Abstract
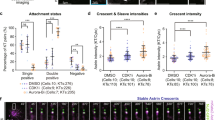
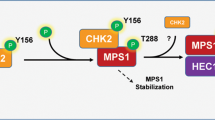
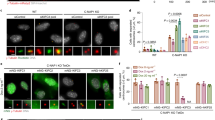
Loss or gain of whole chromosome, the form of chromosome instability commonly associated with cancers is thought to arise from aberrant chromosome segregation during cell division. Chromosome segregation in mitosis is orchestrated by the interaction of kinetochores with spindle microtubules. Our studies show that NEK2A is a kinetochore-associated protein kinase essential for faithful chromosome segregation. However, it was unclear how NEK2A ensures accurate chromosome segregation in mitosis. Here we show that NEK2A-mediated Hec1 (highly expressed in cancer) phosphorylation is essential for faithful kinetochore microtubule attachments in mitosis. Using phospho-specific antibody, our studies show that NEK2A phosphorylates Hec1 at Ser165 during mitosis. Although such phosphorylation is not required for assembly of Hec1 to the kinetochore, expression of non-phosphorylatable mutant Hec1S165 perturbed chromosome congression and resulted in a dramatic increase in microtubule attachment errors, including syntelic and monotelic attachments. Our in vitro reconstitution experiment demonstrated that Hec1 binds to microtubule in low affinity and phosphorylation by NEK2A, which prevents aberrant kinetochore-microtubule connections in vivo, increases the affinity of the Ndc80 complex for microtubules in vitro. Thus, our studies illustrate a novel regulatory mechanism in which NEK2A kinase operates a faithful chromosome attachment to spindle microtubule, which prevents chromosome instability during cell division.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Abbreviations
- Hec1:
-
highly expressed in cancer
References
Ault JG, Nicklas RB . (1989). Tension, microtubule rearrangements, and the proper distribution of chromosomes in mitosis. Chromosoma 98: 33–39.
Ault JG, Rieder CL . (1994). Centrosome and kinetochore movement during mitosis. Curr Opin Cell Biol 6: 41–49.
Cheeseman IM, Chappie JS, Wilson-Kubalek EM, Desai A . (2006). The conserved KMN network constitutes the core microtubule-binding site of the kinetochore. Cell 127: 983–997.
Chen Y, Riley DJ, Zheng L, Chen PL, Lee WH . (2002). Phosphorylation of the mitotic regulator protein Hec1 by Nek2 kinase is essential for faithful chromosome segregation. J Biol Chem 277: 49408–49416.
Ciferri C, De Luca J, Monzani S, Ferrari KJ, Ristic D, Wyman C et al. (2005). Architecture of the human Ndc80-hec1 complex, a critical constituent of the outer kinetochore. J Biol Chem 280: 29088–29095.
Ciferri C, Musacchio A, Petrovic A . (2007). The Ndc80 complex: hub of kinetochore activity. FEBS Lett 581: 2862–2869.
Cleveland DW, Mao Y, Sullivan KF . (2003). Activating and silencing the mitotic checkpoint through CENP-E-dependent activation/inactivation of BubR1. Cell 114: 87–98.
DeLuca JG, Dong Y, Hergert P, Strauss J, Hickey JM, Salmon ED et al. (2005). Hec1 and nuf2 are core components of the kinetochore outer plate essential for organizing microtubule attachment sites. Mol Biol Cell 16: 519–531.
DeLuca JG, Gall WE, Ciferri C, Cimini D, Musacchio A, Salmon ED . (2006). Kinetochore microtubule dynamics and attachment stability are regulated by Hec1. Cell 127: 969–982.
Durfee T, Becherer K, Chen PL, Yeh SH, Yang Y, Kilburn AE et al. (1993). The retinoblastoma protein associates with the protein phosphatase type 1 catalytic subunit. Genes Dev 7: 555–569.
Fang L, Fang G . (2007). Centromere cohesion: regulating the guardian. Cell Res 17: 664–665.
Fry AM . (2002). The Nek2 protein kinase: a novel regulator of centrosome structure. Oncogene 21: 6184–6194.
Fu G, Ding X, Yuan K, Aikhionbare F, Yao J, Cai X et al. (2007). Phosphorylation of human Sgo1 by NEK2A is essential for chromosome congression in mitosis. Cell Res 17: 608–618.
Fujiwara T, Bandi M, Nitta M, Ivanova EV, Bronson RT, Pellman D . (2005). Cytokinesis failure generating tetraploids promotes tumorigenesis in p53-null cells. Nature 437: 1043–1047.
Hayward DG, Fry AM . (2006). Nek2 kinase in chromosome instability and cancer. Cancer Lett 237: 155–166.
Ke Y, Dou Z, Zhang J, Yao X . (2003). Function and regulation of Aurora/Ipl1p kinase family in cell division. Cell Res 13: 69–81.
Lampson MA, Kapoor TM . (2005). The human mitotic checkpoint protein BubR1 regulates chromosome-spindle attachments. Nat Cell Biol 7: 93–98.
Liu D, Ding X, Du J, Cai X, Huang Y, Ward T et al. (2007). Nuf2 interacts with CENP-E and is essential for a stable spindle microtubule-kinetochore attachment. J Biol Chem 282: 21415–21424.
Lou Y, Yao J, Zereshki A, Dou Z, Ahmed K, Wang H et al. (2004). NEK2A interacts with MAD1 and possibly functions as a novel integrator of the spindle checkpoint signaling. J Biol Chem 279: 20049–20057.
Musacchio A, Salmon ED . (2007). The spindle-assembly checkpoint in space and time. Nat Rev Mol Cell Biol 8: 379–393.
Nicklas RB . (1971). Mitosis. Adv Cell Biol 2: 225–297.
Nicklas RB, Kubai DF . (1985). Microtubules, chromosome movement, and reorientation after chromosomes are detached from the spindle by micromanipulation. Chromosoma 92: 313–324.
Nigg EA . (2001). Mitotic kinases as regulators of cell division and its checkpoints. Nat Rev Mol Cell Biol 2: 21–32.
Storchova Z, Breneman A, Cande J, Dunn J, Burbank K, O'Toole E et al. (2006). Genome-wide genetic analysis of polyploidy in yeast. Nature 443: 541–547.
Yao X, Abrieu A, Zheng Y, Sullivan KF, Cleveland DW . (2000). CENP-E forms a link between attachment of spindle microtubules to kinetochores and the mitotic checkpoint. Nat Cell Biol 2: 484–491.
Acknowledgements
We would like to thank Drs W-H Lee and A Musacchio for gift reagents. This work was supported by grants from Chinese 973 project (2002CB713700, 2007CB914503), Chinese Academy of Sciences (KSCX1-YW-R65 and KSCX2-YW-H10), Chinese 863 project (2001AA215331 and), Chinese Ministry of Education (20020358051; 20050358061 and 111 project B07007 to XD), Chinese Natural Science Foundation (90508002, 30121001 to XY; 30500183 to XD; 30570850 to JZ; 30600222 to JY), National Institutes of Health (DK56292, CA132389, CA89019, and CA92080) and a GCC Breast Cancer Research Grant to XY. The facilities were supported in part by NIH/NCRR/RCM1 Grant G-12-RR03034. SER and XY are GCC eminent scholars.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Supplementary Information accompanies the paper on the Oncogene website (http://www.nature.com/onc)
Supplementary information
Rights and permissions
About this article
Cite this article
Du, J., Cai, X., Yao, J. et al. The mitotic checkpoint kinase NEK2A regulates kinetochore microtubule attachment stability. Oncogene 27, 4107–4114 (2008). https://doi.org/10.1038/onc.2008.34
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2008.34
Keywords
This article is cited by
-
Inhibition of the YAP-MMB interaction and targeting NEK2 as potential therapeutic strategies for YAP-driven cancers
Oncogene (2024)
-
NEK2 inhibition triggers anti-pancreatic cancer immunity by targeting PD-L1
Nature Communications (2021)
-
Role of Nek2 on centrosome duplication and aneuploidy in breast cancer cells
Oncogene (2014)
-
Mitosis as an anti-cancer drug target
Chromosoma (2013)
-
Regulatory mechanisms of kinetochore–microtubule interaction in mitosis
Cellular and Molecular Life Sciences (2013)