Abstract
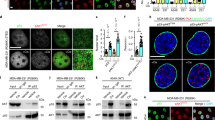
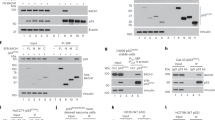
Although p73α induces many of the same cellular events as p53, it is structurally distinct from p53 in that it possesses a unique COOH-terminal domain. To dissect the function of this domain, we performed yeast two-hybrid screening of a HeLa cDNA library using residues 552–636 of p73α as bait. Among the clones that showed a specific interaction with p73α was AMP-activated protein kinase α (AMPKα). Additional yeast two-hybrid assays indicated that the βγ-binding domain of AMPKα is critical for the interaction with p73α. The interaction was further confirmed in vitro by glutathione S-transferase pull-down, and in vivo by immunoprecipitation and immunofluorescence microscopy. Transient coexpression of AMPKα resulted in downregulation of the effect of p73α, but not of p53, on various p53-responsive promoters. Chromatin immunoprecipitation indicated p73α-dependent recruitment of AMPKα to the p21WAF1 promoter. Treatment with 5-aminoimidazole-4-carboxamide ribonucleotide, an agonist of AMPKα, and expression of dominant-negative versions of AMPKα revealed that the repression of p73α was independent of AMPKα kinase activity. In addition, cisplatin-induced growth repression was impaired when AMPKα was overexpressed. Upon the knock down of AMPKα by siRNA, the induction of p21WAF1 by p73α was significantly increased. Taken together, these data indicate that AMPKα specifically regulates p73α by a direct interaction without affecting its phosphorylation status. From these data, we speculate that AMPKα may provide a molecular clue to understand the repressive role of the C-terminus of p73α in transcription and DNA damage response.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Abbreviations
- AMPK:
-
AMP-activated protein kinase
- AICAR:
-
5-aminoimidazole-4-carboxamide ribonucleotide
- DBD:
-
DNA-binding domain
- GST:
-
glutathione S-transferase
- ChIP:
-
chromatin immunoprecipitation
- GFP:
-
green fluorescent protein
- β-gal:
-
β-galactosidase
- MTT:
-
3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazonium) bromide
References
Bentley D . (1999). Coupling RNA polymerase II transcription with pre-mRNA processing. Curr Opin Cell Biol 11: 347–351.
Bronner M, Hertz R, Bar-Tana J . (2004). Kinase-independent transcriptional co-activation of peroxisome proliferator-activated receptor alpha by AMP-activated protein kinase. Biochem J 384: 295–305.
Chen H, Montagnani M, Funahashi T, Shimomura I, Quon MJ . (2003). Adiponectin stimulates production of nitric oxide in vascular endothelial cells. J Biol Chem 278: 45021–45026.
Cho YS, Kim EJ, Park UH, Sin HS, Um SJ . (2006). Additional sex comb-like 1 (ASXL1), in cooperation with SRC-1, acts as a ligand-dependent coactivator for retinoic acid receptor. J Biol Chem 281: 17588–17598.
Culmsee C, Monnig J, Kemp BE, Mattson MP . (2001). AMP-activated protein kinase is highly expressed in neurons in the developing rat brain and promotes neuronal survival following glucose deprivation. J Mol Neurosci 17: 45–58.
Da Silva Xavier G, Leclerc I, Salt IP, Doiron B, Hardie DG, Kahn A et al. (2000). Role of AMP-activated protein kinase in the regulation by glucose of islet beta cell gene expression. Proc Natl Acad Sci USA 97: 4023–4028.
Dai JM, Wang ZY, Sun DC, Lin RX, Wang SQ . (2007). SIRT1 interacts with p73 and suppresses p73-dependent transcriptional activity. J Cell Physiol 210: 161–166.
Di Como CJ, Gaiddon C, Prives C . (1999). p73 function is inhibited by tumor-derived p53 mutants in mammalian cells. Mol Cell Biol 19: 1438–1449.
Dietz S, Rother K, Bamberger C, Schmale H, Mossner J, Engeland K . (2002). Differential regulation of transcription and induction of programmed cell death by human p53-family members p63 and p73. FEBS Lett 525: 93–99.
Hardie DG, Carling D, Carlson M . (1998). The AMP-activated/SNF1 protein kinase subfamily: metabolic sensors of the eukaryotic cell? Annu Rev Biochem 67: 821–855.
Hardie DG, Salt IP, Hawley SA, Davies SP . (1999). AMP-activated proteinkinase: an ultrasensitive system for monitoring cellular energy charge. Biochem J 338: 717–722.
Hawley SA, Pan DA, Mustard KJ, Ross L, Bain J, Edelman AM et al. (2005). Ca2+/calmodulin-dependent protein kinase kinase-beta acts upstream of AMP-activated protein kinase in mammalian cells. Cell Metab 2: 9–19.
Hurley RL, Anderson KA, Franzone JM, Kemp BE, Means AR, Witters LA . (2005). Ca2+/calmodulin-dependent protein kinase kinases are AMP-activated protein kinase kinases. J Biol Chem 280: 29060–29066.
Imamura K, Ogura T, Kishimoto A, Kaminishi M, Esumi H . (2001). Cell cycle regulation via p53 phosphorylation by a 5′-AMP activated protein kinase activator, 5-aminoimidazole-4-carboxamide-1-beta-D-ribofuranoside, in a human hepatocellular carcinoma cell line. Biochem Biophys Res Commun 287: 562–567.
Jones RG, Plas DR, Kubek S, Buzzai M, Mu J, Xu Y et al. (2005). AMP-activated protein kinase induces a p53-dependent metabolic checkpoint. Mol Cell 18: 283–293.
Jost CA, Marin MC, Kaelin WG . (1997). p73 is a simian [correction of human] p53-related protein that can induce apoptosis. Nature 389: 191–194.
Kaghad M, Bonnet H, Yang A, Creancier L, Biscan JC, Valent A et al. (1997). Monoallelically expressed gene related to p53 at 1p36, a region frequently deleted in neuroblastoma and other human cancers. Cell 90: 809–819.
Kemp BE, Mitchelhill KI, Stapleton D, Michell BJ, Chen ZP, Witters LA . (1999). Dealing with energy demand: the AMP-activated protein kinase. Trends Biochem Sci 24: 22–25.
Kemp BE, Stapleton D, Campbell DJ, Chen ZP, Murthy S, Walter M et al. (2003). AMP-activated protein kinase, super metabolic regulator. Biochem Soc Trans 31: 162–168.
Kim EJ, Park JS, Um SJ . (2002). Identification and characterization of HIPK2 interacting with p73 and modulating functions of the p53 family in vivo. J Biol Chem 277: 32020–32028.
Lau LM, Nugent JK, Zhao X, Irwin MS . (2008). HDM2 antagonist Nutlin-3 disrupts p73-HDM2 binding and enhances p73 function. Oncogene 27: 997–1003.
Leclerc I, Viollet B, da Silva Xavier G, Kahn A, Rutter GA . (2002). Role of AMP-activated protein kinase in the regulation of gene transcription. Biochem Soc Trans 30: 307–311.
Lee HK, Park UH, Kim EJ, Um SJ . (2007). MED25 is distinct from TRAP220/MED1 in cooperating with CBP for retinoid receptor activation. EMBO J 26: 3545–3557.
Lee M, Hwang JT, Lee HJ, Jung SN, Kang I, Chi SG et al. (2003). AMP-activated protein kinase activity is critical for hypoxia-inducible factor-1 transcriptional activity and its target gene expression under hypoxic conditions in DU145 cells. J Biol Chem 278: 39653–39661.
Leff T . (2003). AMP-activated protein kinase regulates gene expression by direct phosphorylation of nuclear proteins. Biochem Soc Trans 31: 224–227.
Liu G, Chen X . (2005). The C-terminal sterile alpha motif and the extreme C terminus regulate the transcriptional activity of the alpha isoform of p73. J Biol Chem 280: 20111–20119.
Lizcano JM, Goransson O, Toth R, Deak M, Morrice NA, Boudeau J et al. (2004). LKB1 is a master kinase that activates 13 kinases of the AMPK subfamily, including MARK/PAR-1. EMBO J 23: 833–843.
Lo WS, Duggan L, Emre NC, Belotserkovskya R, Lane WS, Shiekhattar R et al. (2001). Snf1–a histone kinase that works in concert with the histone acetyltransferase Gcn5 to regulate transcription. Science 293: 1142–1146.
McGrath JA, Duijf PH, Doetsch V, Irvine AD, de Waal R, Vanmolkot KR et al. (2001). Hay-Wells syndrome is caused by heterozygous missense mutations in the SAM domain of p63. Hum Mol Genet 10: 221–229.
Osada M, Ohba M, Kawahara C, Ishioka C, Kanamaru R, Katoh I et al. (1998). Cloning and functional analysis of human p51, which structurally and functionally resembles p53. Nat Med 4: 839–843.
Ozaki T, Naka M, Takada N, Tada M, Sakiyama S, Nakagawara A . (1999). Deletion of the COOH-terminal region of p73alpha enhances both its transactivation function and DNA-binding activity but inhibits induction of apoptosis in mammalian cells. Cancer Res 59: 5902–5907.
Pilon G, Dallaire P, Marette A . (2004). Inhibition of inducible nitric-oxide synthase by activators of AMP-activated protein kinase: a new mechanism of action of insulin-sensitizing drugs. J Biol Chem 279: 20767–20774.
Pozniak CD, Barnabe-Heider F, Rymar VV, Lee AF, Sadikot AF, Miller FD . (2002). p73 is required for survival and maintenance of CNS neurons. J Neurosci 22: 9800–9809.
Pozniak CD, Radinovic S, Yang A, McKeon F, Kaplan DR, Miller FD . (2000). An anti-apoptotic role for the p53 family member, p73, during developmental neuron death. Science 289: 304–306.
Salt I, Celler JW, Hawley SA, Prescott A, Woods A, Carling D et al. (1998). AMP-activated protein kinase: greater AMP dependence, and preferential nuclear localization, of complexes containing the alpha2 isoform. Biochem J 334: 177–187.
Sbisà E, Catalano D, Grillo G, Licciulli F, Turi A, Liuni S et al. (2007). p53FamTaG: a database resource of human p53, p63 and p73 direct target genes combining in silico prediction and microarray data. BMC Bioinformatics 8: s20.
Shaw RJ, Kosmatka M, Bardeesy N, Hurley RL, Witters LA, DePinho RA et al. (2004). The tumor suppressor LKB1 kinase directly activates AMP-activated kinase and regulates apoptosis in response to energy stress. Proc Natl Acad Sci USA 101: 3329–3335.
Solaz-Fuster MC, Gimeno-Alcaniz JV, Casado M, Sanz P . (2006). TRIP6 transcriptional co-activator is a novel substrate of AMP-activated protein kinase. Cell Signal 18: 1702–1712.
Stein SC, Woods A, Jones NA, Davison MD, Carling D . (2000). The regulation of AMP-activated protein kinase by phosphorylation. Biochem J 345: 437–443.
Thanos CD, Bowie JU . (1999). p53 Family members p63 and p73 are SAM domain-containing proteins. Protein Sci 8: 1708–1710.
Turnley AM, Stapleton D, Mann RJ, Witters LA, Kemp BE, Bartlett PF . (1999). Cellular distribution and developmental expression of AMP-activated protein kinase isoforms in mouse central nervous system. J Neurochem 72: 1707–1716.
Wang S, Yan-Neale Y, Zeremski M, Cohen D . (2004). Transcription regulation by histone deacetylases. Novartis Found Symp 259: 238–245.
Weiske J, Huber O . (2006). The histidine triad protein Hint1 triggers apoptosis independent of its enzymatic activity. J Biol Chem 281: 27356–27366.
Woods A, Dickerson K, Heath R, Hong SP, Momcilovic M, Johnstone SR et al. (2005). Calmodulin-dependent protein kinase kinase-beta is an alternative upstream kinase for AMP-activated protein kinase. Cell Metab 2: 21–33.
Woods A, Johnstone SR, Dickerson K, Leiper FC, Fryer LG, Neumann D et al. (2003). LKB1 is the upstream kinase in the AMP-activated protein kinase cascade. Curr Biol 13: 2004–2008.
Yang A, Kaghad M, Wang Y, Gillett E, Fleming MD, Dötsch V et al. (1998). p63, a p53 homolog at 3q27-29, encodes multiple products with transactivating, death-inducing, and dominant-negative activities. Mol Cell 2: 305–316.
Yang A, Walker N, Bronson R, Kaghad M, Oosterwegel M, Bonnin J et al. (2000). p73-deficient mice have neurological, pheromonal and inflammatory defects but lack spontaneous tumours. Nature 404: 99–103.
Acknowledgements
This study was supported by a grant of the National R&D Program for Cancer Control, Ministry of Health and Welfare, Republic of Korea (0420030-3). All authors were supported by BK21 project from Ministry of Education and Human Resources Development.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Supplementary Information accompanies the paper on the Oncogene website (http://www.nature.com/onc)
Rights and permissions
About this article
Cite this article
Lee, YG., Lee, SW., Sin, HS. et al. Kinase activity-independent suppression of p73α by AMP-activated kinase α (AMPKα). Oncogene 28, 1040–1052 (2009). https://doi.org/10.1038/onc.2008.452
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2008.452
Keywords
This article is cited by
-
Context-dependent AMPK activation distinctly regulates TAp73 stability and transcriptional activity
Signal Transduction and Targeted Therapy (2018)
-
AMPK couples p73 with p53 in cell fate decision
Cell Death & Differentiation (2014)
-
Over-expressions of AMPK subunits in ovarian carcinomas with significant clinical implications
BMC Cancer (2012)
-
AMP-activated protein kinase-α1 as an activating kinase of TGF-β-activated kinase 1 has a key role in inflammatory signals
Cell Death & Disease (2012)