles MV, Vauti F, Otte J, Fuchtbauer EM, Ruiz P, Fuchtbauer A et al. (2000). Establishment of a gene-trap sequence tag library to generate mutant mice from embryonic stem cells. Nat Genet 24: 13–14.
Yoshioka K, Yoshioka Y, Hsieh P . (2006). ATR kinase activation mediated by MutSalpha and MutLalpha in response to cytotoxic O6-methylguanine adducts. Mol Cell 22: 501–510.
Acknowledgements
We thank the Kazusa DNA Research Institute for providing hKIAA1450 cDNA. This study was supported by grants (including a Frontier Research Grant) from the Ministry of Education, Culture, Sports, Science and Technology of Japan.
Author information
Authors and Affiliations
Corresponding author
Additional information
Conflict of interest
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Komori, K., Takagi, Y., Sanada, M. et al. A novel protein, MAPO1, that functions in apoptosis triggered by O6-methylguanine mispair in DNA. Oncogene 28, 1142–1150 (2009). https://doi.org/10.1038/onc.2008.462
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2008.462
Keywords
This article is cited by
-
Function of high-mobility group A proteins in the DNA damage signaling for the induction of apoptosis
Scientific Reports (2016)
-
Molecular genetics and clinical features of Birt–Hogg–Dubé syndrome
Nature Reviews Urology (2015)