Abstract
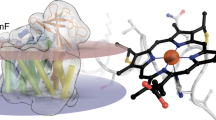
We have isolated a soluble cytochrome from Shewanella oneidensis that contains eight covalently attached heme groups and determined its crystal structure. One of these hemes exhibits novel ligation of the iron atom by the ε-amino group of a lysine residue, despite its attachment via a typical CXXCH motif. This heme is most likely the active site for tetrathionate reduction, a reaction catalyzed efficiently by this enzyme.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Heidelberg, J.F. et al. Nat. Biotechnol. 20, 1118–1123 (2002).
Gordon, E.H.J., Pealing, S.L., Chapman, S.K., Ward, F.B. & Reid, G.A. Microbiology 144, 937–945 (1998).
Lo Conte, L., Brenner, S.E., Hubbard, T.J.P., Chothia, C. & Murzin, A. Nucleic Acids Res. 30, 264–267 (2002).
Einsle, O. et al. J. Biol. Chem. 275, 39608–39616 (2000).
Simon, J. FEMS Microbiol. Rev. 26, 285–309 (2002).
Hensel, M., Hinsley, A.P., Nikolaus, T., Sawers, G. & Berks, B.C. Mol. Microbiol. 32, 275–287 (1999).
Claiborne, A., Mallett, T.C., Yeh, J.I., Luba, J. & Parsonage, D. Adv. Protein Chem. 58, 215–276 (2001).
Bamford V.A. et al. EMBO J. 21, 5599–5610 (2002).
Igarashi, N., Moriyama, H., Fujiwara, T., Fukumori, Y. & Tanaka, N. Nat. Struct. Biol. 4, 276–284 (1997).
Acknowledgements
We thank the UK Biotechnology and Biological Sciences Research Council for grant funding to G.A.R. and S.K.C. and the Wellcome Trust for their support to the Edinburgh Protein Interaction Centre. Synchrotron access at European Molecular Biology Laboratory Hamburg was supported by the European Commission (contract HPRI-CT-1999-00017).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Table
Data collection, phasing and refinement statistics for OTR (PDF 18 kb)
Rights and permissions
About this article
Cite this article
Mowat, C., Rothery, E., Miles, C. et al. Octaheme tetrathionate reductase is a respiratory enzyme with novel heme ligation. Nat Struct Mol Biol 11, 1023–1024 (2004). https://doi.org/10.1038/nsmb827
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nsmb827
This article is cited by
-
Microbial communities of Auka hydrothermal sediments shed light on vent biogeography and the evolutionary history of thermophily
The ISME Journal (2022)
-
Nature's nitrite-to-ammonia expressway, with no stop at dinitrogen
JBIC Journal of Biological Inorganic Chemistry (2022)
-
Novel taxa of Acidobacteriota implicated in seafloor sulfur cycling
The ISME Journal (2021)
-
Unprecedented Microbial Conversion of Biliverdin into Bilirubin-10-sulfonate
Scientific Reports (2019)
-
Ein altes Paar in neuem Glanz: Thiosulfat und Tetrathionat
BIOspektrum (2017)