Abstract
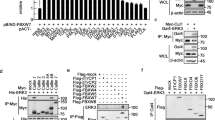
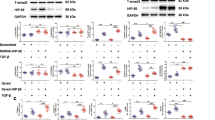
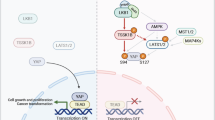
Transforming growth factor-beta (TGF-β) elicits a variety of cellular activities primarily through a signaling cascade mediated by two key transcription factors, Smad2 and Smad3. Numerous regulatory mechanisms exist to control the activity of Smad3, thereby modulating the strength and specificity of TGF-β responses. In search for potential regulators of Smad3 through a yeast two-hybrid screen, we identified casein kinase 1 gamma 2 (CKIγ2) as a novel Smad3-interacting protein. In mammalian cells, CKIγ2 selectively and constitutively binds Smad3 but not Smad1, -2 or -4. Functionally, CKIγ2 inhibits Smad3-mediated TGF-β responses including induction of target genes and cell growth arrest, and this inhibition is dependent on CKIγ2 kinase activity. Mechanistically, CKIγ2 does not affect the basal levels of Smad proteins or activity of the receptors. Rather, CKIγ2 preferentially promotes the ubiquitination and degradation of activated Smad3 through direct phosphorylation of its MH2 domain at Ser418. Importantly, mutation of Ser418 to alanine or aspartic acid causes an increase or decrease of Smad3 activity, respectively, in the presence of TGF-β. CKIγ2 is the first kinase known to mark activated Smad3 for destruction. Given its negative function in TGF-β signaling and its reported overexpression in human cancers, CKIγ2 may act as an oncoprotein during tumorigenesis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Bierie B, Moses HL . (2006). Tumour microenvironment: TGFbeta: the molecular Jekyll and Hyde of cancer. Nat Rev Cancer 6: 506–520.
Davidson G, Wu W, Shen J, Bilic J, Fenger U, Stannek P et al. (2005). Casein kinase 1 gamma couples Wnt receptor activation to cytoplasmic signal transduction. Nature 438: 867–872.
Dennler S, Huet S, Gauthier JM . (1999). A short amino-acid sequence in MH1 domain is responsible for functional differences between Smad2 and Smad3. Oncogene 18: 1643–1648.
Dunn NR, Koonce CH, Anderson DC, Islam A, Bikoff EK, Robertson EJ . (2005). Mice exclusively expressing the short isoform of Smad2 develop normally and are viable and fertile. Genes Dev 19: 152–163.
Fukuchi M, Imamura T, Chiba T, Ebisawa T, Kawabata M, Tanaka K et al. (2001). Ligand-dependent degradation of Smad3 by a ubiquitin ligase complex of ROC1 and associated proteins. Mol Biol Cell 12: 1431–1443.
Gross SD, Anderson RA . (1998). Casein kinase I: spatial organization and positioning of a multifunctional protein kinase family. Cell Signal 10: 699–711.
Guo X, Ramirez A, Waddell DS, Li Z, Liu X, Wang X-F . (2008). Axin and GSK3-β control Smad3 protein stability and modulate TGF-β signaling. Genes Dev 22: 106–120.
Heldin C-H, Miyazono K, Dijke PT . (1997). TGF-β signalling from cell membrane to nucleus through SMAD proteins. Nature 390: 465.
Heldin CH, ten Dijke P . (1999). SMAD destruction turns off signalling. Nat Cell Biol 1: E195–E197.
Imoto S, Sugiyama K, Sekine Y, Matsuda T . (2005). Roles for lysine residues of the MH2 domain of Smad3 in transforming growth factor-beta signaling. FEBS Lett 579: 2853–2862.
Inman GJ, Nicolas FJ, Hill CS . (2002). Nucleocytoplasmic shuttling of Smads 2, 3, and 4 permits sensing of TGF-beta receptor activity. Mol Cell 10: 283–294.
Izzi L, Attisano L . (2006). Ubiquitin-dependent regulation of TGF signaling in cancer. Neoplasia 8: 677–688.
Jayaraman L, Massagué J . (2000). Distinct oligomeric states of SMAD proteins in the transforming growth factor-beta pathway. J Biol Chem 275: 40710–40717.
Kitabayashi AN, Kusuda J, Hirai M, Hashimoto K . (1997). Cloning and chromosomal mapping of human casein kinase I gamma 2 (CSNK1G2). Genomics 46: 133–137.
Knippschild U, Gocht A, Wolff S, Huber N, Lohler J, Stoter M . (2005). The casein kinase 1 family: participation in multiple cellular processes in eukaryotes. Cell Signal 17: 675–689.
Komuro A, Imamura T, Saitoh M, Yoshida Y, Yamori T, Miyazono K et al. (2004). Negative regulation of transforming growth factor-[beta] (TGF-[beta]) signaling by WW domain-containing protein 1 (WWP1). Oncogene 23: 6914–6923.
Kretschmer A, Moepert K, Dames S, Sternberger M, Kaufmann J, Klippel A . (2003). Differential regulation of TGF-beta signaling through Smad2, Smad3 and Smad4. Oncogene 22: 6748–6763.
Liberati NT, Datto MB, Frederick JP, Shen X, Wong C, Rougier-Chapman EM et al. (1999). Smads bind directly to the Jun family of AP-1 transcription factors. Proc Natl Acad Sci USA 96: 4844–4849.
Liberati NT, Moniwa M, Borton AJ, Davie JR, Wang XF . (2001). An essential role for Mad homology domain 1 in the association of Smad3 with histone deacetylase activity. J Biol Chem 276: 22595–22603.
Lin X, Duan X, Liang YY, Su Y, Wrighton KH, Long J et al. (2006). PPM1A functions as a Smad phosphatase to terminate TGFbeta signaling. Cell 125: 915–928.
Lin X, Liang M, Feng X-H . (2000). Smurf2 is a ubiquitin E3 ligase mediating proteasome-dependent degradation of Smad2 in transforming growth factor-beta signaling. J Biol Chem 275: 36818–36822.
Liu C, Li Y, Semenov M, Han C, Baeg G-H, Tan Y et al. (2002). Control of β-catenin phosphorylation/degradation by a dual-kinase mechanism. Cell 108: 837–847.
Lo RS, Massagué J . (1999). Ubiquitin-dependent degradation of TGF-beta-activated Smad2. Nat Cell Biol 1: 472–478.
Long J, Wang G, Matsuura I, He D, Liu F . (2004). Activation of Smad transcriptional activity by protein inhibitor of activated STAT3 (PIAS3). Proc Natl Acad Sci USA 101: 99–104.
Massagué J . (2000). How cells read TGF-beta signals. Nat Rev Mol Cell Biol 1: 169–178.
Massagué J, Blain SW, Lo RS . (2000). TGFβ signaling in growth control, cancer, and heritable disorders. Cell 103: 295–309.
Massagué J, Seoane J, Wotton D . (2005). Smad transcription factors. Genes Dev 19: 2783–2810.
Peters JM, McKay RM, McKay JP, Graff JM . (1999). Casein kinase I transduces Wnt signals. Nature 401: 345–350.
Piek E, Ju WJ, Heyer J, Escalante-Alcalde D, Stewart CL, Weinstein M et al. (2001). Functional characterization of transforming growth factor beta signaling in Smad2- and Smad3-deficient fibroblasts. J Biol Chem 276: 19945–19953.
Poncelet A-C, Schnaper HW, Tan R, Liu Y, Runyan CE . (2007). Cell phenotype-specific down-regulation of Smad3 involves decreased gene activation as well as protein degradation. J Biol Chem 282: 15534–15540.
Price MA . (2006). CKI, there's more than one: casein kinase I family members in Wnt and Hedgehog signaling. Genes Dev 20: 399–410.
Price MA, Kalderon D . (2002). Proteolysis of the Hedgehog signaling effector Cubitus interruptus requires phosphorylation by Glycogen Synthase Kinase 3 and Casein Kinase 1. Cell 108: 823–835.
Reguly T, Wrana JL . (2003). In or out? The dynamics of Smad nucleocytoplasmic shuttling. Trends Cell Biol 13: 216–220.
Roberts AB, Tian F, Byfield SD, Stuelten C, Ooshima A, Saika S et al. (2006). Smad3 is key to TGF-[beta]-mediated epithelial-to-mesenchymal transition, fibrosis, tumor suppression and metastasis. Cytokine Growth Factor Rev 17: 19–27.
Schilling SH, Datto MB, Wang XF . (2006). A phosphatase controls the fate of receptor-regulated Smads. Cell 125: 838–840.
Shi Y, Massagué J . (2003). Mechanisms of TGF-beta signaling from cell membrane to the nucleus. Cell 113: 685–700.
Shi Y, Wang YF, Jayaraman L, Yang H, Massagué J, Pavletich NP . (1998). Crystal structure of a Smad MH1 domain bound to DNA: insights on DNA binding in TGF-beta signaling. Cell 94: 585–594.
Schmierer B, Hill CS . (2005). Kinetic analysis of Smad nucleocytoplasmic shuttling reveals a mechanism for transforming growth factor beta-dependent nuclear accumulation of Smads. Mol Cell Biol 25: 9845–9858.
Vielhaber E, Virshup DM . (2001). Casein kinase I: from obscurity to center stage. IUBMB Life 51: 73–78.
Waddell DS, Liberati NT, Guo X, Frederick JP, Wang X-F . (2004). Casein kinase Iɛ plays a functional role in the transforming growth factor-β signaling pathway. J Biol Chem 279: 29236–29246.
Wang W, Koka V, Lan HY . (2005). Transforming growth factor-β and Smad signalling in kidney diseases. Review article. Nephrology 10: 48–56.
Weinstein M, Yang X, Deng C . (2000). Functions of mammalian Smad genes as revealed by targeted gene disruption in mice. Cytokine Growth Factor Rev 11: 49–58.
Xu L, Kang Y, Col S, Massagué J . (2002). Smad2 nucleocytoplasmic shuttling by nucleoporins CAN/Nup214 and Nup153 feeds TGFβ signaling complexes in the cytoplasm and nucleus. Mol Cell 10: 271–282.
Yagi K, Goto D, Hamamoto T, Takenoshita S, Kato M, Miyazono K . (1999). Alternatively spliced variant of Smad2 lacking exon 3. Comparison with wild-type Smad2 and Smad3. J Biol Chem 274: 703–709.
Zeng X, Tamai K, Doble B, Li S, Huang H, Habas R et al. (2005). A dual-kinase mechanism for Wnt co-receptor phosphorylation and activation. Nature 438: 873–877.
Zhai L, Graves PR, Robinson LC, Italiano M, Culbertson MR, Rowles J et al. (1995). Casein kinase I gamma subfamily. Molecular cloning, expression, and characterization of three mammalian isoforms and complementation of defects in the Saccharomyces cerevisiae YCK genes. J Biol Chem 270: 12717–12724.
Acknowledgements
We thank Drs Jun Kusuda, Joan Massagué, Xin-Hua Feng, Rik Derynck, Jun-Lin Guan, James Woodgett and Anita Roberts for valuable reagents. We appreciate the Wang laboratory members for insightful scientific discussions and excellent technical support. We thank Natalie Ahn, Kathryn Resing and Will Old for MS facility and support. This work was supported by NIH grants DK064113 and GM083000 to X-F W, and an NIH Grant GM083172 to XL. DSW was supported by Department of Defense Breast Cancer Predoctoral Fellowship DAMD17-00-1-0299. NTL was supported by a National Science Foundation Predoctoral Fellowship.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary Information accompanies the paper on the Oncogene website (http://www.nature.com/onc)
Supplementary information
Rights and permissions
About this article
Cite this article
Guo, X., Waddell, D., Wang, W. et al. Ligand-dependent ubiquitination of Smad3 is regulated by casein kinase 1 gamma 2, an inhibitor of TGF-β signaling. Oncogene 27, 7235–7247 (2008). https://doi.org/10.1038/onc.2008.337
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2008.337
Keywords
This article is cited by
-
Adriamycin induces cardiac fibrosis in mice via PRMT5-mediated cardiac fibroblast activation
Acta Pharmacologica Sinica (2023)
-
miR-615-3p promotes the epithelial-mesenchymal transition and metastasis of breast cancer by targeting PICK1/TGFBRI axis
Journal of Experimental & Clinical Cancer Research (2020)
-
Blockade of Smad signaling by 3′-deoxyadenosine: a mechanism for its anti-fibrotic potential
Laboratory Investigation (2013)
-
Role of Smads in TGFβ signaling
Cell and Tissue Research (2012)
-
Phospho-control of TGF-β superfamily signaling
Cell Research (2009)