Abstract
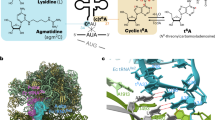
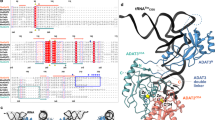
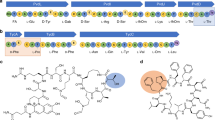
We report the crystal structure of an archaea-specific editing domain of threonyl-tRNA synthetase that reveals a marked structural similarity to D-amino acid deacylases found in eubacteria and eukaryotes. The domain can bind D-amino acids despite a low sequence identity to other D-amino acid deacylases. These results together indicate the presence of these deacylases in all three kingdoms of life. This underlines an important role they may have played in enforcing homochirality during translation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Miller, S.L. & Orgel, L.E. The Origin of Life on Earth (Prentice Hall, River Edge, New Jersey, USA, 1974).
Martyn Bailey, J. FASEB J. 12, 503–507 (1998).
Tamura, K. & Schimmel, P. Science 305, 1253 (2004).
Schimmel, P. & Söll, D. Annu. Rev. Biochem. 48, 601–648 (1979).
Sankaranarayanan, R. et al. Nat. Struct. Biol. 7, 461–465 (2000).
Dock-Bregeon, A.C. et al. Cell 103, 877–884 (2000).
Beebe, K., Merriman, E., Pouplana, L.R. & Schimmel, P. Proc. Natl. Acad. Sci. USA 101, 5958–5963 (2004).
Korencic, D. et al. Proc. Natl. Acad. Sci. USA 101, 10260–10265 (2004).
Ferri-Fioni, M-L. et al. J. Biol. Chem. 276, 47285–47290 (2001).
Rigden, D.J. RNA 10, 1845–1851 (2004).
Soutourina, J., Plateau, P. & Blanquet, S. J. Biol. Chem. 275, 32535–32542 (2000).
Agmon, I. et al. FEBS Lett. 567, 20–26 (2004).
Milton, R.C., Milton, S.C.F. & Kent, S.B.H. Science 256, 1445–1448 (1992).
Calendar, R. & Berg, P. Biochemistry 5, 1681–1690 (1966).
Soutourina, J., Plateau, P., Delort, F., Peirotes, A. & Blanquet, S. J. Biol. Chem. 274, 19109–19114 (1999).
Soutourina, O., Soutourina, J., Blanquet, S. & Plateau, P. J. Biol. Chem. 279, 42560–42565 (2004).
Acknowledgements
S.D. thanks the Council of Scientific and Industrial Research (CSIR), India, for a senior research fellowship. We thank V.M. Shanmugam and Y. Sharma for help during X-ray data collection and fluorescence experiments, respectively. We thank D. Moras and his lab members, particularly A.-C. Dock-Bregeon, from Institut de Génétique et de Biologie Moléculaire et Cellulaire, Strasbourg, for helpful discussions and providing Pyrococcus abyssi genomic DNA. R.S. is an international senior research fellow of The Wellcome Trust, UK, in Biomedical Science in India.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Fig. 1
Structural superposition of Pab-NTD (cyan and yellow) and DTD (pink and blue) dimers as observed in their respective crystal structures. (PDF 538 kb)
Supplementary Fig. 2
Amino acid binding studies using Bis-ANS fluorescence. (PDF 551 kb)
Supplementary Fig. 3
Crystal soaks with different amino acids. (PDF 509 kb)
Supplementary Fig. 4
Structure-based sequence alignment of the editing domain of a few archaeal ThrRSs along with DTD homologs. (PDF 904 kb)
Supplementary Table 1
Summary of crystallographic data and refinement statistics. (PDF 67 kb)
Rights and permissions
About this article
Cite this article
Dwivedi, S., Kruparani, S. & Sankaranarayanan, R. A D-amino acid editing module coupled to the translational apparatus in archaea. Nat Struct Mol Biol 12, 556–557 (2005). https://doi.org/10.1038/nsmb943
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nsmb943
This article is cited by
-
A chiral selectivity relaxed paralog of DTD for proofreading tRNA mischarging in Animalia
Nature Communications (2018)
-
Specificity and catalysis hardwired at the RNA–protein interface in a translational proofreading enzyme
Nature Communications (2015)
-
A genomic glimpse of aminoacyl-tRNA synthetases in malaria parasite Plasmodium falciparum
BMC Genomics (2009)
-
Post-transfer editing mechanism of a D-aminoacyl-tRNA deacylase-like domain in threonyl-tRNA synthetase from archaea
The EMBO Journal (2006)